Question Bank for Pre Board Exam(XII Chemistry)
... 37.In Corundum, O2– ions from hcp and Al3+ occupy two third of octahedral voids. Determine the formula of corundum. 38.Why is Frenkel defect not found in pure alkali metal halides? 39.Which point defect is observed in a crystal when a vacancy is created by an atom missing from a lattice site. 40. Wh ...
... 37.In Corundum, O2– ions from hcp and Al3+ occupy two third of octahedral voids. Determine the formula of corundum. 38.Why is Frenkel defect not found in pure alkali metal halides? 39.Which point defect is observed in a crystal when a vacancy is created by an atom missing from a lattice site. 40. Wh ...
Visible Light Photoredox Catalysis with Transition
... light in the visible region to give excited states capable of singleelectron transfer. These catalysts have been employed to achieve a vast range of bond-forming reactions of broad utility in organic synthesis.16 Visible light photocatalysis has also been carried out with heterogeneous semiconductor ...
... light in the visible region to give excited states capable of singleelectron transfer. These catalysts have been employed to achieve a vast range of bond-forming reactions of broad utility in organic synthesis.16 Visible light photocatalysis has also been carried out with heterogeneous semiconductor ...
Ether C-O Bond Cleavage w
... No β-hydride elimination products observed ◦ Transmetalation occurs rapidly ...
... No β-hydride elimination products observed ◦ Transmetalation occurs rapidly ...
metal ions in solution
... M2+ solutions are very weakly acidic (pH about 6) due to the following equilibrium: [M(H2O)6]2+ + H2O ⇋ [M(H2O)5OH]+ + H3O+ ...
... M2+ solutions are very weakly acidic (pH about 6) due to the following equilibrium: [M(H2O)6]2+ + H2O ⇋ [M(H2O)5OH]+ + H3O+ ...
- University at Albany
... Similar to that of alkenes, except the ending of the root name corresponding to the longest continuous chain containing the triple bond is changed from “ene” to “yne”. E.g. ethene becomes ethyne, propene becomes propyne. The chain is numbered from the end closest to the triple bond. When a double an ...
... Similar to that of alkenes, except the ending of the root name corresponding to the longest continuous chain containing the triple bond is changed from “ene” to “yne”. E.g. ethene becomes ethyne, propene becomes propyne. The chain is numbered from the end closest to the triple bond. When a double an ...
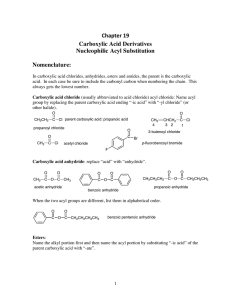
Chapter 19 Carboxylic Acid Derivatives Nucleophilic Acyl
... After acid chlorides, the next most reactive derivatives are the anhydrides. They can be used to form the esters and amides and are also subject to hydrolysis. It is best to think of acid chlorides and anhydrides as reagents used for the preparation of the more stable end products, the esters and th ...
... After acid chlorides, the next most reactive derivatives are the anhydrides. They can be used to form the esters and amides and are also subject to hydrolysis. It is best to think of acid chlorides and anhydrides as reagents used for the preparation of the more stable end products, the esters and th ...
Caboxylic acid Derivatives
... Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are esters, amides and nitriles, although acid halides and anhydrides are also derivatives (really activated forms of a carboxyl ...
... Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are esters, amides and nitriles, although acid halides and anhydrides are also derivatives (really activated forms of a carboxyl ...
Simultaneous decolorization of ternary dye mixture from aqueous
... destabilized and they start attracting each other and generate bigger particles. EC process depends on pH, particle size, concentrations, and the conductivity of the continuous phase. These experimental factors affect the results in different ways. Electrocoagulation has some advantages including si ...
... destabilized and they start attracting each other and generate bigger particles. EC process depends on pH, particle size, concentrations, and the conductivity of the continuous phase. These experimental factors affect the results in different ways. Electrocoagulation has some advantages including si ...
APPROACHES TO CARBOHYDRATE-BASED CHEMICAL LIBRARIES: THE
... Although having been somewhat less than blissfully content with my surroundings in Cambridge, I have thoroughly enjoyed my five years as a graduate student. My lab has always been a very positive environment, and much of the credit for that should go to Peter Lansbury. He has always gone to extremes ...
