m5zn_307118e6dc84400
... Salts of Carboxylic Acids Carboxylic acids are neutralized by strong bases to ...
... Salts of Carboxylic Acids Carboxylic acids are neutralized by strong bases to ...
pH and pOH (cont.)
... − NH4Cl is the salt of a weak base (NH3) and strong acid (HCl). − When dissolved in water, the salt dissociates into ammonium ions and chloride ions. ...
... − NH4Cl is the salt of a weak base (NH3) and strong acid (HCl). − When dissolved in water, the salt dissociates into ammonium ions and chloride ions. ...
Full answers
... • Draw a Lewis structure and thus determine the geometry of the ICl4– ion. (The I is the central atom.) ...
... • Draw a Lewis structure and thus determine the geometry of the ICl4– ion. (The I is the central atom.) ...
Are You suprised ?
... For the following reaction 2 NH3(g) ⇌ N2(g) + 3 H2(g) if, Kc = 2.6×10-5 mol2 L-2 at 127oC. Then Kp at the same temperature is: A) 2.8×10-2 atm2 C) 8.2×10-4 atm2 ...
... For the following reaction 2 NH3(g) ⇌ N2(g) + 3 H2(g) if, Kc = 2.6×10-5 mol2 L-2 at 127oC. Then Kp at the same temperature is: A) 2.8×10-2 atm2 C) 8.2×10-4 atm2 ...
Masterton and Hurley Chapter 4
... Simple Solubility Rules: SAP (compounds containing sodium, ammonium, and potassium are soluble) CAN (chlorate, acetate, and nitrate containing compounds are soluble) ...
... Simple Solubility Rules: SAP (compounds containing sodium, ammonium, and potassium are soluble) CAN (chlorate, acetate, and nitrate containing compounds are soluble) ...
Chemical Bonding
... Since both H2O and CH3OH are hydrogen bonded the viscosities are high, but when there is substitution of second methyl group to produce the non-hydrogen-bonded dimethyl-ether, (CH3)2O, the viscosity drops to a low value. Polyhydroxy alcohols such as ethylene glycol, CH2OH.CH2OH and glycerol, CH2OH.C ...
... Since both H2O and CH3OH are hydrogen bonded the viscosities are high, but when there is substitution of second methyl group to produce the non-hydrogen-bonded dimethyl-ether, (CH3)2O, the viscosity drops to a low value. Polyhydroxy alcohols such as ethylene glycol, CH2OH.CH2OH and glycerol, CH2OH.C ...
Dr. Arrington Exam 3
... (c) pH is determined in three ways during a titration: (i) pH calculated from the excess concentration of H+ or OH− in solution. (ii) pH calculated from the Henderson-Hasselbalch equation (buffer equation) (iii) pH calculated from a weak acid or weak base equilibrium calculation. Place the calculati ...
... (c) pH is determined in three ways during a titration: (i) pH calculated from the excess concentration of H+ or OH− in solution. (ii) pH calculated from the Henderson-Hasselbalch equation (buffer equation) (iii) pH calculated from a weak acid or weak base equilibrium calculation. Place the calculati ...
Solution
... Nuclear chemistry is used to treat some forms of disease. For example, thyroid diseases can be treated using iodine-131 (I-131). Once absorbed, the radioisotope decays emitting beta particles that destroy the surrounding tissue. a) Write the nuclear equation for this decay. ...
... Nuclear chemistry is used to treat some forms of disease. For example, thyroid diseases can be treated using iodine-131 (I-131). Once absorbed, the radioisotope decays emitting beta particles that destroy the surrounding tissue. a) Write the nuclear equation for this decay. ...
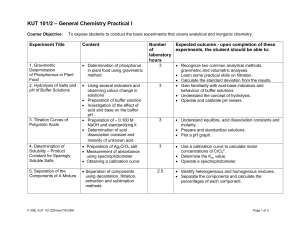
KUT 101/2 – General Chemistry Practical I
... • Preparation of Na2S2O3 solution and standardizing it • Determination of the oxidizing capacity of an unknown liquid bleach • Preparation of Cu(NO)3 and performing basic laboratory procedures • Reduction of copper with zinc • Preparation of ∼ 0.100 M NaOH and standardizing it. • Analysis of an unkn ...
