Tutorial 6 Writing Chemical Formulas for Molecular Compounds and
... • Molecular compounds form when nonmetals share electrons to form covalent bonds. The formula for a molecular compound shows the number of atoms that are combined in one molecule. Molecular compounds ...
... • Molecular compounds form when nonmetals share electrons to form covalent bonds. The formula for a molecular compound shows the number of atoms that are combined in one molecule. Molecular compounds ...
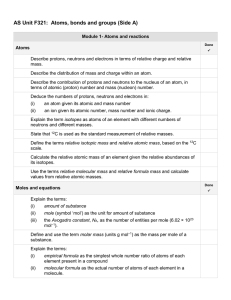
AS Unit F321 Unit 1 Side A check list
... State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. State the formulae of the common alkalis: sodium hydroxide, pot ...
... State the formulae of the common acids: hydrochloric, 2ulphuric and nitric acids. State that common bases are metal oxides, metal hydroxides and ammonia. State that an alkali is a soluble base that releases OH– ions in aqueous solution. State the formulae of the common alkalis: sodium hydroxide, pot ...
Chapters 6, 8
... Metal gives away its electron(s) and becomes positively charged cation. Nonmetal accepts electron(s) and becomes negatively charged anion. Ionic compound is a result of attraction between oppositely charged ions. ...
... Metal gives away its electron(s) and becomes positively charged cation. Nonmetal accepts electron(s) and becomes negatively charged anion. Ionic compound is a result of attraction between oppositely charged ions. ...
Chapter 2 Notes - Duplin County Schools
... nucleus in energy levels First energy level -- 2e Second energy level -- 8e Third energy level -- 18e ...
... nucleus in energy levels First energy level -- 2e Second energy level -- 8e Third energy level -- 18e ...
Atoms, molecules and ions
... • When two elements form several compounds, Greek prefixes are used to specify the number of atoms of each element – e.g.; CO : carbon monoxide CO2 : carbon dioxide NO2 : nitrogen dioxide N2O4 : dinitrogen tetroxide • Many covalent compounds containing hydrogen are referred to by their common name – ...
... • When two elements form several compounds, Greek prefixes are used to specify the number of atoms of each element – e.g.; CO : carbon monoxide CO2 : carbon dioxide NO2 : nitrogen dioxide N2O4 : dinitrogen tetroxide • Many covalent compounds containing hydrogen are referred to by their common name – ...
Chapter 4
... Completely ionized in water to give either H3O+ or OHGood conductors of electricity. Directional arrow () indicates dissociation is complete ...
... Completely ionized in water to give either H3O+ or OHGood conductors of electricity. Directional arrow () indicates dissociation is complete ...
3.10 Neutralization
... • Net ionic equations for reactions between strong acids and bases HCl(aq) + KOH(aq) → KCl(aq) + H2O(l) H+ + Cl- + K+ + OH- → K+ + Cl- + H2O(l) ⇒H+ + OH- → H2O(l) – H+ is present in the form of H3O+ ...
... • Net ionic equations for reactions between strong acids and bases HCl(aq) + KOH(aq) → KCl(aq) + H2O(l) H+ + Cl- + K+ + OH- → K+ + Cl- + H2O(l) ⇒H+ + OH- → H2O(l) – H+ is present in the form of H3O+ ...
Review #7: Solutions, Acids and Bases 1. Definitions: a) Solution: a
... are very good conductors of electricity and have very low pH (<4.5). The six common strong acids are: HCl (aq), HBr (aq), HI (aq), HClO3 (aq), HNO3 (aq), and H2SO4 (aq). A weak acid is an acid that does not ionize very much when it is dissolved in water, so most of the acid molecules stay intact as ...
... are very good conductors of electricity and have very low pH (<4.5). The six common strong acids are: HCl (aq), HBr (aq), HI (aq), HClO3 (aq), HNO3 (aq), and H2SO4 (aq). A weak acid is an acid that does not ionize very much when it is dissolved in water, so most of the acid molecules stay intact as ...
Weathering and Erosion - School District 67 Okanagan Skaha
... • Water weathers rock by dissolving it • Water combines with some minerals (eg. Feldspar) to produce clay • Chemical reaction of water with other substances is called hydrolysis ...
... • Water weathers rock by dissolving it • Water combines with some minerals (eg. Feldspar) to produce clay • Chemical reaction of water with other substances is called hydrolysis ...
chemical reaction
... the oxidation state(O.S) of an atom in the pure (uncombined) element is 0. The total (sum) of the oxidation state of all the atoms in a molecule or formula unit is 0. For an ion total of the oxidation state is equal to the charge on the ion. In their compounds the alkali metals (1a groups Li, Na, K, ...
