File
... Alex’s hypothesis was that the rate will be affected by changing the concentrations of the propanone and the iodine, as the reaction can happen without a catalyst. Hannah’s hypothesis was that as the catalyst is involved in the reaction, the concentrations of the propanone, iodine and the hydrogen i ...
... Alex’s hypothesis was that the rate will be affected by changing the concentrations of the propanone and the iodine, as the reaction can happen without a catalyst. Hannah’s hypothesis was that as the catalyst is involved in the reaction, the concentrations of the propanone, iodine and the hydrogen i ...
AP Chemistry Review Assignment Brown and LeMay: Chemistry the
... 11. Classify each of the following as a pure substance or a mixture; if a mixture, indicate whether it is homogeneous or heterogeneous: a) rice pudding b) seawater c) magnesium d) gasoline 15. A solid white substance A is heated strongly in the absence of air. It decomposes to form a new white subst ...
... 11. Classify each of the following as a pure substance or a mixture; if a mixture, indicate whether it is homogeneous or heterogeneous: a) rice pudding b) seawater c) magnesium d) gasoline 15. A solid white substance A is heated strongly in the absence of air. It decomposes to form a new white subst ...
Grade XII Foreign SET 2 Chemistry (Theory)
... (i) Nitrogen is chemically less reactive. This is because of the high stability of its molecule, N2. In N2, the two nitrogen atoms form a triple bond. This triple bond has very high bond strength, which is very difficult to break. It is because of nitrogen’s small size that it is able to form p– p ...
... (i) Nitrogen is chemically less reactive. This is because of the high stability of its molecule, N2. In N2, the two nitrogen atoms form a triple bond. This triple bond has very high bond strength, which is very difficult to break. It is because of nitrogen’s small size that it is able to form p– p ...
Group 2 - UC Davis Canvas
... 11. The bond energy of the noble gas fluorine is too small to offset the energy required to break the F—F bond. 13. Iodide ion is slowly oxidized to iodine, which is yellow-brown in aqueous solution, by oxygen in the air: 4 I − ( aq ) + O 2 ( g ) + 4 H + ( aq ) → 2 I 2 ( aq ) + 2 H 2 O(l) . 15. D ...
... 11. The bond energy of the noble gas fluorine is too small to offset the energy required to break the F—F bond. 13. Iodide ion is slowly oxidized to iodine, which is yellow-brown in aqueous solution, by oxygen in the air: 4 I − ( aq ) + O 2 ( g ) + 4 H + ( aq ) → 2 I 2 ( aq ) + 2 H 2 O(l) . 15. D ...
Acidity Test Kit Alkalinity Test Kit 9
... ·· Readings from 0 to 100 mg/L are determined to 1 mg/L resolution. ·· Readings from 0 to 500 mg/L are determined to 5 mg/L resolution. • Replacement reagents available ·· There is no need to buy a new kit when reagents are exhausted. The HI3820-100 can be ordered to replace the reagents supplied w ...
... ·· Readings from 0 to 100 mg/L are determined to 1 mg/L resolution. ·· Readings from 0 to 500 mg/L are determined to 5 mg/L resolution. • Replacement reagents available ·· There is no need to buy a new kit when reagents are exhausted. The HI3820-100 can be ordered to replace the reagents supplied w ...
What is matter?
... Transparency – the degree to which a substance freely permits the passage of light transparent butterfly ...
... Transparency – the degree to which a substance freely permits the passage of light transparent butterfly ...
AQA Additional Sci C2 Revision Guide
... form positively charged ions. Non-metal atoms gain electrons to form negatively charged ions. Ions have the electronic structure of a noble gas i.e. they have full outer shells. Oppositely charged ions are strongly attracted to each other and are held together by ionic bonds. The diagram below shows ...
... form positively charged ions. Non-metal atoms gain electrons to form negatively charged ions. Ions have the electronic structure of a noble gas i.e. they have full outer shells. Oppositely charged ions are strongly attracted to each other and are held together by ionic bonds. The diagram below shows ...
Document
... concentrations of excess hydrochloric acid and the time taken for the magnesium to completely react, recorded. A graph of the student's results is shown below. ...
... concentrations of excess hydrochloric acid and the time taken for the magnesium to completely react, recorded. A graph of the student's results is shown below. ...
Advanced Placement Chemistry
... D) It is the first order in [Y]. E) The overall order of the reaction is 2. 64. Equal numbers of moles of He(g), Ar(g), and Ne(g) are placed in a glass vessel at room temperature. If the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial ...
... D) It is the first order in [Y]. E) The overall order of the reaction is 2. 64. Equal numbers of moles of He(g), Ar(g), and Ne(g) are placed in a glass vessel at room temperature. If the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial ...
Chemistry Review Fill in the blank
... 4. Ionic compounds involve ____________ and _______________. They are characterized by the __________________ of electrons. 5. Covalent molecules involve _____________________. They are characterized by the ______________ of electrons. 6. Type of bonding and electronegativity differences. Type Elect ...
... 4. Ionic compounds involve ____________ and _______________. They are characterized by the __________________ of electrons. 5. Covalent molecules involve _____________________. They are characterized by the ______________ of electrons. 6. Type of bonding and electronegativity differences. Type Elect ...