... • Preparation of Na2S2O3 solution and standardizing it • Determination of the oxidizing capacity of an unknown liquid bleach • Preparation of Cu(NO)3 and performing basic laboratory procedures • Reduction of copper with zinc • Preparation of ∼ 0.100 M NaOH and standardizing it. • Analysis of an unkn ...
CHEMISTRY FINAL EXAM REVIEW SHEET
... Chapter 5: Models of the Atom Terms: Proton: +1 charge, located in nucleus, relative mass = 1 amu Neutron: No charge, located in nucleus, relative mass = 1 amu Electron: -1 charge, located outside of nucleus, relative mass = 1/1840 amu Atomic Number = number of protons in an element. Mass Number = n ...
... Chapter 5: Models of the Atom Terms: Proton: +1 charge, located in nucleus, relative mass = 1 amu Neutron: No charge, located in nucleus, relative mass = 1 amu Electron: -1 charge, located outside of nucleus, relative mass = 1/1840 amu Atomic Number = number of protons in an element. Mass Number = n ...
Notes on QA - Scarsdale Public Schools
... To understand what happens in the reaction we need to also review LeChatelier’s principle for systems at equilibrium: If a stress is applied to an equilibrium system, the system moves (shifts) in the direction that relieves the stress. Consider our equilibrium system: Cu(OH)2(s) = Cu2+(aq) + 2OH-(aq ...
... To understand what happens in the reaction we need to also review LeChatelier’s principle for systems at equilibrium: If a stress is applied to an equilibrium system, the system moves (shifts) in the direction that relieves the stress. Consider our equilibrium system: Cu(OH)2(s) = Cu2+(aq) + 2OH-(aq ...
Review Package
... 5. Ionic Compounds (Textbook p. 139-146; 148-149) Terminology (ion, cation, anion, ionic charge/combining capacity, valence electron, stable octet, polyatomic ion, binary compound, ternary compound, ionic bond) Draw Bohr-Rutherford diagrams/Lewis Dot structures to show the formation of ionic com ...
... 5. Ionic Compounds (Textbook p. 139-146; 148-149) Terminology (ion, cation, anion, ionic charge/combining capacity, valence electron, stable octet, polyatomic ion, binary compound, ternary compound, ionic bond) Draw Bohr-Rutherford diagrams/Lewis Dot structures to show the formation of ionic com ...
Chemical reaction
... • Polar nature of water causes water molecules to be attracted to each other. • This attraction: hydrogen bond. ...
... • Polar nature of water causes water molecules to be attracted to each other. • This attraction: hydrogen bond. ...
Acid-on-the-Fly
... © 2009 Halliburton. All rights reserved. Sales of Halliburton products and services will be in accord solely with the terms and conditions contained in the contract between Halliburton and the customer that is applicable to the sale. ...
... © 2009 Halliburton. All rights reserved. Sales of Halliburton products and services will be in accord solely with the terms and conditions contained in the contract between Halliburton and the customer that is applicable to the sale. ...
Introduction to Organic Chemistry 2 ed William H. Brown
... • the relative acidity of HA within a period is determined by the stability of A• the greater the electronegativity, the greater the ...
... • the relative acidity of HA within a period is determined by the stability of A• the greater the electronegativity, the greater the ...
Final Review
... 4. What is the volume of one mole of gas at STP? a. 1 liter b. 12 liters c. 22.4 liters d. It depends on the gas since all gases have different densities 5. Which of the following is NOT true at STP conditions? a. Temperature is at 0°C and pressure is at 1 atm b. Temperature is at 273 K and pressure ...
... 4. What is the volume of one mole of gas at STP? a. 1 liter b. 12 liters c. 22.4 liters d. It depends on the gas since all gases have different densities 5. Which of the following is NOT true at STP conditions? a. Temperature is at 0°C and pressure is at 1 atm b. Temperature is at 273 K and pressure ...
CAMBRIDGE INTERNATIONAL EXAMINATIONS
... A gas Y, is less dense than air, very soluble in water and is an alkali. Which method is used to collect a dry sample of the gas? A ...
... A gas Y, is less dense than air, very soluble in water and is an alkali. Which method is used to collect a dry sample of the gas? A ...