File
... The smallest atomic unit The process of combining two light nuclei to form a heavier more stable nucleus The process of using a neutron to split a heavy nucleus into two nuclei with smaller mass numbers Brittle versus soft Stretchable Metal-like but does not contain all metal characteristics A posit ...
... The smallest atomic unit The process of combining two light nuclei to form a heavier more stable nucleus The process of using a neutron to split a heavy nucleus into two nuclei with smaller mass numbers Brittle versus soft Stretchable Metal-like but does not contain all metal characteristics A posit ...
Exam 2, Fall 2001
... Substitute Question for Page 4: Cobalt(III) ion forms many compounds with ammonia. To find the formula of one of these compounds, you titrate the NH3 in the compound with standardized acid. Co(NH3)xCl3(aq) + x HCl(aq) → x NH4+(aq) + Co3+(aq) + (x + 3) Cl-(aq) Assume that 23.63 mL of 1.500 M HCl is u ...
... Substitute Question for Page 4: Cobalt(III) ion forms many compounds with ammonia. To find the formula of one of these compounds, you titrate the NH3 in the compound with standardized acid. Co(NH3)xCl3(aq) + x HCl(aq) → x NH4+(aq) + Co3+(aq) + (x + 3) Cl-(aq) Assume that 23.63 mL of 1.500 M HCl is u ...
Name: Date: AP Chemistry/Chemistry 145 Summer Assignment
... 14. Calculate the percent composition of C12H22O11 (sugar). (Give Percent of each element.) Show all work. ...
... 14. Calculate the percent composition of C12H22O11 (sugar). (Give Percent of each element.) Show all work. ...
CHEMISTRY 1710 - Practice Exam #2
... A) strong electrolyte, weak acid C) strong electrolyte, strong acid ...
... A) strong electrolyte, weak acid C) strong electrolyte, strong acid ...
Chemistry Unit Review
... 4. Describe the difference between a physical change and a chemical change. ...
... 4. Describe the difference between a physical change and a chemical change. ...
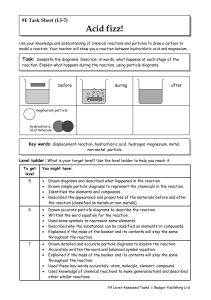
Year 9 Homework Task 9E-5 Reactions 5-7
... Explained if the mass of the beaker and its contents will stay the same throughout the reaction. Used these key words accurately: atom, molecule, element, compound. Used knowledge of chemical reactions to make generalisations and described other similar reactions. ...
... Explained if the mass of the beaker and its contents will stay the same throughout the reaction. Used these key words accurately: atom, molecule, element, compound. Used knowledge of chemical reactions to make generalisations and described other similar reactions. ...
Chapter 3
... • Molecular formulas show the kind and # of atoms in a molecule – H2O = 2 hydrogen atoms + 1 oxygen atom – CO2 = 1 carbon atom + 2 oxygen atoms – C6H12O6 = 6 carbon atoms + 12 hydrogen atoms + 6 oxygen atoms – NaCl = 1 sodium ion(Na) + 1 chlorine ion(Cl) ...
... • Molecular formulas show the kind and # of atoms in a molecule – H2O = 2 hydrogen atoms + 1 oxygen atom – CO2 = 1 carbon atom + 2 oxygen atoms – C6H12O6 = 6 carbon atoms + 12 hydrogen atoms + 6 oxygen atoms – NaCl = 1 sodium ion(Na) + 1 chlorine ion(Cl) ...
makeup2
... 5. If the hydrocarbon C2H4 is burned in oxygen gas, carbon dioxide and water are formed as described by the unbalanced chemical equation C2H4 + O2 ----> CO2 + H2O When this equation is balanced properly, we predict that one mole of C2H4 will (A) react with one mole of O2 (B) form two moles of CO2. ( ...
