WRL3502.tmp
... Relevant sections in the text: General Concepts Alkyl halides are most commonly synthesized from alcohols by replacing the hydroxyl group with a halide substituent. This is an example of nucleophilic aliphatic substitution, which is part of a very important group of reactions. The overall reaction i ...
... Relevant sections in the text: General Concepts Alkyl halides are most commonly synthesized from alcohols by replacing the hydroxyl group with a halide substituent. This is an example of nucleophilic aliphatic substitution, which is part of a very important group of reactions. The overall reaction i ...
9. E1: Alkenes from alcohols - Web Pages
... The substrate or starting material is cyclohexanol (R-OH). Sulfuric acid is present as a catalyst, which promotes the reaction but is not consumed in it. The hydroxyl group in R-OH is a poor-leaving group because it would have to leave as a hydroxide ion (HO-). Therefore, an acid is used to protona ...
... The substrate or starting material is cyclohexanol (R-OH). Sulfuric acid is present as a catalyst, which promotes the reaction but is not consumed in it. The hydroxyl group in R-OH is a poor-leaving group because it would have to leave as a hydroxide ion (HO-). Therefore, an acid is used to protona ...
7. Alkenes: Reactions and Synthesis
... Br+ adds to an alkene producing a cyclic ion Bromonium ion, bromine shares charge with carbon ...
... Br+ adds to an alkene producing a cyclic ion Bromonium ion, bromine shares charge with carbon ...
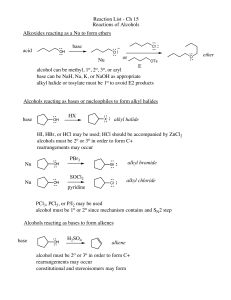
Reaction List - Ch 15 Reactions of Alcohols Alkoxides reacting as a
... alcohol can be methyl, 1o, 2o, 3o, or aryl base can be NaH, Na, K, or NaOH as appropriate alkyl halide or tosylate must be 1o to avoid E2 products Alcohols reacting as bases or nucleophiles to form alkyl halides base ...
... alcohol can be methyl, 1o, 2o, 3o, or aryl base can be NaH, Na, K, or NaOH as appropriate alkyl halide or tosylate must be 1o to avoid E2 products Alcohols reacting as bases or nucleophiles to form alkyl halides base ...
CHEMISTRY 3.5 Paper 1 Describe the structure and reactions of
... acidic conditions, the amino acid forms an ion that will move towards one electrode. In basic conditions, it forms another ion that will move towards the other electrode. Explain how the conditions described above give rise to two ions that will move towards the two different electrodes and state wh ...
... acidic conditions, the amino acid forms an ion that will move towards one electrode. In basic conditions, it forms another ion that will move towards the other electrode. Explain how the conditions described above give rise to two ions that will move towards the two different electrodes and state wh ...
Chapter 15
... • Aromatic hydrocarbons are known generally as arenes. • An Aryl group is formed by the removal of a hydrogen, and is symbolized with Ar• We have already talked about how stable the double bonds are in arenes due to conjugation. • They resist typical addition reactions ...
... • Aromatic hydrocarbons are known generally as arenes. • An Aryl group is formed by the removal of a hydrogen, and is symbolized with Ar• We have already talked about how stable the double bonds are in arenes due to conjugation. • They resist typical addition reactions ...
Chapter 20: Reactions of Complexes (Mechanisms)
... Often, trends can be obtained by “fine‐tuning” the ligand. Most useful are NR3 and PR3 ligands, where R groups are easily changed to vary the nucleophilicity of the donor atom without affecting the sterics too much. For systems where no intermediate is observable (an I mechanism), the rds may be ...
... Often, trends can be obtained by “fine‐tuning” the ligand. Most useful are NR3 and PR3 ligands, where R groups are easily changed to vary the nucleophilicity of the donor atom without affecting the sterics too much. For systems where no intermediate is observable (an I mechanism), the rds may be ...
Exam 2
... worth studying. Also problems assigned for the text may also be helpful. Chap 9 &10-- Sn2, Sn1, E2 and E1 reactions. -Know the definitions of Sn2, Sn1, E2 and E1. -Be able to depict the reaction coordinate diagrams of each reaction -Be able to draw the mechanism, products and the stereochemical resu ...