... Although having been somewhat less than blissfully content with my surroundings in Cambridge, I have thoroughly enjoyed my five years as a graduate student. My lab has always been a very positive environment, and much of the credit for that should go to Peter Lansbury. He has always gone to extremes ...
From Kinetics to Equilibrium
... salt and wet snow somewhat increases the rate of the reaction. Even so, it takes several years for a significant amount of rust to form on the body of a car. This is a good thing for car owners—if rusting occurred as fast as the reaction in an inflating air bag, cars would flake to pieces in seconds ...
... salt and wet snow somewhat increases the rate of the reaction. Even so, it takes several years for a significant amount of rust to form on the body of a car. This is a good thing for car owners—if rusting occurred as fast as the reaction in an inflating air bag, cars would flake to pieces in seconds ...
Small Reactive Sulfur-Nitrogen Compounds and Their Transition
... ~ ~ , ~ ~ development of the field was certainly hampered by the fact that the free ligand is not available under normal conditions, and indirect methods are required to build the thionitrosyl ligand within the coordination sphere, e.g., by sulfurization of nitrido-metal complexes or by halide abstr ...
... ~ ~ , ~ ~ development of the field was certainly hampered by the fact that the free ligand is not available under normal conditions, and indirect methods are required to build the thionitrosyl ligand within the coordination sphere, e.g., by sulfurization of nitrido-metal complexes or by halide abstr ...
History of Organic Chemistry
... Confused, the student tells the naive chemistry-challenged person that the task is impossible. a) Why can=t the student show the structure of C4H10? (2) b) Rephrase the question by changing one word so that the question can be answered, and then answer it. (Keep the flavor of the question - don't ch ...
... Confused, the student tells the naive chemistry-challenged person that the task is impossible. a) Why can=t the student show the structure of C4H10? (2) b) Rephrase the question by changing one word so that the question can be answered, and then answer it. (Keep the flavor of the question - don't ch ...
Chemistry 30 – Organic Chemistry
... • polar but lack of OH bond means no hydrogen bonding, so lower boiling points than alcohols and carboxylic acids • esters with few carbons are polar enough to be soluble in water ...
... • polar but lack of OH bond means no hydrogen bonding, so lower boiling points than alcohols and carboxylic acids • esters with few carbons are polar enough to be soluble in water ...
Document
... to chemical reaction • they do not react with oxidizing agents such as potassium dichromate • they do not react with reducing agents such as H2 in the presence of a transition metal catalyst • they are not affected by most acids or bases at moderate temperatures • Because of their general inertness ...
... to chemical reaction • they do not react with oxidizing agents such as potassium dichromate • they do not react with reducing agents such as H2 in the presence of a transition metal catalyst • they are not affected by most acids or bases at moderate temperatures • Because of their general inertness ...
$doc.title
... calculations 65 seem to indicate accumulation of negative charge on the 2- and 5-carbon atoms and a positive charge on the sulfur atom. Coordination can occur through the sulfur atom (8), through the 2- or 5-carbon or both (r] 1) or through the n-clouds of the C(2)=C(3) or C(4)=C(5) bonds (fl2) or b ...
... calculations 65 seem to indicate accumulation of negative charge on the 2- and 5-carbon atoms and a positive charge on the sulfur atom. Coordination can occur through the sulfur atom (8), through the 2- or 5-carbon or both (r] 1) or through the n-clouds of the C(2)=C(3) or C(4)=C(5) bonds (fl2) or b ...
Two-electron Quenching of Dinuclear Ruthenium(II)
... The excited state decays involve several processes (Fig 1.4). By irradiating Ru complexes, for example [Ru(bpy)3]2+, at the appropriate wavelength, a lowest photoexcited singlet metal-toligand charge transfer state (1MLCT) is produced, 1[Ru(bpy)3]2+* , which then undergoes intersystem crossing (kis ...
... The excited state decays involve several processes (Fig 1.4). By irradiating Ru complexes, for example [Ru(bpy)3]2+, at the appropriate wavelength, a lowest photoexcited singlet metal-toligand charge transfer state (1MLCT) is produced, 1[Ru(bpy)3]2+* , which then undergoes intersystem crossing (kis ...
