Ch. 2-2 Properties of Water
... • Because of their partial positive and negative charges, polar molecules such as water can attract each other. The attraction between the hydrogen atom on one water molecule and the oxygen atom on another water molecule is an example of a hydrogen bond ...
... • Because of their partial positive and negative charges, polar molecules such as water can attract each other. The attraction between the hydrogen atom on one water molecule and the oxygen atom on another water molecule is an example of a hydrogen bond ...
Chapter 2 (Hill/Petrucci/McCreary/Perry This chapter deals with
... Chemical compounds have constant, unvarying compositions with respect to weights of elements which make them up. Law of Constant Composition (Definite Proportions) "In a compound, elements are always present in a definite proportion by weight." Thus, water, a compound is always 11 parts H and 89 par ...
... Chemical compounds have constant, unvarying compositions with respect to weights of elements which make them up. Law of Constant Composition (Definite Proportions) "In a compound, elements are always present in a definite proportion by weight." Thus, water, a compound is always 11 parts H and 89 par ...
SG2 Atoms and Atomic Structure
... d) Use periodic table to identify the mass number of the most abundant isotope of an element e) Use periodic table to determine the number of protons, neutrons, and electrons in a neutral atom f) Discovery of isotopes and what they are g) Calculate average atomic mass of an element from mass number ...
... d) Use periodic table to identify the mass number of the most abundant isotope of an element e) Use periodic table to determine the number of protons, neutrons, and electrons in a neutral atom f) Discovery of isotopes and what they are g) Calculate average atomic mass of an element from mass number ...
Unit 1 Review, pages 138–145
... 34. If an atom has high ionization energy, its electron affinity is also high. 35. (a) An electrolyte is a compound that conducts an electric current when it is dissolved in water. (b) Ionic compounds are electrolytes because they separate into ions when they are in solution. These charged entities ...
... 34. If an atom has high ionization energy, its electron affinity is also high. 35. (a) An electrolyte is a compound that conducts an electric current when it is dissolved in water. (b) Ionic compounds are electrolytes because they separate into ions when they are in solution. These charged entities ...
Chemistry Name______________________________________
... electrons move only in specific circular orbits. While there are many different orbits they can only occupy one at a time. They will gain energy to jump to higher orbit and lose energy to fall to lower lowest energy for atom (all electrons in orbits closest to nucleus) couldnot explain other atom’s ...
... electrons move only in specific circular orbits. While there are many different orbits they can only occupy one at a time. They will gain energy to jump to higher orbit and lose energy to fall to lower lowest energy for atom (all electrons in orbits closest to nucleus) couldnot explain other atom’s ...
Chemistry Study Guide
... react with other forms of matter. For example, some substances are flammable. If they are heated with oxygen, they will react and burst into flames. The ability of a substance to combine with oxygen is an example of chemical property. ...
... react with other forms of matter. For example, some substances are flammable. If they are heated with oxygen, they will react and burst into flames. The ability of a substance to combine with oxygen is an example of chemical property. ...
Chemistry Study Guide
... react with other forms of matter. For example, some substances are flammable. If they are heated with oxygen, they will react and burst into flames. The ability of a substance to combine with oxygen is an example of chemical property. ...
... react with other forms of matter. For example, some substances are flammable. If they are heated with oxygen, they will react and burst into flames. The ability of a substance to combine with oxygen is an example of chemical property. ...
The Chemical Context of Life
... number of protons in its nucleus (ex. 2He) • An element’s mass number 質量數 is the sum of protons + neutrons in the nucleus (ex. 24He or 1123Na) • Atomic mass 原子量, the atom’s total mass, can be approximated by the mass number (ex. Na is 23 daltons (22.9898 daltons precisely)) Copyright © 2008 Pearson ...
... number of protons in its nucleus (ex. 2He) • An element’s mass number 質量數 is the sum of protons + neutrons in the nucleus (ex. 24He or 1123Na) • Atomic mass 原子量, the atom’s total mass, can be approximated by the mass number (ex. Na is 23 daltons (22.9898 daltons precisely)) Copyright © 2008 Pearson ...
Semester Exam Review Guide
... b. the total number of protons, electrons, and neutrons is increasing c. electrons are repelling from each other in the valence shell d. elements are becoming very reactive 17. The atomic mass number is equal to the number of a. protons b. neutrons c. protons and neutrons d. protons and electrons 18 ...
... b. the total number of protons, electrons, and neutrons is increasing c. electrons are repelling from each other in the valence shell d. elements are becoming very reactive 17. The atomic mass number is equal to the number of a. protons b. neutrons c. protons and neutrons d. protons and electrons 18 ...
Formula and The Mole
... arrangement which is the same as the noble gases. 3. A covalent bond is the attraction of the positive nucleus for a shared pair of electrons. 4. Covalent bonds are strong bonds between two non-metal atoms. ...
... arrangement which is the same as the noble gases. 3. A covalent bond is the attraction of the positive nucleus for a shared pair of electrons. 4. Covalent bonds are strong bonds between two non-metal atoms. ...
Introduction to Oxidation Reduction
... The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or ...
... The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or ...
