Chemistry
... To be able to: Identify the position of the metallic elements in the periodic system, to characterize the metallic bond, metal lattices, physical properties of metals. Distinguish between metallic and non-metallic elements according to the atomic structure. Write electronic formula of the atoms of ...
... To be able to: Identify the position of the metallic elements in the periodic system, to characterize the metallic bond, metal lattices, physical properties of metals. Distinguish between metallic and non-metallic elements according to the atomic structure. Write electronic formula of the atoms of ...
practice exercise - Needham.K12.ma.us
... noble-gas atom. From the periodic table, we see that barium has atomic number 56. The nearest noble gas is xenon, atomic number 54. Barium can attain a stable arrangement of 54 electrons by losing two of its electrons, forming the Ba2 + cation. Oxygen has atomic number 8. The nearest noble gas is ne ...
... noble-gas atom. From the periodic table, we see that barium has atomic number 56. The nearest noble gas is xenon, atomic number 54. Barium can attain a stable arrangement of 54 electrons by losing two of its electrons, forming the Ba2 + cation. Oxygen has atomic number 8. The nearest noble gas is ne ...
Practical and Efficient Synthesis of -Aminophosphonic Acids
... rigid unusual secondary α-amino acids, where the nitrogen is involved in a ring, may result in significant consequences for the conformation of peptidomimetics as synthetic tools for drug discovery [1,2]. Some of the most important molecules are the 1,2,3,4-tetrahydroquinoline-2-carboxylic acid 1 [3 ...
... rigid unusual secondary α-amino acids, where the nitrogen is involved in a ring, may result in significant consequences for the conformation of peptidomimetics as synthetic tools for drug discovery [1,2]. Some of the most important molecules are the 1,2,3,4-tetrahydroquinoline-2-carboxylic acid 1 [3 ...
8B. Acid-base titration of Ibuprofen in tablets
... content of the active component C12H17COOH. Tablets usually contain different neutral components, like starch, which however do not obscure the endpoint. Ibuprofen has one ionizable hydrogen (one-carboxylic acid) and the dissociation constant is pKa1=4.40 [1]. This makes its acid-base titration rela ...
... content of the active component C12H17COOH. Tablets usually contain different neutral components, like starch, which however do not obscure the endpoint. Ibuprofen has one ionizable hydrogen (one-carboxylic acid) and the dissociation constant is pKa1=4.40 [1]. This makes its acid-base titration rela ...
NATURAL RUBBER
... •Ammonia aqueous contains hydroxide ion, OH•Hydroxide ion can neutralize the acid produced by the bacteria • Hence, the rubber particles remain negatively charged and the coagulation is prevented ...
... •Ammonia aqueous contains hydroxide ion, OH•Hydroxide ion can neutralize the acid produced by the bacteria • Hence, the rubber particles remain negatively charged and the coagulation is prevented ...
ic199p5a
... (b) Suppose that one of these salts actually had the cesium chloride structure (C.N. = 8) and another, the zinc blende structure (C.N. = 4). Choose the most likely salts (2) from this list to have these two other structures and briefly explain the reason for your choice (assume that the anion is big ...
... (b) Suppose that one of these salts actually had the cesium chloride structure (C.N. = 8) and another, the zinc blende structure (C.N. = 4). Choose the most likely salts (2) from this list to have these two other structures and briefly explain the reason for your choice (assume that the anion is big ...
Types of Reactions Lab
... 1.) List the 5 types of reactions. For each type, describe them in terms of elements and compounds and describe what generally happens. 2.) A) What is bromothymol blue and how can it be used to identify an acid or a base (use your book or the internet to look this up)? B) Look up the same for univer ...
... 1.) List the 5 types of reactions. For each type, describe them in terms of elements and compounds and describe what generally happens. 2.) A) What is bromothymol blue and how can it be used to identify an acid or a base (use your book or the internet to look this up)? B) Look up the same for univer ...
chem 111 practice exam
... THERE ARE 6 PAGES TO THIS EXAM (including the cover page) Significant Figures must be correct. All set-ups must be shown (where applicable) or you will receive no credit. (10 points) 1. Myoglobin stores oxygen for metabolic processes in muscle. Chemical analysis shows that it contains 0.34 % Fe by m ...
... THERE ARE 6 PAGES TO THIS EXAM (including the cover page) Significant Figures must be correct. All set-ups must be shown (where applicable) or you will receive no credit. (10 points) 1. Myoglobin stores oxygen for metabolic processes in muscle. Chemical analysis shows that it contains 0.34 % Fe by m ...
14 Enthalpy of neutralization
... In this lab a similar experiment will be performed to find the enthalpy change (H) that occurs during a neutralization reaction between sodium hydroxide and one of three acids (HA) that will be assigned to you. HnA + nNaOH NanA + nH2O The calorimeter will be a “coffee cup” calorimeter, using two ...
