Glencoe Chapter 4 Structure of the Atom for the Wiki
... Atomists and Democritus : 400 B.C. From Thrace in Greece. Atoms- “Uncut-Table” Indivisible parts which cannot be broken down further. Aristotle proposed that matter was continuous and was not made up of smaller particles. ...
... Atomists and Democritus : 400 B.C. From Thrace in Greece. Atoms- “Uncut-Table” Indivisible parts which cannot be broken down further. Aristotle proposed that matter was continuous and was not made up of smaller particles. ...
Chemistry Standards Checklist
... analyzing data and developing reasonable scientific explanations. a. Trace the source on any large disparity between estimated and calculated answers to problems. b. Consider possible effects of measurement errors on calculations. ...
... analyzing data and developing reasonable scientific explanations. a. Trace the source on any large disparity between estimated and calculated answers to problems. b. Consider possible effects of measurement errors on calculations. ...
Study Guide for Ch. 1
... Identify the benefits of the metric system versus classical measurement. Temperature scales and their details. Differentiate between solutions, colloids, and suspensions. Understand the physical properties involved in determining solids, liquids, & gases. Use significant figures in calculations and ...
... Identify the benefits of the metric system versus classical measurement. Temperature scales and their details. Differentiate between solutions, colloids, and suspensions. Understand the physical properties involved in determining solids, liquids, & gases. Use significant figures in calculations and ...
SOL Essential Knowledge
... A. Recognize the following relative to the Kinetic Molecular Theory: 1. Atoms and molecules are in constant motion. 2. The theory is a model for predicting and explaining gas behavior. 3. Forces of attraction between molecules determine the physical changes of state. 4. Pressure, temperature and vol ...
... A. Recognize the following relative to the Kinetic Molecular Theory: 1. Atoms and molecules are in constant motion. 2. The theory is a model for predicting and explaining gas behavior. 3. Forces of attraction between molecules determine the physical changes of state. 4. Pressure, temperature and vol ...
chemistry form iii - Covington Latin School
... to introduce the students to the principles of chemistry to increase the students' knowledge of the chemical world around them to ensure passage of all college-level chemistry courses to equip the student with hands-on laboratory skills to train the students to write scientific laboratory reports ...
... to introduce the students to the principles of chemistry to increase the students' knowledge of the chemical world around them to ensure passage of all college-level chemistry courses to equip the student with hands-on laboratory skills to train the students to write scientific laboratory reports ...
Chemistry Vocab for Quiz 12/21 or 12/22 Atom – The smallest
... Compound – A substance made of 2 or elements chemically combined in a specific ratio. Chemical bond – the force that holds 2 atoms together. Mixture – Two or more substances that are mixed together but not chemically bound, Physical change – A change that alters the form or appearance of a material ...
... Compound – A substance made of 2 or elements chemically combined in a specific ratio. Chemical bond – the force that holds 2 atoms together. Mixture – Two or more substances that are mixed together but not chemically bound, Physical change – A change that alters the form or appearance of a material ...
chapters 1-4
... Periodic table: symbol, atomic number; elements organized in groups (e.g. noble gases, noble metals) Diatomic molecules exist as natural state of seven elements (all nonmetals). ...
... Periodic table: symbol, atomic number; elements organized in groups (e.g. noble gases, noble metals) Diatomic molecules exist as natural state of seven elements (all nonmetals). ...
BASIC CHEMISTRY
... number of protons and neutrons in the nucleus and the electrons in the energy level. ...
... number of protons and neutrons in the nucleus and the electrons in the energy level. ...
Chemistry Final Study Guide
... Boiling can occur only when the liquid reaches a certain temperature, called the __________ __________. The process by which a gas changes its state to become a liquid is called __________. Physical properties can be used to identify a substance, such as __________, __________, and __________ proper ...
... Boiling can occur only when the liquid reaches a certain temperature, called the __________ __________. The process by which a gas changes its state to become a liquid is called __________. Physical properties can be used to identify a substance, such as __________, __________, and __________ proper ...
CHAPTER 1 Practice Exercises 1.1 12.3 g Cd 1.3 26.9814 u 1.5
... atom, which does not affect the chemistry of the elements as much as the number of protons. ...
... atom, which does not affect the chemistry of the elements as much as the number of protons. ...
Metric Unit – Chapter 1
... Distinguishing Elements and Compounds: Element = the ___________________________________________________ _______________________________________________________________ Oxygen and hydrogen are 2 of the more than 100 known elements. Compound = a substance that contains _____________________________ ...
... Distinguishing Elements and Compounds: Element = the ___________________________________________________ _______________________________________________________________ Oxygen and hydrogen are 2 of the more than 100 known elements. Compound = a substance that contains _____________________________ ...
Chemistry Content Standards
... evolution, water production, and changes in energy to the system. c. Apply concepts of the mole and Avogadro’s number to conceptualize and calculate • Empirical/molecular formulas, • Mass, moles and molecules relationships, • Molar volumes of gases. d. Identify and solve different types of stoichiom ...
