1 Structure of Atom
... upper plate. The air between the plates is then exposed to X-rays which eject electrons from air molecules. Some of these electrons are captured by the oil droplet and it acquires a negative charge. When the plates are earthed, the droplet falls under the influence of gravity. He adjusted the streng ...
... upper plate. The air between the plates is then exposed to X-rays which eject electrons from air molecules. Some of these electrons are captured by the oil droplet and it acquires a negative charge. When the plates are earthed, the droplet falls under the influence of gravity. He adjusted the streng ...
Chapter 4
... Substances that cannot be broken down by simple chemical means 115 known: 88 found in nature, others are man ...
... Substances that cannot be broken down by simple chemical means 115 known: 88 found in nature, others are man ...
Chapter 2 ATOMS AND ELEMENTS
... • Stretching between Groups 2A and 3A is a series of elements called the transition elements. • These fill the B-groups (1B through 8B) in the fourth through the seventh periods in the center of the periodic table. • All are metals and 13 of them are in the top 30 elements in terms of abundance in t ...
... • Stretching between Groups 2A and 3A is a series of elements called the transition elements. • These fill the B-groups (1B through 8B) in the fourth through the seventh periods in the center of the periodic table. • All are metals and 13 of them are in the top 30 elements in terms of abundance in t ...
Chapter 2 - San Joaquin Memorial High School
... applications of chemical phenomena that were utilized prior to 1000 B.C. The Greeks were the first to try to explain why chemical changes occur. By about 400 B.C. they had proposed that all matter was composed of four fundamental substances: fire, earth, water, and air. The Greeks also considered th ...
... applications of chemical phenomena that were utilized prior to 1000 B.C. The Greeks were the first to try to explain why chemical changes occur. By about 400 B.C. they had proposed that all matter was composed of four fundamental substances: fire, earth, water, and air. The Greeks also considered th ...
Atom Building - Campbell County Schools
... • Neils Bohr hypothesized that the atom was like a tiny solar system, with electrons circling the nucleus in well defined orbits like planets • The speculated paths of the electrons were called orbitals, shells, or energy ...
... • Neils Bohr hypothesized that the atom was like a tiny solar system, with electrons circling the nucleus in well defined orbits like planets • The speculated paths of the electrons were called orbitals, shells, or energy ...
TOPIC 12. THE ELEMENTS
... An irreplaceable gas with vital applications in magnetic resonance imaging machines is used and lost - filling party balloons! An object made from some metals retains a memory of its initial shape and if distorted, it will return to the original shape when heated. Origin of the elements. As discusse ...
... An irreplaceable gas with vital applications in magnetic resonance imaging machines is used and lost - filling party balloons! An object made from some metals retains a memory of its initial shape and if distorted, it will return to the original shape when heated. Origin of the elements. As discusse ...
Has the Periodic Table Been Successfully Axiomatized?
... established that the correct ordering principle for the elements consists in atomic number instead of a atomic weight. This development is crucial for the arguments contained in Hettema and Kuipers’ article, since they go on to discuss whether the ordering number z (implicit in the work of Mendeleev ...
... established that the correct ordering principle for the elements consists in atomic number instead of a atomic weight. This development is crucial for the arguments contained in Hettema and Kuipers’ article, since they go on to discuss whether the ordering number z (implicit in the work of Mendeleev ...
s_block - ilc.edu.hk
... Reasons : 1. Absence of low-lying vacant d-orbtals to accept lone pairs from ligands. For Na+, 1s2, 2s2, 2p6, 3s, 3p, 3d High-lying relative to 2p ...
... Reasons : 1. Absence of low-lying vacant d-orbtals to accept lone pairs from ligands. For Na+, 1s2, 2s2, 2p6, 3s, 3p, 3d High-lying relative to 2p ...
Follow this presentation to draw atoms 1-13
... There are two apparent flaws with such a model. Firstly, there is no obvious quantization. That is, the final equation does not quantize r, since we can find the electromagnetic analog a Kepplerian orbits for any r, given the appropriate value of v (and thus T). A related problem that does not appea ...
... There are two apparent flaws with such a model. Firstly, there is no obvious quantization. That is, the final equation does not quantize r, since we can find the electromagnetic analog a Kepplerian orbits for any r, given the appropriate value of v (and thus T). A related problem that does not appea ...
FREE Sample Here
... The atomic mass of naturally occurring cobalt, which exists in nature as a single isotope, is listed as 58.9332 u. This means that a. all cobalt atoms found in nature have a mass which is 58.9332/12.000 times as great as that of a 12C atom. b. all cobalt atoms found in nature have a mass which is 58 ...
... The atomic mass of naturally occurring cobalt, which exists in nature as a single isotope, is listed as 58.9332 u. This means that a. all cobalt atoms found in nature have a mass which is 58.9332/12.000 times as great as that of a 12C atom. b. all cobalt atoms found in nature have a mass which is 58 ...
FREE Sample Here
... The atomic mass of naturally occurring cobalt, which exists in nature as a single isotope, is listed as 58.9332 u. This means that a. all cobalt atoms found in nature have a mass which is 58.9332/12.000 times as great as that of a 12C atom. b. all cobalt atoms found in nature have a mass which is 58 ...
... The atomic mass of naturally occurring cobalt, which exists in nature as a single isotope, is listed as 58.9332 u. This means that a. all cobalt atoms found in nature have a mass which is 58.9332/12.000 times as great as that of a 12C atom. b. all cobalt atoms found in nature have a mass which is 58 ...