IntroRedoxDCIAns
... The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or ...
... The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. General statement-the reactions show the disappearance or ...
Exam Review - hrsbstaff.ednet.ns.ca
... Rutherford's observation that a gold foil scatters some alpha particle through angles greater than 90º enabled him to conclude that a) all atoms are electrically neutral. b) the nucleus of the atom contains the positive charge. c) an electron has a very small mass. d) electrons are a part of all mat ...
... Rutherford's observation that a gold foil scatters some alpha particle through angles greater than 90º enabled him to conclude that a) all atoms are electrically neutral. b) the nucleus of the atom contains the positive charge. c) an electron has a very small mass. d) electrons are a part of all mat ...
Electron Configurations
... probably didn’t have much trouble with these concepts. Otherwise, you may want some extra information on the subject. Most of this below is “borrowed” from Sparknotes.com. The first and most important rule to remember when attempting to determine how electrons will be arranged in the atom is Hund’s ...
... probably didn’t have much trouble with these concepts. Otherwise, you may want some extra information on the subject. Most of this below is “borrowed” from Sparknotes.com. The first and most important rule to remember when attempting to determine how electrons will be arranged in the atom is Hund’s ...
SCIENCE 10: Chemical Reactions – Atomic Structure
... The element copper forms two different compounds with chlorine. Chlorine always forms a 1- ion. Copper can form either a 1+ ion or a 2+ ion. CuCl = copper (I) chloride CuCl2 = copper (II) chloride Naming Ionic Compounds: (p.194) o Metal name first, non-metal name second o Change the ending of the ...
... The element copper forms two different compounds with chlorine. Chlorine always forms a 1- ion. Copper can form either a 1+ ion or a 2+ ion. CuCl = copper (I) chloride CuCl2 = copper (II) chloride Naming Ionic Compounds: (p.194) o Metal name first, non-metal name second o Change the ending of the ...
Physics 107: Ideas of Modern Physics
... 1. A scientist is trying to eject electrons from a metal by shining a light on it, but none are coming out. To eject electrons, she should change the light by… a. decreasing the frequency b. increasing the frequency c. increasing the intensity d. increasing the wavelength e. asking Einstein 2. A bet ...
... 1. A scientist is trying to eject electrons from a metal by shining a light on it, but none are coming out. To eject electrons, she should change the light by… a. decreasing the frequency b. increasing the frequency c. increasing the intensity d. increasing the wavelength e. asking Einstein 2. A bet ...
Ideas of Modern Physics
... 1. A scientist is trying to eject electrons from a metal by shining a light on it, but none are coming out. To eject electrons, she should change the light by… a. decreasing the frequency b. increasing the frequency c. increasing the intensity d. increasing the wavelength e. asking Einstein 2. A bet ...
... 1. A scientist is trying to eject electrons from a metal by shining a light on it, but none are coming out. To eject electrons, she should change the light by… a. decreasing the frequency b. increasing the frequency c. increasing the intensity d. increasing the wavelength e. asking Einstein 2. A bet ...
First Law of Thermodynamics:
... The steric number of an inner atom uniquely determines the number and type of hybrid orbitals. Hybrid orbitals form localized bonds by overlap with atomic orbitals or with other hybrid orbitals. There is no need to hybridize orbitals on outer atoms, because atoms do not have limiting geometries. The ...
... The steric number of an inner atom uniquely determines the number and type of hybrid orbitals. Hybrid orbitals form localized bonds by overlap with atomic orbitals or with other hybrid orbitals. There is no need to hybridize orbitals on outer atoms, because atoms do not have limiting geometries. The ...
Electron Configuration Notes
... • electrons move around nucleus in orbits similar to how planets orbit the sun • energy levels for electrons are quantized Major developments that put Bohr’s Model into question: Einstein: Light energy exhibits properties of matter. Matter and energy are different forms of the same thing. De Broglie ...
... • electrons move around nucleus in orbits similar to how planets orbit the sun • energy levels for electrons are quantized Major developments that put Bohr’s Model into question: Einstein: Light energy exhibits properties of matter. Matter and energy are different forms of the same thing. De Broglie ...
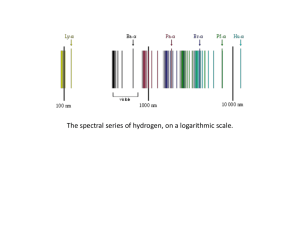
Rutherford–Bohr model
... The Rutherford–Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jump between orbits is accompanied by an emitted or absorbed amount of ...
... The Rutherford–Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jump between orbits is accompanied by an emitted or absorbed amount of ...
List of Definitions for AS Chemistry
... protein involving folding or coiling of the chains. It shows how protein molecules are arranged in relation to each other. There are four types of R group interactions which hold the tertiary structure in its shape. van der Waals’ forces (induced dipole-induced dipole bonding) exist when non-polar ...
... protein involving folding or coiling of the chains. It shows how protein molecules are arranged in relation to each other. There are four types of R group interactions which hold the tertiary structure in its shape. van der Waals’ forces (induced dipole-induced dipole bonding) exist when non-polar ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.