acids - WordPress.com
... • In this method, both the reactants used are in the aqueous state thus excess reagent cannot be easily removed from the product which is in the same state. Thus exact quantities of reactants must be used to ensure that product is not contaminated by excess reagent. • The two main steps involved ar ...
... • In this method, both the reactants used are in the aqueous state thus excess reagent cannot be easily removed from the product which is in the same state. Thus exact quantities of reactants must be used to ensure that product is not contaminated by excess reagent. • The two main steps involved ar ...
Review for Physical Science Test #2
... To tell a strong acid from a weak acid To tell an acid from a neutral solution To tell a strong base from a weak base To create a temporary tattoo on your little sister’s face right before picture day. ...
... To tell a strong acid from a weak acid To tell an acid from a neutral solution To tell a strong base from a weak base To create a temporary tattoo on your little sister’s face right before picture day. ...
Answers to Review Questions
... 1. Do all atoms of an element have the same atomic number? The same atomic mass? Explain. Atomic number is based on the number of protons, hence all atoms of a particular element have the same atomic number. Isotopes vary in the number of neutrons, hence a different atomic mass. 2. How many atoms wo ...
... 1. Do all atoms of an element have the same atomic number? The same atomic mass? Explain. Atomic number is based on the number of protons, hence all atoms of a particular element have the same atomic number. Isotopes vary in the number of neutrons, hence a different atomic mass. 2. How many atoms wo ...
Ei otsikkoa
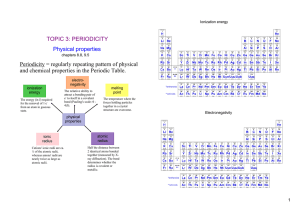
... Transition elements show characteristic properties: The acidity of aqueous solutions of chlorides increases across the period: NaCl : MgCl2 : AlCl3 : SiCl4 : PCl3 : PCl5 : Cl2 : ...
... Transition elements show characteristic properties: The acidity of aqueous solutions of chlorides increases across the period: NaCl : MgCl2 : AlCl3 : SiCl4 : PCl3 : PCl5 : Cl2 : ...
Semester 2 Review
... A. How will an increase in temperature change the concentration of hydrogen gas? ________ B. How will an increase in pressure affect the system? ___________________ C. Which direction will the addition of iodine gas shift the system? _________________ What does this do to the concentration of hydrog ...
... A. How will an increase in temperature change the concentration of hydrogen gas? ________ B. How will an increase in pressure affect the system? ___________________ C. Which direction will the addition of iodine gas shift the system? _________________ What does this do to the concentration of hydrog ...
Water Chemistry - Biology12-Lum
... Although a water molecule has an overall neutral charge (having the same number of electrons and protons), the electrons are asymmetrically distributed, which makes the molecule polar. The oxygen nucleus draws electrons away from the hydrogen nuclei, leaving these nuclei with a small net positive ch ...
... Although a water molecule has an overall neutral charge (having the same number of electrons and protons), the electrons are asymmetrically distributed, which makes the molecule polar. The oxygen nucleus draws electrons away from the hydrogen nuclei, leaving these nuclei with a small net positive ch ...
IGCSE Revision document
... 1. empirical formula = CH3 Mr = 30 2. empirical formula = NH3 Mr = 17 3. empirical formula = CH2 Mr = 98 4. empirical formula = CH Mr = 78 Calculate the percentage of the elements shown in the following compounds: C in CO N in (NH4)2SO4 O in Al(OH)3 O in Na2CO3.10H2O ...
... 1. empirical formula = CH3 Mr = 30 2. empirical formula = NH3 Mr = 17 3. empirical formula = CH2 Mr = 98 4. empirical formula = CH Mr = 78 Calculate the percentage of the elements shown in the following compounds: C in CO N in (NH4)2SO4 O in Al(OH)3 O in Na2CO3.10H2O ...
Summary notes - Kelso High School
... You can find out if a substance is an acid or and alkali by dissolving it in water and adding an indicator. An indicator is a chemical which changes colour in different pH environments so by matching the colour of the solution to the colour chart, the pH of the substance can be found. The two most c ...
... You can find out if a substance is an acid or and alkali by dissolving it in water and adding an indicator. An indicator is a chemical which changes colour in different pH environments so by matching the colour of the solution to the colour chart, the pH of the substance can be found. The two most c ...