... the oxidation state(O.S) of an atom in the pure (uncombined) element is 0. The total (sum) of the oxidation state of all the atoms in a molecule or formula unit is 0. For an ion total of the oxidation state is equal to the charge on the ion. In their compounds the alkali metals (1a groups Li, Na, K, ...
The Ka values of water and the hydronium ion for comparison with
... Journal of Chemical Education ...
... Journal of Chemical Education ...
Unit 2: Atoms and Ions Homework Booklet
... c. When coal is burned a gas is given off that is weakly soluble in water. What is the gas and what is formed when it dissolves in water? ...
... c. When coal is burned a gas is given off that is weakly soluble in water. What is the gas and what is formed when it dissolves in water? ...
AP Matter Class Packet Unit 5
... Predict the products of and balance the following reactions: ___ HF (aq) + ___ LiOH (aq) → __________ (aq) + ___ HOH (l) ___ HCl (aq) + ___ Ca(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ HClO3 (aq) + ___ Mg(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ H2CO3 (aq) + ___ NaOH (aq) → __________(aq) + _ ...
... Predict the products of and balance the following reactions: ___ HF (aq) + ___ LiOH (aq) → __________ (aq) + ___ HOH (l) ___ HCl (aq) + ___ Ca(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ HClO3 (aq) + ___ Mg(OH)2 (aq) → __________(aq) + ___ HOH (l) ___ H2CO3 (aq) + ___ NaOH (aq) → __________(aq) + _ ...
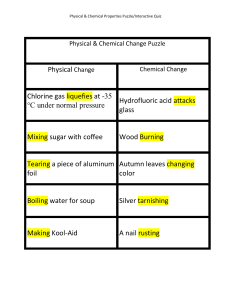
Physical Change Chlorine gas liquefies at
... Physical & Chemical Properties Puzzle/Interactive Quiz ...
... Physical & Chemical Properties Puzzle/Interactive Quiz ...
Percent Ionization
... 3.) A salt of a weak base and a strong acid. - the cation of the salt is the conjugate of the weak base. It hydrolyzes to give an acidic solution - eg. NH4Cl 4.) A salt of a weak base and a weak acid. - both ions hydrolyze. You must compare the Ka of the cation with the Kb of the anion - if the Ka o ...
... 3.) A salt of a weak base and a strong acid. - the cation of the salt is the conjugate of the weak base. It hydrolyzes to give an acidic solution - eg. NH4Cl 4.) A salt of a weak base and a weak acid. - both ions hydrolyze. You must compare the Ka of the cation with the Kb of the anion - if the Ka o ...
Slide 1
... Acidic proton on a hydroxyl group bonded or coordinated to a central atom on which there is an oxo (=O) group ...
... Acidic proton on a hydroxyl group bonded or coordinated to a central atom on which there is an oxo (=O) group ...
honors final key
... 2. An ionic compound is made up of a __cation_______ and a _anion_______. The first element is ALWAYS the _cation______, which forms a _ion______ with a _positive______ charge. The second element is the __anion______, which forms an _ion_________ with a __negative________ charge. a. Give three examp ...
... 2. An ionic compound is made up of a __cation_______ and a _anion_______. The first element is ALWAYS the _cation______, which forms a _ion______ with a _positive______ charge. The second element is the __anion______, which forms an _ion_________ with a __negative________ charge. a. Give three examp ...
Prelim Revision Paper 4
... Which line in the table describes what happens to a dilute solution of hydrochloric acid when water is ...
... Which line in the table describes what happens to a dilute solution of hydrochloric acid when water is ...
Inorganic Chemistry Basics
... Inorganic Chemistry Basics Lewis acid = electron pair acceptor (e.g. H+, Al3+, BF3, CO2, SiF4) Lewis base = electron pair donor (e.g. NH3, PF3, Hal-, HS-, H2O, OH-) Lewis acids and bases form Lewis acid/base adducts (e.g. BF3←NH3) ...
... Inorganic Chemistry Basics Lewis acid = electron pair acceptor (e.g. H+, Al3+, BF3, CO2, SiF4) Lewis base = electron pair donor (e.g. NH3, PF3, Hal-, HS-, H2O, OH-) Lewis acids and bases form Lewis acid/base adducts (e.g. BF3←NH3) ...
Carboxylic Acids
... the name of the parent chain and is replaced -anoic acid. Since a carboxylic acid group must always lie at the end of a carbon chain, it is always is given the #1 location position in numbering and it is not necessary to include it in the name. Many carboxylic acids are called by the common names. T ...
... the name of the parent chain and is replaced -anoic acid. Since a carboxylic acid group must always lie at the end of a carbon chain, it is always is given the #1 location position in numbering and it is not necessary to include it in the name. Many carboxylic acids are called by the common names. T ...