... evolution, water production, and changes in energy to the system. c. Apply concepts of the mole and Avogadro’s number to conceptualize and calculate • Empirical/molecular formulas, • Mass, moles and molecules relationships, • Molar volumes of gases. d. Identify and solve different types of stoichiom ...
chapter 7 – cyu
... Geissler develop the gas discharge tube. This tube, when it has all the air pumped out of it, initially glowed blue then green at one end when under very low pressure. The green glow at the anode end was the result of the electrons being released from the cathode at the opposite end of the tube. The ...
... Geissler develop the gas discharge tube. This tube, when it has all the air pumped out of it, initially glowed blue then green at one end when under very low pressure. The green glow at the anode end was the result of the electrons being released from the cathode at the opposite end of the tube. The ...
PS7aChemistryReviewRevised
... Can you model a) the molecular changes, b) potential and kinetic changes for each indicated time interval? ...
... Can you model a) the molecular changes, b) potential and kinetic changes for each indicated time interval? ...
PowerPoint
... All one kind of atom. Compounds are substances that can be broken down by chemical methods • When they are broken down, the pieces have completely different properties than the compound. • Made of molecules- two or more atoms ...
... All one kind of atom. Compounds are substances that can be broken down by chemical methods • When they are broken down, the pieces have completely different properties than the compound. • Made of molecules- two or more atoms ...
Earth`s Chemistry
... Chemical bonds = forces that hold atoms together to make compounds Ionic bonds = electrons are transferred from one atom to another Ion = an atom or group of atoms that carry an electrical charge ( positive or negative) ...
... Chemical bonds = forces that hold atoms together to make compounds Ionic bonds = electrons are transferred from one atom to another Ion = an atom or group of atoms that carry an electrical charge ( positive or negative) ...
Deconstructed HS-PS1-2
... could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical reactions involving main group elements and combustion reactions.] ...
... could include the reaction of sodium and chlorine, of carbon and oxygen, or of carbon and hydrogen.] [Assessment Boundary: Assessment is limited to chemical reactions involving main group elements and combustion reactions.] ...
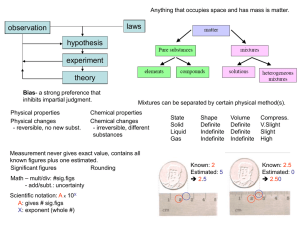
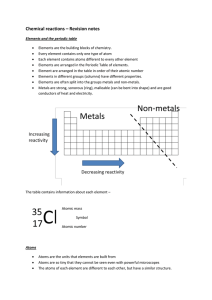
Chemical reactions revision
... Mixtures can be separated more easily than compounds The technique used depends on the properties of the substances Iron can be separated by sulphur by a magnet. Some liquids can be separated by differences in their boiling points This is distillation ...
... Mixtures can be separated more easily than compounds The technique used depends on the properties of the substances Iron can be separated by sulphur by a magnet. Some liquids can be separated by differences in their boiling points This is distillation ...
Inside the Atom connections to the lower secondary (KS3
... The periodic table Pupils are taught about: • the varying physical and chemical properties of different elements. During KS3 students will learn about the Periodic Table and the properties of the periods and groups. This is a good link to how the discovery of the structure of the atom, led to the e ...
... The periodic table Pupils are taught about: • the varying physical and chemical properties of different elements. During KS3 students will learn about the Periodic Table and the properties of the periods and groups. This is a good link to how the discovery of the structure of the atom, led to the e ...
Chem vocab quiz definitons
... Liquid is the state of matter that is described as having a definite volume but an indefinite shape. Gas is the state of matter that is described as having no definite shape, or volume. Solid is the state of matter that is described as having a definite shape and volume. Viscosity is a property of l ...
... Liquid is the state of matter that is described as having a definite volume but an indefinite shape. Gas is the state of matter that is described as having no definite shape, or volume. Solid is the state of matter that is described as having a definite shape and volume. Viscosity is a property of l ...
History of chemistry

The history of chemistry represents a time span from ancient history to the present. By 1000 BC, civilizations used technologies that would eventually form the basis to the various branches of chemistry. Examples include extracting metals from ores, making pottery and glazes, fermenting beer and wine, extracting chemicals from plants for medicine and perfume, rendering fat into soap, making glass, and making alloys like bronze.The protoscience of chemistry, alchemy, was unsuccessful in explaining the nature of matter and its transformations. However, by performing experiments and recording the results, alchemists set the stage for modern chemistry. The distinction began to emerge when a clear differentiation was made between chemistry and alchemy by Robert Boyle in his work The Sceptical Chymist (1661). While both alchemy and chemistry are concerned with matter and its transformations, chemists are seen as applying scientific method to their work.Chemistry is considered to have become an established science with the work of Antoine Lavoisier, who developed a law of conservation of mass that demanded careful measurement and quantitative observations of chemical phenomena. The history of chemistry is intertwined with the history of thermodynamics, especially through the work of Willard Gibbs.