Instructions for AP/IB 2 Chem Summer Assignment Note
... Learn the general formula for each type of reaction. If the reaction occurs in water solution, you must give the net ionic equation. If it doesn't occur in aqueous solution, the atoms/molecules do not exist as ions. ...
... Learn the general formula for each type of reaction. If the reaction occurs in water solution, you must give the net ionic equation. If it doesn't occur in aqueous solution, the atoms/molecules do not exist as ions. ...
Aqueous chemistry is a very important component to laboratory
... The partially negative oxygen atom in water will surround the positive cations, while the partially positive hydrogen atoms will surround the negative anions. If two electrodes (conductors of electricity) are placed in a solution and connected to a battery, the cations will migrate through the solut ...
... The partially negative oxygen atom in water will surround the positive cations, while the partially positive hydrogen atoms will surround the negative anions. If two electrodes (conductors of electricity) are placed in a solution and connected to a battery, the cations will migrate through the solut ...
Decomposition Reaction
... Activity Series of Metals 1. Each element on the list replaces from a compound any of the elements below it. The larger the interval between elements, the more vigorous the reaction. 2. The first five elements (lithium - sodium) are known as very active metals and they react with cold water to produ ...
... Activity Series of Metals 1. Each element on the list replaces from a compound any of the elements below it. The larger the interval between elements, the more vigorous the reaction. 2. The first five elements (lithium - sodium) are known as very active metals and they react with cold water to produ ...
Chapter 4: Solution Chemistry and the Hydrosphere
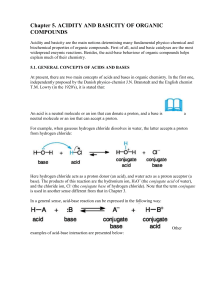
... • An acid and base that differ only by the presence of H+ are conjugate acid-base pairs. • The general reaction for the dissociation (or ionization) of an acid can be represented as above, where the double-arrow indicates both the forward and reverse reactions can occur. • Note: The double arrow (⇄) ...
... • An acid and base that differ only by the presence of H+ are conjugate acid-base pairs. • The general reaction for the dissociation (or ionization) of an acid can be represented as above, where the double-arrow indicates both the forward and reverse reactions can occur. • Note: The double arrow (⇄) ...
Homework Exercises
... The concentrated ammonia solution is diluted exactly 100 times. The diluted solution of ammonia is titrated with 0.1 moll-1 hydrochloric acid. ...
... The concentrated ammonia solution is diluted exactly 100 times. The diluted solution of ammonia is titrated with 0.1 moll-1 hydrochloric acid. ...
Slide 1
... D(DG) = -RTefflnkeq = -RTeffln(k3/k2) = RTeffln(k2/k3) The ratio is equal to the product ratio, which is to a good approximation equal to the ratio of the intensities for these two ions in the MS/MS spectrum: ...
... D(DG) = -RTefflnkeq = -RTeffln(k3/k2) = RTeffln(k2/k3) The ratio is equal to the product ratio, which is to a good approximation equal to the ratio of the intensities for these two ions in the MS/MS spectrum: ...
2009-10 Chemistry 1st Semester Final Exam Topics and Review
... e. One of the problems with space travel is the building up of carbon dioxide produced by the astronauts. The typical procedure is to react the carbon dioxide with lithium hydroxide to form lithium carbonate and liquid water. 36. Write and balance a chemical equation for this reaction. a. Aluminum m ...
... e. One of the problems with space travel is the building up of carbon dioxide produced by the astronauts. The typical procedure is to react the carbon dioxide with lithium hydroxide to form lithium carbonate and liquid water. 36. Write and balance a chemical equation for this reaction. a. Aluminum m ...
Chapter 5. ACIDITY AND BASICITY OF ORGANIC COMPOUNDS
... Another stabilizing factor is the polarizability (opposed to polarity) of an element in the acidic site. This term means the ability of the electrons to respond to a changing electric field, as a result of its interaction with solvent or with other polar reagents. Relative polarizability increases ...
... Another stabilizing factor is the polarizability (opposed to polarity) of an element in the acidic site. This term means the ability of the electrons to respond to a changing electric field, as a result of its interaction with solvent or with other polar reagents. Relative polarizability increases ...
Introduction
... Molarity and normality vary with T, whereas molality and formality are independent of T ...
... Molarity and normality vary with T, whereas molality and formality are independent of T ...