THEORY AND IMPORTANCE OF OXYGEN
... 24.58 eV respectively. This, and the fact that the electronegativity of nitrogen with respect to two electrons is negative, results in an excessive ability of the nitrogen to delocalize electrons. Nitrogen as a bridge should be therefore stabilized by means of a third acceptor, for instance by H whi ...
... 24.58 eV respectively. This, and the fact that the electronegativity of nitrogen with respect to two electrons is negative, results in an excessive ability of the nitrogen to delocalize electrons. Nitrogen as a bridge should be therefore stabilized by means of a third acceptor, for instance by H whi ...
Transition Metal Series
... Isomerism in Coordination Compounds 1. Identify each of the following pairs of coordination compounds or complex ions as coordination isomers, linkage isomers, or geometric isomers. (a) [Cr(H2O)5(SCN)]Cl2 and [Cr(H2O)5(NCS)]Cl2 (b) [Cr(NH3)6][Co(CN)6] and [Co(NH3)6][Cr(CN)6] (c) [Cu(NH3)4][PtCl4] an ...
... Isomerism in Coordination Compounds 1. Identify each of the following pairs of coordination compounds or complex ions as coordination isomers, linkage isomers, or geometric isomers. (a) [Cr(H2O)5(SCN)]Cl2 and [Cr(H2O)5(NCS)]Cl2 (b) [Cr(NH3)6][Co(CN)6] and [Co(NH3)6][Cr(CN)6] (c) [Cu(NH3)4][PtCl4] an ...
Syracuse Syllabus
... 2. No student will be refused admission because he or she is unable to participate in a course requirement because of his or her religious holy day requirements. Again, you must make provisions before such absences. According to University policy, “an opportunity to make up examinations and other cl ...
... 2. No student will be refused admission because he or she is unable to participate in a course requirement because of his or her religious holy day requirements. Again, you must make provisions before such absences. According to University policy, “an opportunity to make up examinations and other cl ...
CHAPTER 22 TRANSITION METAL CHEMISTRY AND
... that appear green or blue to the eye are absorbing light from the lower energy red or orange part of the spectrum. Cyanide ion is a very strong field ligand. It causes a larger crystal field splitting than water, resulting in the absorption of higher energy (shorter wavelength) radiation when a d el ...
... that appear green or blue to the eye are absorbing light from the lower energy red or orange part of the spectrum. Cyanide ion is a very strong field ligand. It causes a larger crystal field splitting than water, resulting in the absorption of higher energy (shorter wavelength) radiation when a d el ...
Ionic Bonding
... eight electrons in their outer energy levels (or two in the case of helium). These noble gas structures are thought of as being in some way a "desirable" thing for an atom to have. You may well have been left with the strong impression that when other atoms react, they try to organize things such th ...
... eight electrons in their outer energy levels (or two in the case of helium). These noble gas structures are thought of as being in some way a "desirable" thing for an atom to have. You may well have been left with the strong impression that when other atoms react, they try to organize things such th ...
JCE0198 p0098 The Ligand Field Spectra of Copper(II) Complexes
... for the third solution (containing [Cu(en)2 (H2O)2 ]2+ ) is approximately 7000 cm{1 wide at half-height. Note that spectra which arise from d–d electronic transitions are very weak (low molar absorptivities) because the transitions are quantum-mechanically forbidden (Laporte selection rule). For a p ...
... for the third solution (containing [Cu(en)2 (H2O)2 ]2+ ) is approximately 7000 cm{1 wide at half-height. Note that spectra which arise from d–d electronic transitions are very weak (low molar absorptivities) because the transitions are quantum-mechanically forbidden (Laporte selection rule). For a p ...
3UE-Exam Review-June2010 - Savita Pall and Chemistry
... 37. Which statement is true about ionization energies? a) The first ionization energies in a period increase smoothly from left to right across the periodic table. b) The first ionization energies of the elements in a family increase smoothly down the family. c) Successive ionization energies increa ...
... 37. Which statement is true about ionization energies? a) The first ionization energies in a period increase smoothly from left to right across the periodic table. b) The first ionization energies of the elements in a family increase smoothly down the family. c) Successive ionization energies increa ...
Synthesis and magnetic characterization of Ln(III) complexes with 4
... (crot, crotonate (C4H5O2); bpy, 4,4 0 -bipyridine (C10H8N2); Ln, Nd, Gd, Ho, Er, Y) is presented. The ninefold Ln coordination polyhedra form dimeric entities that are connected throughout the bpy units into infinite polymeric chains. All (but the yttrium) reported compounds present a weak antiferrom ...