Acids and Bases
... shared with a proton. In other words, stable bases are weak bases—they don’t share their electrons well. So we can say, the stronger the acid, the weaker is its conjugate base or, the stronger the acid, the more stable is its conjugate base. The stability of an acid’s conjugate base is determined by ...
... shared with a proton. In other words, stable bases are weak bases—they don’t share their electrons well. So we can say, the stronger the acid, the weaker is its conjugate base or, the stronger the acid, the more stable is its conjugate base. The stability of an acid’s conjugate base is determined by ...
Energy and Matter in Chemical Change Science 10
... 2. Reactivity: Metals are very reactive, some more than others, but most form compounds with other elements quite easily. Sodium (Na) and potassium (K) are some of the most reactive metals. 3. Chemical: Metals usually make positive ions when the compounds are dissolved in solution. Also, their metal ...
... 2. Reactivity: Metals are very reactive, some more than others, but most form compounds with other elements quite easily. Sodium (Na) and potassium (K) are some of the most reactive metals. 3. Chemical: Metals usually make positive ions when the compounds are dissolved in solution. Also, their metal ...
Final Exam - Seattle Central College
... – Recognize how IMF’s influence vapor pressure and boiling point. – Given different substances, determine which has the highest boiling point based on IMF’s. • Given a bond or intermolecular force, identify it a polar covalent, nonpolar covalent, ionic, metallic, ion-dipole forces, London/dispersion ...
... – Recognize how IMF’s influence vapor pressure and boiling point. – Given different substances, determine which has the highest boiling point based on IMF’s. • Given a bond or intermolecular force, identify it a polar covalent, nonpolar covalent, ionic, metallic, ion-dipole forces, London/dispersion ...
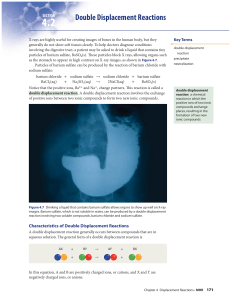
Double Displacement Reactions
... double displacement reaction produces a gas. Give the general form of each reaction. ...
... double displacement reaction produces a gas. Give the general form of each reaction. ...
9. The Copigmentation Interactions between Strawberry
... anthocyanins was examined in model solutions, with high concentrations caffeic acid as a copigment at different temperatures at heating and at cooling. In the temperature range 20-50oC at heating was confirm that the system was the most stable at 20oC. With increasing temperature to 50 o C and follo ...
... anthocyanins was examined in model solutions, with high concentrations caffeic acid as a copigment at different temperatures at heating and at cooling. In the temperature range 20-50oC at heating was confirm that the system was the most stable at 20oC. With increasing temperature to 50 o C and follo ...
ACP Chemistry Semester 1 Final Exam - Doc-U-Ment
... 20) A mixture of 0.220 moles CO, 0.350 moles H2 and 0.640 moles He has a total pressure of 2.95 atm. What is the pressure of CO? A) 1.86 atm B) 0.649 atm C) 0.536 atm D) 1.54 atm E) 0.955 atm 21) Determine the volume of SO2 (at STP) formed from the reaction of 96.7 g of FeS2 and 55.0 L of O2 (at 398 ...
... 20) A mixture of 0.220 moles CO, 0.350 moles H2 and 0.640 moles He has a total pressure of 2.95 atm. What is the pressure of CO? A) 1.86 atm B) 0.649 atm C) 0.536 atm D) 1.54 atm E) 0.955 atm 21) Determine the volume of SO2 (at STP) formed from the reaction of 96.7 g of FeS2 and 55.0 L of O2 (at 398 ...
Formulae and equations
... The number of atoms or groups of atoms in a formula is given by putting a small number just below and behind the symbol(s). As the appearance of a symbol indicates one atom is present, a 1 isn’t written (you put NaCl not Na1Cl1). In some formulae brackets are used to avoid ambiguity. Aluminium sulph ...
... The number of atoms or groups of atoms in a formula is given by putting a small number just below and behind the symbol(s). As the appearance of a symbol indicates one atom is present, a 1 isn’t written (you put NaCl not Na1Cl1). In some formulae brackets are used to avoid ambiguity. Aluminium sulph ...
CHM1 Review for Exam 9 Topics 1. Reaction Types a. Combustion
... 2Halides (Cl , Br and Ag , Pb or Hg2 Sulfide (S ) Group 1 ions and I) ammonium ion Sulfates (SO42-) Ag+, Sr2+, Ba2+, Pb2+ Hydroxides (OH-) Group 1 ions and 2and oxides (O ) ammonium ion ...
... 2Halides (Cl , Br and Ag , Pb or Hg2 Sulfide (S ) Group 1 ions and I) ammonium ion Sulfates (SO42-) Ag+, Sr2+, Ba2+, Pb2+ Hydroxides (OH-) Group 1 ions and 2and oxides (O ) ammonium ion ...
Honors Unit 3 - Stoichiometry
... 1. Zinc metal and aqueous lead (II) nitrate react to form aqueous zinc nitrate and solid lead. ...
... 1. Zinc metal and aqueous lead (II) nitrate react to form aqueous zinc nitrate and solid lead. ...