... worth studying. Also problems assigned for the text may also be helpful. Chap 9 &10-- Sn2, Sn1, E2 and E1 reactions. -Know the definitions of Sn2, Sn1, E2 and E1. -Be able to depict the reaction coordinate diagrams of each reaction -Be able to draw the mechanism, products and the stereochemical resu ...
Chemistry 322 Experiment #3 Data Sheet
... 5. In the first part of this experiment, you performed Lucas tests on several alcohols. a) Write a complete mechanism for the reaction occurring between the Lucas reagent and tbutanol. ...
... 5. In the first part of this experiment, you performed Lucas tests on several alcohols. a) Write a complete mechanism for the reaction occurring between the Lucas reagent and tbutanol. ...
Topic 16 Assessed Homework - A
... State what you would observe when propanoic acid reacts with this reagent. Reagent ............................................................................................. Observation ....................................................................................... ...
... State what you would observe when propanoic acid reacts with this reagent. Reagent ............................................................................................. Observation ....................................................................................... ...
ASYMMETRIC CATALYSIS
... strates, catalyst precursors, and metal-ligand complexes; and searching for appropriate reaction conditions. A clear understanding of catalytic mechanisms and characteristics of metal species from an inorganic chemistry perspective can provide insights into selecting chiral ligands, substrates, and ...
... strates, catalyst precursors, and metal-ligand complexes; and searching for appropriate reaction conditions. A clear understanding of catalytic mechanisms and characteristics of metal species from an inorganic chemistry perspective can provide insights into selecting chiral ligands, substrates, and ...
EXPERIMENT 3: Preparation and Reactivity of Alkyl Halides
... Although one might expect such a reaction to be reversible, it can be driven to formation of R-I by using anhydrous acetone as the solvent. Sodium iodide (NaI) is soluble in this solvent, but sodium chloride and sodium bromide are not. If a reaction occurs, a precipitate of sodium chloride or sodium ...
... Although one might expect such a reaction to be reversible, it can be driven to formation of R-I by using anhydrous acetone as the solvent. Sodium iodide (NaI) is soluble in this solvent, but sodium chloride and sodium bromide are not. If a reaction occurs, a precipitate of sodium chloride or sodium ...
Chemistry - Choithram School
... iii)Reaction of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in presence of alcoholic KOH , alkenes are the major products. iv)Chloroform is stored in closed dark coloured bottles completely filled so that air is kept out. v)Grignard’s reagent should be prepared under anhy ...
... iii)Reaction of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in presence of alcoholic KOH , alkenes are the major products. iv)Chloroform is stored in closed dark coloured bottles completely filled so that air is kept out. v)Grignard’s reagent should be prepared under anhy ...
Section 07 - Section Practice Exam II Solutions
... Thus, we would expect X groups that stabilize the generation of the positive charge to speed the reaction. This hypothesis is supported by the data for X=NO2 and X=Me. The nitro group is electron withdrawing and will destabilize the carbocation, explaining why it slows the reaction. The electron don ...
... Thus, we would expect X groups that stabilize the generation of the positive charge to speed the reaction. This hypothesis is supported by the data for X=NO2 and X=Me. The nitro group is electron withdrawing and will destabilize the carbocation, explaining why it slows the reaction. The electron don ...
Exam 3 Review Sheet
... You should feel comfortable with the following topics: Ch. 16 and 17: Aromatic Compounds • Concepts of aromaticity: MO diagrams, Hückel Rules for Aromaticity, resonance structures • Reactions at the benzylic position: o Addition of Br or Cl with NBS/NCS in the presence of peroxide. o Oxidation of a ...
... You should feel comfortable with the following topics: Ch. 16 and 17: Aromatic Compounds • Concepts of aromaticity: MO diagrams, Hückel Rules for Aromaticity, resonance structures • Reactions at the benzylic position: o Addition of Br or Cl with NBS/NCS in the presence of peroxide. o Oxidation of a ...
ch07 by Dr. Dina
... The hydroxyl is the group with higher priority and must be given the lowest possible number ...