... (crot, crotonate (C4H5O2); bpy, 4,4 0 -bipyridine (C10H8N2); Ln, Nd, Gd, Ho, Er, Y) is presented. The ninefold Ln coordination polyhedra form dimeric entities that are connected throughout the bpy units into infinite polymeric chains. All (but the yttrium) reported compounds present a weak antiferrom ...
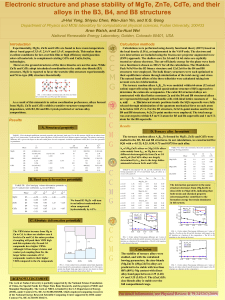
Electronic structure and phase stability of MgTe, ZnTe, CdTe, and
... core interactions are included using the frozen-core projector augmented wave (PAW) approach. The shallow-core Zn 3d and Cd 4d states are explicitly treated as valence electrons. The cut-off kinetic energy for the plane wave basis wave functions is chosen as 300 eV for all the calculations. The Monk ...
... core interactions are included using the frozen-core projector augmented wave (PAW) approach. The shallow-core Zn 3d and Cd 4d states are explicitly treated as valence electrons. The cut-off kinetic energy for the plane wave basis wave functions is chosen as 300 eV for all the calculations. The Monk ...
Bonding in minerals - The Mineralogical Society of Great Britain and
... elements being relatively more intense than those derived from 2p orbitals (Scofield, 1976). It can, therefore, be seen that XP spectroscopy provides basic information about the ionization energies of the molecular orbitals present in a mineral and some limited information about their structure in t ...
... elements being relatively more intense than those derived from 2p orbitals (Scofield, 1976). It can, therefore, be seen that XP spectroscopy provides basic information about the ionization energies of the molecular orbitals present in a mineral and some limited information about their structure in t ...
CHAPTER 5 Electronic Properties of −Dawson Anions Wells
... MOs is the same, which suggests that the energy difference between both site reductions should be very close to the value obtained for the D3h form. All these results fully agree with the experimental data of Pope6b and Contant22 who demonstrated that equatorial sites are more easily reduced than po ...
... MOs is the same, which suggests that the energy difference between both site reductions should be very close to the value obtained for the D3h form. All these results fully agree with the experimental data of Pope6b and Contant22 who demonstrated that equatorial sites are more easily reduced than po ...
Chapter 4 The Structure of Matter
... atom to another atom − (2) be more flexible to bend and stretch without breaking. – (a) atoms can slide past each other without breaking their bonds ...
... atom to another atom − (2) be more flexible to bend and stretch without breaking. – (a) atoms can slide past each other without breaking their bonds ...
5000176793-5000311155-1-SP - Gazi University Journal of
... al. used ENDOR to measure Gd3+ center in CsCaF3 [10]. Brik and Avram investigated fine structure of lasing 4T2g level in a CsCaF3: V2+ compounds [11]. Falin et al. used magnetic resonance and optical spectroscopy to study a trigonal Yb3+ paramagnetic center in CsCaF3 single crystal [12]. They establ ...
... al. used ENDOR to measure Gd3+ center in CsCaF3 [10]. Brik and Avram investigated fine structure of lasing 4T2g level in a CsCaF3: V2+ compounds [11]. Falin et al. used magnetic resonance and optical spectroscopy to study a trigonal Yb3+ paramagnetic center in CsCaF3 single crystal [12]. They establ ...
Head-Gordon`s
... However, it has been known for three decades that the exact energy is in fact a functional of only the electron density (a function of only 3, rather than 3n, variables). The only catch is that the functional is not known. In section 4, I discuss density functional theory (DFT) which focuses on the ...
... However, it has been known for three decades that the exact energy is in fact a functional of only the electron density (a function of only 3, rather than 3n, variables). The only catch is that the functional is not known. In section 4, I discuss density functional theory (DFT) which focuses on the ...
Original
... Electrostatic attraction between closely packed, oppositely charged metal and nonmetal ions, form ionic bonds. The energy of ionic bonds can be calculated using Coulomb’s Law, where Q = the charge of each ion, and r = distance between ions (nm): (Negative answer = attraction, positive answer = repul ...
