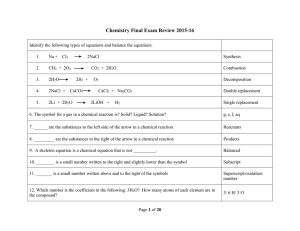
Solution FRQs Practice
... 2003 D Required For each of the following, use appropriate chemical principles to explain the observations. Include chemical equations as appropriate. (a) In areas affected by acid rain, statues and structures made of limestone (calcium carbonate) often show signs of considerable deterioration. (b) ...
... 2003 D Required For each of the following, use appropriate chemical principles to explain the observations. Include chemical equations as appropriate. (a) In areas affected by acid rain, statues and structures made of limestone (calcium carbonate) often show signs of considerable deterioration. (b) ...
Spring 2014
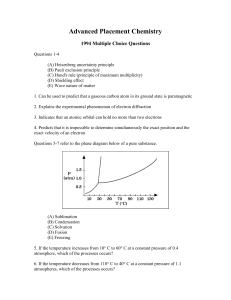
... 13. NO2 has 5 + 2*6 = 17 valence electrons. Which statement about NO2 is correct? A) Since it cannot form octets with an odd number of electrons, the molecule doesn’t exist. B) The molecule exists because nitrogen can form stable 3-electron bonds. C) The molecule does exist, but is pretty reactive b ...
... 13. NO2 has 5 + 2*6 = 17 valence electrons. Which statement about NO2 is correct? A) Since it cannot form octets with an odd number of electrons, the molecule doesn’t exist. B) The molecule exists because nitrogen can form stable 3-electron bonds. C) The molecule does exist, but is pretty reactive b ...
1) abcde 2) abcde 3) abcde 4) abcde 5) abcde 6) abcde 7) abcde 8
... 19) What is the concentration of H3 O+ in a solution whose pH is 4? (a) 104 M (b) 10-4 M (c) 10-10 M (d) 4 M 20) The random motion of molecules helps to explain (a) the compressibility of gases (b) the fact that gases fill their container (c) the low density of gases (d) the transparency of gases 21 ...
... 19) What is the concentration of H3 O+ in a solution whose pH is 4? (a) 104 M (b) 10-4 M (c) 10-10 M (d) 4 M 20) The random motion of molecules helps to explain (a) the compressibility of gases (b) the fact that gases fill their container (c) the low density of gases (d) the transparency of gases 21 ...
Section 4.8: Acid-Base Reactions
... Two compounds react to form two new compounds. All double replacement reactions must have a "driving force" that removes a pair of ions from solution. Ions in a precipitation reaction will keep their same charges as reactants and products. Formation of a precipitate: A precipitate is an insoluble su ...
... Two compounds react to form two new compounds. All double replacement reactions must have a "driving force" that removes a pair of ions from solution. Ions in a precipitation reaction will keep their same charges as reactants and products. Formation of a precipitate: A precipitate is an insoluble su ...
Chemistry II Exams and Answer Keys 2015 Season
... 4. A 1.000 g sample of nickel ore is dissolved in acid. The nickel(II) ions are precipitated by DMGO (dimethyl glyoxime) solution according to the following equation: Ni2+(aq) + 2C4H8N2O2 → Νi(C8H14ON4O4)2(s) + 2H+(aq) The precipitate is filtered and washed. Then, it is heated gently to dryness. The ...
... 4. A 1.000 g sample of nickel ore is dissolved in acid. The nickel(II) ions are precipitated by DMGO (dimethyl glyoxime) solution according to the following equation: Ni2+(aq) + 2C4H8N2O2 → Νi(C8H14ON4O4)2(s) + 2H+(aq) The precipitate is filtered and washed. Then, it is heated gently to dryness. The ...
Researches of the system of neutralization process control in the
... During the rise of temperature T1 in the reaction zone, conditioned by increase of nitric acid consumption Fk , decrease of ammonia gas consumption Fa or increase of nitric acid concentration Qk , thermal EMF E1 and E2 of thermocouples 6 and 7 and their difference E E1 E 2 , which is being carr ...
... During the rise of temperature T1 in the reaction zone, conditioned by increase of nitric acid consumption Fk , decrease of ammonia gas consumption Fa or increase of nitric acid concentration Qk , thermal EMF E1 and E2 of thermocouples 6 and 7 and their difference E E1 E 2 , which is being carr ...
Fundamentals of Theoretical Organic Chemistry Lecture 1
... Some of the mathematical models given in Figure1.1.2-8, together with other formulas not shown on figure, can be combined to compute internal potential energies. The equations may be combined in a number of possible ways. The combined equations, collectively referred to as “force - fields”, incorpor ...
... Some of the mathematical models given in Figure1.1.2-8, together with other formulas not shown on figure, can be combined to compute internal potential energies. The equations may be combined in a number of possible ways. The combined equations, collectively referred to as “force - fields”, incorpor ...
Final review packet
... 7. Using the solubility rules, tell which of these compounds are soluble: (S) = Soluble or (I) = Insoluble in water _____ a. magnesium hydroxide ______g. (NH4)3PO4 _____ b. silver chloride ...
