1. Define each of the following terms: a.Alkaline earth metals
... 9. What is the difference between an ionic compound and a molecular compound in terms of: a. The bonds formed between them An ionic compound is formed because electrons are transferred from one element to another using ionic bonds. A molecular compound is formed when elements share electrons t ...
... 9. What is the difference between an ionic compound and a molecular compound in terms of: a. The bonds formed between them An ionic compound is formed because electrons are transferred from one element to another using ionic bonds. A molecular compound is formed when elements share electrons t ...
File - Science With BLT
... c. changes the formula so that it no longer represents that compound. d. has no effect on the formula. Which formula does NOT represent a molecule? a. H2O (water) c. CO2 (carbon dioxide) b. NH3 (ammonia) d. NaCl (table salt) What is the formula for zinc fluoride? a. ZnF c. Zn2F b. ZnF2 d. Zn2F3 What ...
... c. changes the formula so that it no longer represents that compound. d. has no effect on the formula. Which formula does NOT represent a molecule? a. H2O (water) c. CO2 (carbon dioxide) b. NH3 (ammonia) d. NaCl (table salt) What is the formula for zinc fluoride? a. ZnF c. Zn2F b. ZnF2 d. Zn2F3 What ...
Ch. 2 - Ltcconline.net
... 2. electrons vary in energy 3. electrons occur at certain energy levels F. Ionic bonds are attractions between ions of opposite charges 1. ion = loss or gain of electron 2. ions with opposite charges attract each other 3. resulting compound is electrically neutral 4. sodium and chloride ions are alw ...
... 2. electrons vary in energy 3. electrons occur at certain energy levels F. Ionic bonds are attractions between ions of opposite charges 1. ion = loss or gain of electron 2. ions with opposite charges attract each other 3. resulting compound is electrically neutral 4. sodium and chloride ions are alw ...
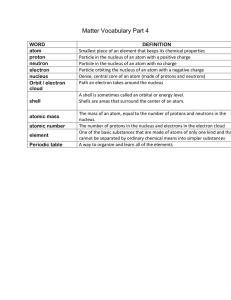
Matter Vocab Part 4
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
Properties of Metals vs. Nonmetals vs. Metalloids
... Quantum mechanical model (Werner, Heisenberg, Schrodinger): Energy levels (n=1, 2, 3, 4,…) – represented by periods on the periodic table Sublevels: (s, p, d, f) – represented by blocks on the periodic table Orbitals – region of space where up to 2 electrons may be found ...
... Quantum mechanical model (Werner, Heisenberg, Schrodinger): Energy levels (n=1, 2, 3, 4,…) – represented by periods on the periodic table Sublevels: (s, p, d, f) – represented by blocks on the periodic table Orbitals – region of space where up to 2 electrons may be found ...
Chemistry Vocab for Quiz 12/21 or 12/22 Atom – The smallest
... Atom – The smallest particle of an element. Atomic number - The number of protons in the nucleus of an element Atomic mass – The average mass of one atom of an element Proton – A small positively particle in the nucleus Neutron – a small particle in the nucleus with no charge Electron – A tiny negat ...
... Atom – The smallest particle of an element. Atomic number - The number of protons in the nucleus of an element Atomic mass – The average mass of one atom of an element Proton – A small positively particle in the nucleus Neutron – a small particle in the nucleus with no charge Electron – A tiny negat ...
MatterPP4
... The number of electrons in the outermost energy level determines the chemical behavior of the different elements. Valence electrons are the outermost electrons in an atom. Elements with the same number of valence electrons have similar chemical properties. Group 1 has 1 valence electron, group 2 has ...
... The number of electrons in the outermost energy level determines the chemical behavior of the different elements. Valence electrons are the outermost electrons in an atom. Elements with the same number of valence electrons have similar chemical properties. Group 1 has 1 valence electron, group 2 has ...
matter crct/final exam review
... 41. Why do atoms share valence electrons or transfer valence electrons? 42. What is the difference between a compound and an element? ...
... 41. Why do atoms share valence electrons or transfer valence electrons? 42. What is the difference between a compound and an element? ...
TEST REVIEW S Valence Electrons TEST REVIEW SHEET 2017
... ionic bond and become a positive ion. If >4, it will take them and become a negative ion For the most part…. metals will give away their valence electrons and nonmetals will take enough valence electrons to fill their outer shell. Determine the charge (ionization number) for the following elements w ...