THE SYNTHESIS AND CHARACTERIZATION OF PHOSPHONIUM INDENYLIDE COMPLEXES OF RUTHENIUM(II)
... Figure 10. Transition metal complexes of phosphonium cyclopentadienylides which have been charactereized by X-ray crystallography (excludes complexes of group 8 metals) ........................ 9 Figure 11. Synthesis of (C5H4PPh3)M(CO)3 (M = Cr, Mo, W) ............................................... ...
... Figure 10. Transition metal complexes of phosphonium cyclopentadienylides which have been charactereized by X-ray crystallography (excludes complexes of group 8 metals) ........................ 9 Figure 11. Synthesis of (C5H4PPh3)M(CO)3 (M = Cr, Mo, W) ............................................... ...
Electroless Deposition of Copper
... reaction (17.1) and the partial anodic reaction (17.7) and thus influence these reactions in a direct way (primary pH effects). Second, pH affects various phenomena associated with the structure and composition of the metal–solution interphase [14]. Those phenomena include (1) adsorption, (2) the st ...
... reaction (17.1) and the partial anodic reaction (17.7) and thus influence these reactions in a direct way (primary pH effects). Second, pH affects various phenomena associated with the structure and composition of the metal–solution interphase [14]. Those phenomena include (1) adsorption, (2) the st ...
Rubidium
... Rubidium is not made by the same method as sodium as might have been expected. This is because the rubidium metal, once formed by electrolysis of liquid rubidium chloride (RbCl), is too soluble in the molten salt. cathode: Rb+(l) + e- Rb (l) anode: Cl-(l) 1/2Cl2 (g) + e- ...
... Rubidium is not made by the same method as sodium as might have been expected. This is because the rubidium metal, once formed by electrolysis of liquid rubidium chloride (RbCl), is too soluble in the molten salt. cathode: Rb+(l) + e- Rb (l) anode: Cl-(l) 1/2Cl2 (g) + e- ...
Unit 2
... H CH3 H CH3 H H ethene or ethylene propene or propylene 1-butene 2-butene Members of the alkene family also have distinct chemical and physical properties. They have lower boiling points than their respective alkanes because there are less hydrogen atoms present to form intermolecular bonds. For nam ...
... H CH3 H CH3 H H ethene or ethylene propene or propylene 1-butene 2-butene Members of the alkene family also have distinct chemical and physical properties. They have lower boiling points than their respective alkanes because there are less hydrogen atoms present to form intermolecular bonds. For nam ...
CH 2 CH(CH 3 ) - Parkway C-2
... Always include both the double and triple bond in the longest chain – even if it isn’t the most number of carbons! Start counting from the end closest to the double or triple bond – whichever has the lowest number If there is a tie, and only then, do double bonds take priority over triple ...
... Always include both the double and triple bond in the longest chain – even if it isn’t the most number of carbons! Start counting from the end closest to the double or triple bond – whichever has the lowest number If there is a tie, and only then, do double bonds take priority over triple ...
Naming of Alkanes
... Have the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: Have fewer hydrogen atoms attached to the carbon chain than alkanes. • Containing double bond are alkenes. ...
... Have the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: Have fewer hydrogen atoms attached to the carbon chain than alkanes. • Containing double bond are alkenes. ...
Syntheses, spectral characterization, thermal properties and DNA
... ligands L1/L2 or L3 to give the corresponding metal complexes. The elemental analysis data for ligands and complexes is given in Table.1. All the compounds show the analytical results close to the theoretical values indicating the presence of two types of ligands. 3.2. IR Spectra The IR spectral dat ...
... ligands L1/L2 or L3 to give the corresponding metal complexes. The elemental analysis data for ligands and complexes is given in Table.1. All the compounds show the analytical results close to the theoretical values indicating the presence of two types of ligands. 3.2. IR Spectra The IR spectral dat ...
Organic_1_6.1ed_2012_02nd_module_functional_groups_and_IR
... An unknown compound having the formula C6H13N had a peak in its IR spectrum at 3350cm-1. Which of the following compounds is consistent with this? CH3 A. ...
... An unknown compound having the formula C6H13N had a peak in its IR spectrum at 3350cm-1. Which of the following compounds is consistent with this? CH3 A. ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.