... 7. Using the solubility rules, tell which of these compounds are soluble: (S) = Soluble or (I) = Insoluble in water _____ a. magnesium hydroxide ______g. (NH4)3PO4 _____ b. silver chloride ...
ChemChapter_7sec1_and_section2[1]FORMULA
... not have an exact physical meaning: rather, they serve as useful “bookkeeping” devices to help keep track of electrons. ...
... not have an exact physical meaning: rather, they serve as useful “bookkeeping” devices to help keep track of electrons. ...
Chapter 7 - Angelfire
... Section 1: Names of Binary Compounds • Binary compounds are those formed from only 2 elements. • To write their formulas the positive ion is written first and then the negative. • To name them use the complete name of the positive ion and add the negative ion name but change the ending to “-ide.” ( ...
... Section 1: Names of Binary Compounds • Binary compounds are those formed from only 2 elements. • To write their formulas the positive ion is written first and then the negative. • To name them use the complete name of the positive ion and add the negative ion name but change the ending to “-ide.” ( ...
Type Of Chemical Reaction
... h. What is the independent variable on this graph? Temperature i. What is the dependent variable on this graph? Solubility j. Which substance is the most soluble at all temperatures? KI ...
... h. What is the independent variable on this graph? Temperature i. What is the dependent variable on this graph? Solubility j. Which substance is the most soluble at all temperatures? KI ...
ATOMS, MOLES AND STOICHIOMETRY
... There are several types of titration; we will only focus on ACID-BASE titration’s. Titration’s are used to find the concentration of an acid or alkali (base) An indicator is used to show the ‘end-point’ which is when the acid has reacted with all of the alkali Acid name Formula Hydrochloric acid HCl ...
... There are several types of titration; we will only focus on ACID-BASE titration’s. Titration’s are used to find the concentration of an acid or alkali (base) An indicator is used to show the ‘end-point’ which is when the acid has reacted with all of the alkali Acid name Formula Hydrochloric acid HCl ...
C:\Users\mrh70950\Documents\My Files\WordPerfect
... 1. elimination of 2 HX from geminal, 1,1-dihaloalkanes: double dehydrohalogenation using very strong bases a. E2 twice is usually mechanism b. must use strong base like NaNH2 c. anti-elimination stereochemistry of E2 is followed d. an intermediate haloalkene is generated; this alkene is the major pr ...
... 1. elimination of 2 HX from geminal, 1,1-dihaloalkanes: double dehydrohalogenation using very strong bases a. E2 twice is usually mechanism b. must use strong base like NaNH2 c. anti-elimination stereochemistry of E2 is followed d. an intermediate haloalkene is generated; this alkene is the major pr ...
Acids, Bases and Salts
... However, it was not until a few hundred years ago that it was discovered why these things taste ...
... However, it was not until a few hundred years ago that it was discovered why these things taste ...
Review
... species in all phases must be equal to each other Processes move from a higher chemical potential to a lower one. Expression for the molar Gibbs free energy, the chemical potential, of a gas Calculation of the Equilibrium Constant from Gorxn or the reverse of this. Calculating the Temp dependence ...
... species in all phases must be equal to each other Processes move from a higher chemical potential to a lower one. Expression for the molar Gibbs free energy, the chemical potential, of a gas Calculation of the Equilibrium Constant from Gorxn or the reverse of this. Calculating the Temp dependence ...
Biochemistry I (CHE 418 / 5418)
... If 2.10 mol N2 and 5.70 mol H2 react, what is the limiting reagent. Ask the question, how many mol of H2 is needed to react with .10 ...
... If 2.10 mol N2 and 5.70 mol H2 react, what is the limiting reagent. Ask the question, how many mol of H2 is needed to react with .10 ...
AP Semestar Exam REVIEW
... b. Li+ + I- KCl(s) c. LiOH(aq) + HI(aq) LiI(s) + H2O d. LiOH(s) + H+(aq) Li+(aq) + H2O e. OH-(aq) + H+(aq) H2O ____ 24. When HCl(g) and NH3(g) are mixed, a white solid forms. What is the balanced equation for this reaction? a. HCl(g) + NH3(g) NH4Cl(s) b. HCl(g) + NH3(g) NH2Cl(g) + H2(s) ...
... b. Li+ + I- KCl(s) c. LiOH(aq) + HI(aq) LiI(s) + H2O d. LiOH(s) + H+(aq) Li+(aq) + H2O e. OH-(aq) + H+(aq) H2O ____ 24. When HCl(g) and NH3(g) are mixed, a white solid forms. What is the balanced equation for this reaction? a. HCl(g) + NH3(g) NH4Cl(s) b. HCl(g) + NH3(g) NH2Cl(g) + H2(s) ...
1 ChE 505 WORKSHOP 1 1. Why are chemical reactions important
... What is the relationship between the initial moles of reactants and products, the moles for each of the above after some reaction time, the stoichiometric coefficients and reaction extent? ...
... What is the relationship between the initial moles of reactants and products, the moles for each of the above after some reaction time, the stoichiometric coefficients and reaction extent? ...









![ChemChapter_7sec1_and_section2[1]FORMULA](http://s1.studyres.com/store/data/000546743_1-278f96ccbbfd49e292510ec017e27124-300x300.png)