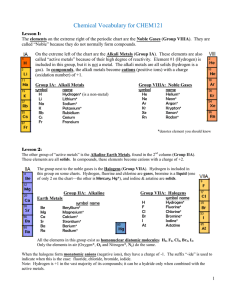
A-level Paper 1 Practice Paper 8 - A
... Octahedral and tetrahedral complex ions are produced by the reaction of transition metal ions with ligands which form co-ordinate bonds with the transition metal ion. Define the term ligand and explain what is meant by the term co-ordinate bond. ...
... Octahedral and tetrahedral complex ions are produced by the reaction of transition metal ions with ligands which form co-ordinate bonds with the transition metal ion. Define the term ligand and explain what is meant by the term co-ordinate bond. ...
Module 3 Exam Review 1. Organic chemistry is the study of which
... 4. What is the name of the functional group shown here? R--O-H hydroxyl 5. What is the name of the functional group shown here? R--S-H sulfhydryl 6. What is the name of the functional group with this formula –PO42--? phosphate 7. An organic molecule with a carbon/water ration of 1:1 is a _____. carb ...
... 4. What is the name of the functional group shown here? R--O-H hydroxyl 5. What is the name of the functional group shown here? R--S-H sulfhydryl 6. What is the name of the functional group with this formula –PO42--? phosphate 7. An organic molecule with a carbon/water ration of 1:1 is a _____. carb ...
Activity Sheet Answers - Middle School Chemistry
... type and number of atoms are in the products of the reaction as are in the reactants. No atoms are created or destroyed. 4. Reducing the amount of vinegar will reduce the amount of carbon dioxide produced because there will be fewer molecules of acetic acid to react with the baking soda and produce ...
... type and number of atoms are in the products of the reaction as are in the reactants. No atoms are created or destroyed. 4. Reducing the amount of vinegar will reduce the amount of carbon dioxide produced because there will be fewer molecules of acetic acid to react with the baking soda and produce ...
2C - Edexcel
... (b) The equation for the reaction between hydrogen and chlorine is H2 + Cl2 o 2HCl Different names are used for the product, depending on its state symbol. (i) What are the names used for HCl(g) and HCl(aq)? ...
... (b) The equation for the reaction between hydrogen and chlorine is H2 + Cl2 o 2HCl Different names are used for the product, depending on its state symbol. (i) What are the names used for HCl(g) and HCl(aq)? ...
Halogens - Cronodon
... The chlorine has displaced the bromide because chlorine is a stronger oxidising agent than bromine (equivalently bromide is a stronger reducing agent than chloride). The oxidising power of the halogens decreases in the order: F2 > Cl2 > Br2 > I2 (oxidising strength) Q.16. When a halogen oxidises ano ...
... The chlorine has displaced the bromide because chlorine is a stronger oxidising agent than bromine (equivalently bromide is a stronger reducing agent than chloride). The oxidising power of the halogens decreases in the order: F2 > Cl2 > Br2 > I2 (oxidising strength) Q.16. When a halogen oxidises ano ...
Household Acids and Bases Lab
... Household Acids and Bases Lab Introduction A visual indicator is a chemical substance that reflects the nature of the chemical system in which it is placed by changing color. Most visual indicators are complex organic molecules that exist in multiple colored forms, one of which could be colorless, d ...
... Household Acids and Bases Lab Introduction A visual indicator is a chemical substance that reflects the nature of the chemical system in which it is placed by changing color. Most visual indicators are complex organic molecules that exist in multiple colored forms, one of which could be colorless, d ...
Name__________________________ Period_______ Word
... example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants were heated. An equation can also indicate the physical state of the reactants and products. The physical states of matter symbols are listed below. ...
... example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants were heated. An equation can also indicate the physical state of the reactants and products. The physical states of matter symbols are listed below. ...
Chapter 3
... ions are pulled apart. By itself, pure water is a poor conductor of electricity. When an ionic compound dissolves, it produces ions. These ions can carry a charge through the solution and are referred to as electrolytes. ...
... ions are pulled apart. By itself, pure water is a poor conductor of electricity. When an ionic compound dissolves, it produces ions. These ions can carry a charge through the solution and are referred to as electrolytes. ...
Lotioncrafter - Stearic Acid
... Use water spray, dry chemical, foam or carbon dioxide. Water may be ineffective but should be used to keep fire-exposed containers cool. If a spill or leak has not ignited, use water spray to disperse the vapors. Water spray may be used to flush spills away from fire. Unusual Fire or Explosion Hazar ...
... Use water spray, dry chemical, foam or carbon dioxide. Water may be ineffective but should be used to keep fire-exposed containers cool. If a spill or leak has not ignited, use water spray to disperse the vapors. Water spray may be used to flush spills away from fire. Unusual Fire or Explosion Hazar ...
Microsoft Word
... Strong bases — include Ba(OH)2 and hydroxides of the alkali metals (NaOH, KOH, etc.), the soluble ionic hydroxides. Other hydroxides are either slightly soluble or insoluble and are weak bases because the OH– ions are mostly tied in the solid. Acid-Base Reactions Reactions of acids ...
... Strong bases — include Ba(OH)2 and hydroxides of the alkali metals (NaOH, KOH, etc.), the soluble ionic hydroxides. Other hydroxides are either slightly soluble or insoluble and are weak bases because the OH– ions are mostly tied in the solid. Acid-Base Reactions Reactions of acids ...
Spring 2014 Chemistry Review
... 101) Adding a solute to a solvent will (increase / decrease) the concentration of the solution. 102) The general rule for solvation is “__________ dissolves __________”. Use the solubility graph to answer questions: 103) How many grams of NaNO3 will dissolve at 30C? _______ 104) Which substance is ...
... 101) Adding a solute to a solvent will (increase / decrease) the concentration of the solution. 102) The general rule for solvation is “__________ dissolves __________”. Use the solubility graph to answer questions: 103) How many grams of NaNO3 will dissolve at 30C? _______ 104) Which substance is ...
Bioenergy basics miller
... (Tradeoff between capital cost (~M0.6) and cost of transporting biomass (~M1.5)). • Process energy provided by lignin combustion • Can choose parameters arbitrarily or use standard values (NREL website). • Possibilities for open-ended design, multiple smaller “feeder” process units in remote locatio ...
... (Tradeoff between capital cost (~M0.6) and cost of transporting biomass (~M1.5)). • Process energy provided by lignin combustion • Can choose parameters arbitrarily or use standard values (NREL website). • Possibilities for open-ended design, multiple smaller “feeder” process units in remote locatio ...
CHEM%1212K% Final%Exam% Summer%2011% K
... B)%The%pH%of%the%solution%is%neutral%because%it%was%made%from%a%strong%base% ...
... B)%The%pH%of%the%solution%is%neutral%because%it%was%made%from%a%strong%base% ...
Chemical Reactions
... Sodium bicarbonate can neutralize or reduce acids in the blood, or urine. It may be used in emergency medical situations (heart attacks, serious kidney or lung problems) to correct the normal acid-base balance in the blood or as an aid in treating overdoses with certain types of medications. ...
... Sodium bicarbonate can neutralize or reduce acids in the blood, or urine. It may be used in emergency medical situations (heart attacks, serious kidney or lung problems) to correct the normal acid-base balance in the blood or as an aid in treating overdoses with certain types of medications. ...