Atomic Radius and Ionization Energy
... • The pattern of properties within a period repeats as you move across a period from left to right… When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties ...
... • The pattern of properties within a period repeats as you move across a period from left to right… When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties ...
r - Department Chemie und Biologie
... ionic bonds are formed by strong Coulomb interactions between two oppositely charged species (e.g. Na+Cl-) the potential energy varies with the distance r between the two ions by 1/r (the force varies with 1/r2) description by formal electron transfer from newly formed cation (usually metals) to new ...
... ionic bonds are formed by strong Coulomb interactions between two oppositely charged species (e.g. Na+Cl-) the potential energy varies with the distance r between the two ions by 1/r (the force varies with 1/r2) description by formal electron transfer from newly formed cation (usually metals) to new ...
Campbell Biology, 10e (Reece) Chapter 2 The Chemical Context of
... B) the number of protons in the element C) the number of protons plus neutrons in the element D) the number of protons plus electrons in the element 6) In what way are elements in the same column of the periodic table the same? They have the same number of _____. A) protons B) electrons when neutral ...
... B) the number of protons in the element C) the number of protons plus neutrons in the element D) the number of protons plus electrons in the element 6) In what way are elements in the same column of the periodic table the same? They have the same number of _____. A) protons B) electrons when neutral ...
Chapter 9 - Fayetteville State University
... 7) Periodic Law: States that elements arranged in order of the atomic number share similar chemical and physical properties. These arrangement are called groups, examples are the alkali metals (Li, Na, K, Rb, Cs), the halogens (F, Cl, Br, I). 8) Groups: A sequence of elements of increasing atomic nu ...
... 7) Periodic Law: States that elements arranged in order of the atomic number share similar chemical and physical properties. These arrangement are called groups, examples are the alkali metals (Li, Na, K, Rb, Cs), the halogens (F, Cl, Br, I). 8) Groups: A sequence of elements of increasing atomic nu ...
chemistry i
... Note: This set of problems does NOT cover all of the concepts that will be on the midterm. Remember to study your class notes and handouts for a full review! 1. Which sequence represents a correct order of historical developments leading to the modern model of the atom? A. The atom is a hard sphere ...
... Note: This set of problems does NOT cover all of the concepts that will be on the midterm. Remember to study your class notes and handouts for a full review! 1. Which sequence represents a correct order of historical developments leading to the modern model of the atom? A. The atom is a hard sphere ...
Intro to Atoms - Freehold Borough Schools
... Matter: anything that has mass and takes up space. Mass: measurement of how much matter is in an object Element: a substance that cannot be broken down into any other substances by chemical or physical means Compound: a substance of 2 or more elements Mixture: 2 or more substances that are mixed tog ...
... Matter: anything that has mass and takes up space. Mass: measurement of how much matter is in an object Element: a substance that cannot be broken down into any other substances by chemical or physical means Compound: a substance of 2 or more elements Mixture: 2 or more substances that are mixed tog ...
Atomic Theory - WaylandHighSchoolChemistry
... • Atom is mostly empty space – first nuclear model ...
... • Atom is mostly empty space – first nuclear model ...
ap chemistry chapter 8 bonding
... • Ionic bonds form when an atom that loses electrons easily reacts with an atom that has a high affinity for electrons. The charged ions are held together by their mutual attraction. • Ionic bonds form because the ion pair has lower energy than the separated ions. All bonds form in order to reach a ...
... • Ionic bonds form when an atom that loses electrons easily reacts with an atom that has a high affinity for electrons. The charged ions are held together by their mutual attraction. • Ionic bonds form because the ion pair has lower energy than the separated ions. All bonds form in order to reach a ...
Study Guide 1st Semester
... 31. What is a valence electron? 32. Where are the alkali metal elements found? How do their electron configurations end? What are some typical behaviors of alkali metals? 33. Where are the alkaline earth metals found? How do their electron configurations end? What are some typical behaviors of alkal ...
... 31. What is a valence electron? 32. Where are the alkali metal elements found? How do their electron configurations end? What are some typical behaviors of alkali metals? 33. Where are the alkaline earth metals found? How do their electron configurations end? What are some typical behaviors of alkal ...
FXM Rev 1 Key - Grande Cache Community High School
... anion This is a negative ion that has form after it gained one or more electrons. S2- is an example. molecule This is the smallest structural unit of a covalent compound. One H2O particle is an example. covalent bond This is the type of bond that forms between two or more atoms that share electrons. ...
... anion This is a negative ion that has form after it gained one or more electrons. S2- is an example. molecule This is the smallest structural unit of a covalent compound. One H2O particle is an example. covalent bond This is the type of bond that forms between two or more atoms that share electrons. ...
Exam 1 Topics to Review (McMurry Chpts 1
... Which of the above subatomic particles determines the size of the atom? c) Which of the above subatomic particles determine the mass of the atom? ...
... Which of the above subatomic particles determines the size of the atom? c) Which of the above subatomic particles determine the mass of the atom? ...