... ionic bond and become a positive ion. If >4, it will take them and become a negative ion For the most part…. metals will give away their valence electrons and nonmetals will take enough valence electrons to fill their outer shell. Determine the charge (ionization number) for the following elements w ...
1 - shawnschmitt
... for comparison f. Hypothesis- a possible explaination for observations, a testable idea g. Mole- the amount of particles in 12g of Carbon-12, also, the amount of substance having 6.022x1023 of any kind of particle h. half-life- the amount of time required for ½ of the mass of an isotope to decay i. ...
... for comparison f. Hypothesis- a possible explaination for observations, a testable idea g. Mole- the amount of particles in 12g of Carbon-12, also, the amount of substance having 6.022x1023 of any kind of particle h. half-life- the amount of time required for ½ of the mass of an isotope to decay i. ...
Remember Question words
... choose, demonstrate, illustrate, interpret, operate, schedule, sketch, solve, use, write ...
... choose, demonstrate, illustrate, interpret, operate, schedule, sketch, solve, use, write ...
Unit Five: Periodic Table Families
... Every element in a family has the same number of valence electrons All elements in a period have the same quantum number (same level of shells) s-Block elements Groups 1 and 2 + Helium p-Block Groups on right of the table d-Block Transition metals f-Block Inner transition metals ...
... Every element in a family has the same number of valence electrons All elements in a period have the same quantum number (same level of shells) s-Block elements Groups 1 and 2 + Helium p-Block Groups on right of the table d-Block Transition metals f-Block Inner transition metals ...
sch3u unit 1 test: matter
... 9. When fluorine forms an ionic bond it tends to a. lose electrons b. gain electrons c. share electrons d. lose protons 10. Copper (II) hydroxide is composed of a. 2 elements, 2 atoms b. 2 elements, 3 atoms c. 3 elements, 4 atoms d. 3 elements, 5 atoms ...
... 9. When fluorine forms an ionic bond it tends to a. lose electrons b. gain electrons c. share electrons d. lose protons 10. Copper (II) hydroxide is composed of a. 2 elements, 2 atoms b. 2 elements, 3 atoms c. 3 elements, 4 atoms d. 3 elements, 5 atoms ...
Chemistry for Bio 11
... • 6CO2(g)+ 6H2O(l) + hν C6H12O6(s) + 6O2(g) • Macromolecules as reactants are broken down for energy: C6H12O6(s) + 6O2(g) 6CO2(g)+ 6H2O(l) All the reactions of a living thing are called its metabolism ...
... • 6CO2(g)+ 6H2O(l) + hν C6H12O6(s) + 6O2(g) • Macromolecules as reactants are broken down for energy: C6H12O6(s) + 6O2(g) 6CO2(g)+ 6H2O(l) All the reactions of a living thing are called its metabolism ...
Pretest 4.3 2008
... evenly distributed throughout. d. According to Bohr, the atom is a sphere in which the positive charges are concentrated in a nucleus and the negative charges travel around the nucleus in orbits. ...
... evenly distributed throughout. d. According to Bohr, the atom is a sphere in which the positive charges are concentrated in a nucleus and the negative charges travel around the nucleus in orbits. ...
Define:
... 40. Write the following numbers in scientific notation. a. 57000 b. 0.000489 41. Determine the number of significant figures in the following numbers. a. 600 b. 600. c. 0.001200 d. 507 42. In the number 0.305 L, which digit is estimated? 43. Express the sum of 8.67 m and 5.2 m to the correct number ...
... 40. Write the following numbers in scientific notation. a. 57000 b. 0.000489 41. Determine the number of significant figures in the following numbers. a. 600 b. 600. c. 0.001200 d. 507 42. In the number 0.305 L, which digit is estimated? 43. Express the sum of 8.67 m and 5.2 m to the correct number ...
Chemical Change
... The chemical properties of elements are related to the energy changes that take place when atoms lose, gain or share electrons to obtain a filled valence shell. ...
... The chemical properties of elements are related to the energy changes that take place when atoms lose, gain or share electrons to obtain a filled valence shell. ...
Note 1.1 Chemistry of Life
... Polar covalent bond - is a bond between two atoms, make up of unequally shared electrons. Polarity is partial positive or negative charge at the ends of a molecule. Covalent bond deals with the sharing of electrons between atoms, there can be an unequal sharing between these atoms. The greater the e ...
... Polar covalent bond - is a bond between two atoms, make up of unequally shared electrons. Polarity is partial positive or negative charge at the ends of a molecule. Covalent bond deals with the sharing of electrons between atoms, there can be an unequal sharing between these atoms. The greater the e ...