... 5. If the hydrocarbon C2H4 is burned in oxygen gas, carbon dioxide and water are formed as described by the unbalanced chemical equation C2H4 + O2 ----> CO2 + H2O When this equation is balanced properly, we predict that one mole of C2H4 will (A) react with one mole of O2 (B) form two moles of CO2. ( ...
Chapter 8 Brønsted-Lowry Theory of Acids and Bases
... Write the chemical reaction for the following acids or bases in water. Identify the conjugate acid base pairs. 1. HF (a weak acid) 2. H2S (a weak acid) 3. HNO3 (a strong acid) 4. CH3NH2 (a weak base) Note: The degree of dissociation also defines weak and strong bases ...
... Write the chemical reaction for the following acids or bases in water. Identify the conjugate acid base pairs. 1. HF (a weak acid) 2. H2S (a weak acid) 3. HNO3 (a strong acid) 4. CH3NH2 (a weak base) Note: The degree of dissociation also defines weak and strong bases ...
Diazotization-Coupling Reaction--
... 6. After all of the sulfanilic acid dissolves completely, remove the Erlenmeyer flask and allow it to cool to room temperature on the bench top. 7. Weigh 0.08-g sodium nitrite, NaNO2, and transfer it to the cooled Erlenmeyer flask; stir the solution until the solid dissolves. 8. Cool the 25-mL Erlen ...
... 6. After all of the sulfanilic acid dissolves completely, remove the Erlenmeyer flask and allow it to cool to room temperature on the bench top. 7. Weigh 0.08-g sodium nitrite, NaNO2, and transfer it to the cooled Erlenmeyer flask; stir the solution until the solid dissolves. 8. Cool the 25-mL Erlen ...
Intro to Soln Stoich
... Water is the “universal solvent” ◦ O-H bonds are covalent, e- not shared equally ◦ Oxygen has a slight negative, hydrogen slight positive Polar molecule Oxygen has a strong attraction to cations, hydrogen to anions ...
... Water is the “universal solvent” ◦ O-H bonds are covalent, e- not shared equally ◦ Oxygen has a slight negative, hydrogen slight positive Polar molecule Oxygen has a strong attraction to cations, hydrogen to anions ...
Solutes
... HCl (aq) H+ (aq) + Cl- (aq) Arrhenius BASES – produces hydroxide ions in aqueous solutions, bitter taste, slippery feel, high pH, and the fact that they turn litmus paper blue ...
... HCl (aq) H+ (aq) + Cl- (aq) Arrhenius BASES – produces hydroxide ions in aqueous solutions, bitter taste, slippery feel, high pH, and the fact that they turn litmus paper blue ...
Chapter 13 - "Water and Solutions"
... • Each division of the pH scale is an increase or decrease in concentration to a power of 10, which makes the scale a logarithmic scale ...
... • Each division of the pH scale is an increase or decrease in concentration to a power of 10, which makes the scale a logarithmic scale ...
Predicting Reactions • AP Chemistry CLASSIFYING REACTIONS
... Visually, a precipitate may go back into solution as a complex ion is formed. For example, Cu2+ + a little ammonia (NH4OH) will form the light blue precipitate, Cu(OH)2. With excess ammonia, the complex, Cu(NH3)42+, forms. Keywords such as "excess" and "concentrated" of any solution may indicate com ...
... Visually, a precipitate may go back into solution as a complex ion is formed. For example, Cu2+ + a little ammonia (NH4OH) will form the light blue precipitate, Cu(OH)2. With excess ammonia, the complex, Cu(NH3)42+, forms. Keywords such as "excess" and "concentrated" of any solution may indicate com ...
1 Q. If ΔrH is positive, what can you say about the reaction? 2 Q If
... strong base and explain how they differ from concentrated and dilute bases. ...
... strong base and explain how they differ from concentrated and dilute bases. ...
Last 4 Digits of USC ID:____ ____ ____ ____ Dr.
... some useful equations can be found on the last page. 5. Good luck!! =) ...
... some useful equations can be found on the last page. 5. Good luck!! =) ...