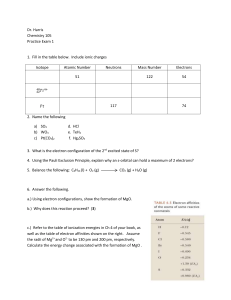
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... c.) Refer to the table of ionization energies in Ch 4 of your book, as well as the table of electron affinities shown on the right. Assume the radii of Mg2+ and O2- to be 130 pm and 200 pm, respectively. Calculate the energy change associated with the formation of MgO . ...
... c.) Refer to the table of ionization energies in Ch 4 of your book, as well as the table of electron affinities shown on the right. Assume the radii of Mg2+ and O2- to be 130 pm and 200 pm, respectively. Calculate the energy change associated with the formation of MgO . ...
Name: Period
... a. Ionic Solids b. Metallic Solids c. Network Solids 7. How are ionic compounds and molecular compounds different? Ionic Compounds ...
... a. Ionic Solids b. Metallic Solids c. Network Solids 7. How are ionic compounds and molecular compounds different? Ionic Compounds ...
BEAT_Sheet_for_Atoms_2016_ACA
... Determine the number of protons, neutrons and electrons in isotopes written in general format and hyphen notation. ...
... Determine the number of protons, neutrons and electrons in isotopes written in general format and hyphen notation. ...
Grade 10 NSC Chemistry Curriculum
... covalent molecules, names and formulae of covalent compounds. • Ionic bonding: transfer of electrons in the formation of ionic bonding, cations and anions, electron diagrams of simple ionic compounds. Ionic structure as illustrated by sodium chloride • Revise the writing of names when given the form ...
... covalent molecules, names and formulae of covalent compounds. • Ionic bonding: transfer of electrons in the formation of ionic bonding, cations and anions, electron diagrams of simple ionic compounds. Ionic structure as illustrated by sodium chloride • Revise the writing of names when given the form ...
2. Essential Chemistry
... o When an atom or molecule gains electrons, it becomes negatively charged. For example when Cl gains an electron it becomes Cl-. o Negatively charged ions are called anions. o An atom or molecule can lose more than one electron. o When molecules loose electrons, polyatomic ions are formed. ...
... o When an atom or molecule gains electrons, it becomes negatively charged. For example when Cl gains an electron it becomes Cl-. o Negatively charged ions are called anions. o An atom or molecule can lose more than one electron. o When molecules loose electrons, polyatomic ions are formed. ...
SCH3U Course Review
... Ionization energies tend to increase with increasing atomic radii decrease with increasing nuclear charge decrease across a period from left to right increase across a period from left to right increase as you go down a family ...
... Ionization energies tend to increase with increasing atomic radii decrease with increasing nuclear charge decrease across a period from left to right increase across a period from left to right increase as you go down a family ...
Safety - Wando High School
... 2. What happens with the electrons in an ionic and covalent bond? 3. Why do atoms bond? 4. In a chemical formula what do the symbols and numbers represent? 5. What is a molecule? Is CO2 a molecule? Is NaCl a molecule? 6. What is an elements oxidation number(s)? 7. What rule does bonding (typically) ...
... 2. What happens with the electrons in an ionic and covalent bond? 3. Why do atoms bond? 4. In a chemical formula what do the symbols and numbers represent? 5. What is a molecule? Is CO2 a molecule? Is NaCl a molecule? 6. What is an elements oxidation number(s)? 7. What rule does bonding (typically) ...
science 1 small-group tutorial scheme
... List all the subshells and orbitals for which the principal quantum number, n, has the values 2 and 3 in order of increasing energy, according to the quantum mechanical rules. Use clearly-labelled diagrams to illustrate the shapes and orientations of the orbitals for which the angular momentum (subs ...
... List all the subshells and orbitals for which the principal quantum number, n, has the values 2 and 3 in order of increasing energy, according to the quantum mechanical rules. Use clearly-labelled diagrams to illustrate the shapes and orientations of the orbitals for which the angular momentum (subs ...
Chapter 18 Resource: Matter
... 1. The building blocks of matter are (atoms, compounds). 2. Isotopes are atoms of the same element that have different numbers of (neutrons, protons). 3. Electrically charged atoms are (electrons, ions). 4. An example of a (compound, mixture) is water. 5. The (chemical, physical) properties of an el ...
... 1. The building blocks of matter are (atoms, compounds). 2. Isotopes are atoms of the same element that have different numbers of (neutrons, protons). 3. Electrically charged atoms are (electrons, ions). 4. An example of a (compound, mixture) is water. 5. The (chemical, physical) properties of an el ...
Infrared Spectroscopy
... will always be a dipole moment unless 1. bonds are composed of the same element (having two atoms with similar electronegativity is not similar enough), or 2. polar covalent bonds are arranged symmetrically. Note that you need to consider all resonance structures when evaluating symmetry. ...
... will always be a dipole moment unless 1. bonds are composed of the same element (having two atoms with similar electronegativity is not similar enough), or 2. polar covalent bonds are arranged symmetrically. Note that you need to consider all resonance structures when evaluating symmetry. ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.