Chemistry Midterm Review Study Guide 2012
... b. The electrons available to be gained, lost, or shared in the formation of chemical compounds are called Valence electrons. c. The measure of the ability of an atom in a chemical compound to attract electrons is called electronegativity d. The energy required to remove an electron from an atom is ...
... b. The electrons available to be gained, lost, or shared in the formation of chemical compounds are called Valence electrons. c. The measure of the ability of an atom in a chemical compound to attract electrons is called electronegativity d. The energy required to remove an electron from an atom is ...
Bonding practice lessons 1-3
... 6. Based on electronegativity values, which type of elements tends to have the greatest attraction for electrons in a bond? A) metals C) nonmetals ...
... 6. Based on electronegativity values, which type of elements tends to have the greatest attraction for electrons in a bond? A) metals C) nonmetals ...
atoms-chemical
... • Atoms with incomplete valence shells interact by either sharing or transferring valence electrons. • These interactions typically result in the atoms remaining close together, held by an attractions called chemical bonds. ...
... • Atoms with incomplete valence shells interact by either sharing or transferring valence electrons. • These interactions typically result in the atoms remaining close together, held by an attractions called chemical bonds. ...
Chemical Bonding
... of only one type of atom. • Isotopes are elements with different numbers of neutrons. • Because isotopes have the same number electrons, all isotopes of an element have the same chemical properties. ...
... of only one type of atom. • Isotopes are elements with different numbers of neutrons. • Because isotopes have the same number electrons, all isotopes of an element have the same chemical properties. ...
Grade 11 Chemistry E.. - hrsbstaff.ednet.ns.ca
... tell what type of reaction it is. a) lead (II) nitrate and sodium iodide react to make lead iodide and sodium nitrate. b) calcium carbonate when you heat it produces calcium oxide and carbon dioxide. c) propane reacts with oxygen to produce carbon dioxide and water d) copper metal and silver nitrate ...
... tell what type of reaction it is. a) lead (II) nitrate and sodium iodide react to make lead iodide and sodium nitrate. b) calcium carbonate when you heat it produces calcium oxide and carbon dioxide. c) propane reacts with oxygen to produce carbon dioxide and water d) copper metal and silver nitrate ...
Electronegativity

Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom or a functional group to attract electrons (or electron density) towards itself. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it. The term ""electronegativity"" was introduced by Jöns Jacob Berzelius in 1811,though the concept was known even before that and was studied by many chemists including Avogadro.In spite of its long history, an accurate scale of electronegativity had to wait till 1932, when Linus Pauling proposed an electronegativity scale, which depends on bond energies, as a development of valence bond theory. It has been shown to correlate with a number of other chemical properties. Electronegativity cannot be directly measured and must be calculated from other atomic or molecular properties. Several methods of calculation have been proposed, and although there may be small differences in the numerical values of the electronegativity, all methods show the same periodic trends between elements. The most commonly used method of calculation is that originally proposed by Linus Pauling. This gives a dimensionless quantity, commonly referred to as the Pauling scale, on a relative scale running from around 0.7 to 3.98 (hydrogen = 2.20). When other methods of calculation are used, it is conventional (although not obligatory) to quote the results on a scale that covers the same range of numerical values: this is known as an electronegativity in Pauling units. As it is usually calculated, electronegativity is not a property of an atom alone, but rather a property of an atom in a molecule. Properties of a free atom include ionization energy and electron affinity. It is to be expected that the electronegativity of an element will vary with its chemical environment, but it is usually considered to be a transferable property, that is to say that similar values will be valid in a variety of situations.On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more ""pull"" it will have on electrons) and the number/location of other electrons present in the atomic shells (the more electrons an atom has, the farther from the nucleus the valence electrons will be, and as a result the less positive charge they will experience—both because of their increased distance from the nucleus, and because the other electrons in the lower energy core orbitals will act to shield the valence electrons from the positively charged nucleus).The opposite of electronegativity is electropositivity: a measure of an element's ability to donate electrons.Caesium is the least electronegative element in the periodic table (=0.79), while fluorine is most electronegative (=3.98). (Francium and caesium were originally assigned both assigned 0.7; caesium's value was later refined to 0.79, but no experimental data allows a similar refinement for francium. However, francium's ionization energy is known to be slightly higher than caesium's, in accordance with the relativistic stabilization of the 7s orbital, and this in turn implies that caesium is in fact more electronegative than francium.)