... Electrostatic attraction between closely packed, oppositely charged metal and nonmetal ions, form ionic bonds. The energy of ionic bonds can be calculated using Coulomb’s Law, where Q = the charge of each ion, and r = distance between ions (nm): (Negative answer = attraction, positive answer = repul ...
- DigitalCommons@USU
... The ground state corresponds to the electronic configuration 关 ¯ 兴 (b 2g ) 2 (a 1u ) 2 (a 1g ) 2 (1e g ) 2 , which yields a 3 A 2g state. The 1e g and a 1g orbitals are located predominantly on the metal, with a high degree of Fe– 3d character; a 1u ( ) and 2e g ( * ) are the HOMO and LUMO, resp ...
... The ground state corresponds to the electronic configuration 关 ¯ 兴 (b 2g ) 2 (a 1u ) 2 (a 1g ) 2 (1e g ) 2 , which yields a 3 A 2g state. The 1e g and a 1g orbitals are located predominantly on the metal, with a high degree of Fe– 3d character; a 1u ( ) and 2e g ( * ) are the HOMO and LUMO, resp ...
lecture 6
... Qualitative picture of two contributions to a transition metal (TM) density of states (DOS) – free electron-like sp-band, and narrow, structured d-band. Due to the large number of d-states, the d-band dominates the DOS and the varying properties over a TM series can be understood as arising from a d ...
... Qualitative picture of two contributions to a transition metal (TM) density of states (DOS) – free electron-like sp-band, and narrow, structured d-band. Due to the large number of d-states, the d-band dominates the DOS and the varying properties over a TM series can be understood as arising from a d ...
Document
... colour in solution is blue. Although we know that reactants lose their properties when forming a product, I wondered why it is blue or not another colour. Interested, I began researching this issue and found that the solutions of not only copper, but whole d-block(transition) elements are coloured. ...
... colour in solution is blue. Although we know that reactants lose their properties when forming a product, I wondered why it is blue or not another colour. Interested, I began researching this issue and found that the solutions of not only copper, but whole d-block(transition) elements are coloured. ...
Chapter 24 Organometallic d
... Organometallic compounds of the d-block Compounds with element-carbon bonds involving metals from the d-block ...
... Organometallic compounds of the d-block Compounds with element-carbon bonds involving metals from the d-block ...
Isomerism of coordination compounds
... Isomerism of coordination compounds Structural isomerism Ionization isomerism When ligands are exchanged in a complex by the counter-ion, it is called ionization isomerism. e.g. [Pt(NH3)4Cl2]Br2 and [Pt(NH3)4Br2]Cl Linkage isomerism A ligand connects with the central metal atom through different ato ...
... Isomerism of coordination compounds Structural isomerism Ionization isomerism When ligands are exchanged in a complex by the counter-ion, it is called ionization isomerism. e.g. [Pt(NH3)4Cl2]Br2 and [Pt(NH3)4Br2]Cl Linkage isomerism A ligand connects with the central metal atom through different ato ...
Unit 5 - INTEC Chemistry Blog
... Tetraamminecopper(II) Cu(NH3)42+(aq) deep blue/purple Tetrachlorocuprate(II) CuCl42-(aq) yellow hydrated thiocyanate complex of iron (III) [Fe(SCN)(H2O)5]2+ blood red [Co(H2O)6]2+(aq) pink hexaaquacobalt(II) [CoCl4]2-(aq) blue tetrachlorocobalte(II) (ii) the formation of a range of compounds in whic ...
... Tetraamminecopper(II) Cu(NH3)42+(aq) deep blue/purple Tetrachlorocuprate(II) CuCl42-(aq) yellow hydrated thiocyanate complex of iron (III) [Fe(SCN)(H2O)5]2+ blood red [Co(H2O)6]2+(aq) pink hexaaquacobalt(II) [CoCl4]2-(aq) blue tetrachlorocobalte(II) (ii) the formation of a range of compounds in whic ...
Jahn–Teller effect
-3D-balls.png?width=300)
The Jahn–Teller effect, sometimes also known as Jahn–Teller distortion, describes the geometrical distortion of molecules and ions that is associated with certain electron configurations. This electronic effect is named after Hermann Arthur Jahn and Edward Teller, who proved, using group theory, that orbital nonlinear spatially degenerate molecules cannot be stable. The Jahn–Teller theorem essentially states that any nonlinear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species. For a description of another type of geometrical distortion that occurs in crystals with substitutional impurities see article off-center ions.