Document
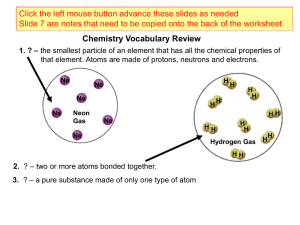
... an atom that have a charge. • Protons and neutrons have essentially the same mass (1 unit each). • The mass of an electron is so small we ignore it. ...
... an atom that have a charge. • Protons and neutrons have essentially the same mass (1 unit each). • The mass of an electron is so small we ignore it. ...
Exam View Benchmark Review sheet for 1st nine weeks
... 74. Calculate the number of protons, electrons, and neutrons in an atom of the element Pb with mass number 207 and atomic number 82. 75. Fluorine (F) contains 9 proton and 10 neutrons. Calculate its mass in amu for 81 atoms. (Given: mass of a proton = 1.007276 amu, mass of a neutron = 1.008665 amu) ...
... 74. Calculate the number of protons, electrons, and neutrons in an atom of the element Pb with mass number 207 and atomic number 82. 75. Fluorine (F) contains 9 proton and 10 neutrons. Calculate its mass in amu for 81 atoms. (Given: mass of a proton = 1.007276 amu, mass of a neutron = 1.008665 amu) ...
Chemistry Benchmark 1 Review
... 74. Calculate the number of protons, electrons, and neutrons in an atom of the element Pb with mass number 207 and atomic number 82. 75. Fluorine (F) contains 9 proton and 10 neutrons. Calculate its mass in amu for 81 atoms. (Given: mass of a proton = 1.007276 amu, mass of a neutron = 1.008665 amu) ...
... 74. Calculate the number of protons, electrons, and neutrons in an atom of the element Pb with mass number 207 and atomic number 82. 75. Fluorine (F) contains 9 proton and 10 neutrons. Calculate its mass in amu for 81 atoms. (Given: mass of a proton = 1.007276 amu, mass of a neutron = 1.008665 amu) ...
Periodic Table
... table is useful as it is possible to predict the properties of an individual element from its position. -l'he Modern Periodic table is a table of all the chemical elements, in order of their atomic numbers so that elements with similar properties arc close to each other. The Modern Periodic ...
... table is useful as it is possible to predict the properties of an individual element from its position. -l'he Modern Periodic table is a table of all the chemical elements, in order of their atomic numbers so that elements with similar properties arc close to each other. The Modern Periodic ...
The Atomic Theory - Academic Magnet High School
... twice as much oxygen as forming A. In other words, if you could make A with 3 grams of carbon and 4 grams of oxygen, B could be made with the same 3 grams of carbon but with 8 grams of oxygen instead. Dalton asked himself – why does B require twice as much oxygen as A does? Why not 1.21 times as muc ...
... twice as much oxygen as forming A. In other words, if you could make A with 3 grams of carbon and 4 grams of oxygen, B could be made with the same 3 grams of carbon but with 8 grams of oxygen instead. Dalton asked himself – why does B require twice as much oxygen as A does? Why not 1.21 times as muc ...
Final Exam - KFUPM Faculty List
... into the central triangle: T-shape, polar 5 electron pairs: Cl needs 5 hybrid orbitals to put them in: 1 (s) + 3 (p3) + 1 (d) = 5 hybrid orbitals (dsp3) ...
... into the central triangle: T-shape, polar 5 electron pairs: Cl needs 5 hybrid orbitals to put them in: 1 (s) + 3 (p3) + 1 (d) = 5 hybrid orbitals (dsp3) ...
Chapter 0 A Very Brief History of Chemistry Multiple Choice Questions
... 38. The atomic mass of naturally occurring silver, which is a mixture of two isotopes, is listed as 107.868 u. This means that a. all silver atoms found in nature have a mass which is 107.868/12.000 times as great as that of a 12C atom. b. all silver atoms found in nature have a mass which is 107.86 ...
... 38. The atomic mass of naturally occurring silver, which is a mixture of two isotopes, is listed as 107.868 u. This means that a. all silver atoms found in nature have a mass which is 107.868/12.000 times as great as that of a 12C atom. b. all silver atoms found in nature have a mass which is 107.86 ...
Chapter 4: The Structure of the Atom
... Conservation of mass Recall from Chapter 3 that the law of conservation of mass states that mass is conserved in any process, such as a chemical reaction. Dalton’s atomic theory easily explains that the conservation of mass in chemical reactions is the result of the separation, combination, or rearr ...
... Conservation of mass Recall from Chapter 3 that the law of conservation of mass states that mass is conserved in any process, such as a chemical reaction. Dalton’s atomic theory easily explains that the conservation of mass in chemical reactions is the result of the separation, combination, or rearr ...
Chapter 2 ATOMS AND ELEMENTS
... • Stretching between Groups 2A and 3A is a series of elements called the transition elements. • These fill the B-groups (1B through 8B) in the fourth through the seventh periods in the center of the periodic table. • All are metals and 13 of them are in the top 30 elements in terms of abundance in t ...
... • Stretching between Groups 2A and 3A is a series of elements called the transition elements. • These fill the B-groups (1B through 8B) in the fourth through the seventh periods in the center of the periodic table. • All are metals and 13 of them are in the top 30 elements in terms of abundance in t ...