... The hydroxyl is the group with higher priority and must be given the lowest possible number ...
Chem 263 Nov 3 2016 notes
... basic conditions is shown below. The first step involves the reversible attack of the nucleophile to form the alkoxide anion, which is then protonated. Most such reactions are done under acidic conditions, in which case the mechanism involves initial protonation of the oxygen. Under basic conditions ...
... basic conditions is shown below. The first step involves the reversible attack of the nucleophile to form the alkoxide anion, which is then protonated. Most such reactions are done under acidic conditions, in which case the mechanism involves initial protonation of the oxygen. Under basic conditions ...
Jan-Erling Bäckvall - The Scripps Research Institute
... (amides and sulfoxides are great ligands for Pd(II), and are compatible with oxidative conditions) ...
... (amides and sulfoxides are great ligands for Pd(II), and are compatible with oxidative conditions) ...
4.5: Bonding in Alcohols and Alkyl Halides
... 4.4: Classes of Alcohols and Alkyl Halides - Alcohols and alkyl halides are classified as according to the degree of substitution of the carbon bearing the halogen or -OH group OH primary (1°) : one alkyl substituent secondary (2°) : two alkyl substituents tertiary (3°) : three alkyl substituents 2- ...
... 4.4: Classes of Alcohols and Alkyl Halides - Alcohols and alkyl halides are classified as according to the degree of substitution of the carbon bearing the halogen or -OH group OH primary (1°) : one alkyl substituent secondary (2°) : two alkyl substituents tertiary (3°) : three alkyl substituents 2- ...
today`s PowerPoint
... Syllabus points 4.1.2 a, b, e Aldehydes and Ketones; identifying their presence using Brady’s reagent (2,4-DNPH) ...
... Syllabus points 4.1.2 a, b, e Aldehydes and Ketones; identifying their presence using Brady’s reagent (2,4-DNPH) ...
Ether And Epoxides
... This reaction is not carried out with moist Ag2O because moist Ag2O is actually AgOH where substitution occurs and formation of alcohols from alkyl halide takes place. Chemical properties (i) Oxidation Ether are less reactive due to absence of polarity, along with an ability to soluble in nonpolar s ...
... This reaction is not carried out with moist Ag2O because moist Ag2O is actually AgOH where substitution occurs and formation of alcohols from alkyl halide takes place. Chemical properties (i) Oxidation Ether are less reactive due to absence of polarity, along with an ability to soluble in nonpolar s ...
Brominations and Alkene Synthesis CHM 233 Review
... look from here, looks like this • ANTI addition, bromines add to opposite sides of the C=C double bond (top and bottom) • note new type of structure for indicating stereochemistry on rings, trans- in this case • NOTE, because of bromonium ion intermediate (no "free" carbocation) there are no rearran ...
... look from here, looks like this • ANTI addition, bromines add to opposite sides of the C=C double bond (top and bottom) • note new type of structure for indicating stereochemistry on rings, trans- in this case • NOTE, because of bromonium ion intermediate (no "free" carbocation) there are no rearran ...
CHM 331 : General Organic Chemistry
... look from here, looks like this • ANTI addition, bromines add to opposite sides of the C=C double bond (top and bottom) • note new type of structure for indicating stereochemistry on rings, trans- in this case • NOTE, because of bromonium ion intermediate (no "free" carbocation) there are no rearran ...
... look from here, looks like this • ANTI addition, bromines add to opposite sides of the C=C double bond (top and bottom) • note new type of structure for indicating stereochemistry on rings, trans- in this case • NOTE, because of bromonium ion intermediate (no "free" carbocation) there are no rearran ...
Sn1 and Sn2 Chemtivity
... Because the rate determining step of an SN1 reaction is the dissociation of the alkyl halide to form a carbocation, two factors affect the rate of an SN1 reaction – the ease with which the leaving group dissociates from the carbon and the stability of the carbocation that is formed. In the previous ...
... Because the rate determining step of an SN1 reaction is the dissociation of the alkyl halide to form a carbocation, two factors affect the rate of an SN1 reaction – the ease with which the leaving group dissociates from the carbon and the stability of the carbocation that is formed. In the previous ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.