... In this lab a similar experiment will be performed to find the enthalpy change (H) that occurs during a neutralization reaction between sodium hydroxide and one of three acids (HA) that will be assigned to you. HnA + nNaOH NanA + nH2O The calorimeter will be a “coffee cup” calorimeter, using two ...
Bellin College Homework Supplement
... D) Sodium Chloride Solution (NaCl) reacts with Silver Nitrate solution (AgNO3) producing solid Silver Chloride (AgCl) and Sodium Nitrate solution (NaNO3). ...
... D) Sodium Chloride Solution (NaCl) reacts with Silver Nitrate solution (AgNO3) producing solid Silver Chloride (AgCl) and Sodium Nitrate solution (NaNO3). ...
Werner-type chromium compounds
... It will be understood, of course, that the acido acido groups, so that for each functional acido group there may be several nuclear metal atoms 60 group is not present alone in any case but that the manner in which it is associated in the solu ‘within the complex, it being necessary only that tion i ...
... It will be understood, of course, that the acido acido groups, so that for each functional acido group there may be several nuclear metal atoms 60 group is not present alone in any case but that the manner in which it is associated in the solu ‘within the complex, it being necessary only that tion i ...
Trends in Physical Properties
... Medicines for the treatment of nervous disorders often contain calcium bromide. Silver nitrate, acidified with dilute nitric acid, can be used together with another reagent to test for the presence of bromide ions in a solution of a medicine. Describe briefly how you would carry out this test and st ...
... Medicines for the treatment of nervous disorders often contain calcium bromide. Silver nitrate, acidified with dilute nitric acid, can be used together with another reagent to test for the presence of bromide ions in a solution of a medicine. Describe briefly how you would carry out this test and st ...
PDF document
... The aim of this work is to develop a new kinetic spectrophotometric method for the determination of acetylsalicylic acid (ASA) in pharmaceutical formulations. In general, ASA analysis is not realised directly, and a previous quantitative hydrolysis in a basic medium is necessary, converting ASA to s ...
... The aim of this work is to develop a new kinetic spectrophotometric method for the determination of acetylsalicylic acid (ASA) in pharmaceutical formulations. In general, ASA analysis is not realised directly, and a previous quantitative hydrolysis in a basic medium is necessary, converting ASA to s ...
- Catalyst
... – a solid precipitate forms when aqueous solutions of certain ions are mixed • Acid-Base: proton transfer reactions – acid donates a proton to a base, forming a molecule (water or another weak acid) and an aqueous salt – Acid: proton-donor; Base: proton-acceptor • Oxidation-Reduction: electron trans ...
... – a solid precipitate forms when aqueous solutions of certain ions are mixed • Acid-Base: proton transfer reactions – acid donates a proton to a base, forming a molecule (water or another weak acid) and an aqueous salt – Acid: proton-donor; Base: proton-acceptor • Oxidation-Reduction: electron trans ...
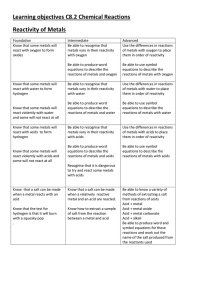
Learning objectives C8.2 Chemical Reactions Reactivity of Metals
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
CHM1 Exam 16 Name 2222222222222222222222222222 Multiple
... What is the total number of moles of NaNO3 formed when 2 moles of Na2CrO4 react completely with excess lead (II) nitrate? (1) 1 mole (2) 2 moles ...
... What is the total number of moles of NaNO3 formed when 2 moles of Na2CrO4 react completely with excess lead (II) nitrate? (1) 1 mole (2) 2 moles ...
SAT - mvhs-fuhsd.org
... straight line until they hit something, they bounce off without losing any energy, they are so far apart from each other that they have effectively no attractive forces and their speed is directly proportional to the Kelvin temperature (KineticMolecular Theory, Ideal Gas Theory) ...
... straight line until they hit something, they bounce off without losing any energy, they are so far apart from each other that they have effectively no attractive forces and their speed is directly proportional to the Kelvin temperature (KineticMolecular Theory, Ideal Gas Theory) ...
How to Make a Collage
... and division should be second nature when you walk into the room and you should be able to do these in your head. Practice these skills using simple flash cards. Build the numbers as you go. Do not just stick to the basics. Expand your horizons and work with larger numbers. You also need to be compe ...
... and division should be second nature when you walk into the room and you should be able to do these in your head. Practice these skills using simple flash cards. Build the numbers as you go. Do not just stick to the basics. Expand your horizons and work with larger numbers. You also need to be compe ...
chemical reaction
... (aq) – aqueous (dissolved in water, exists as ions) ↓ - a precipitate has formed ...
... (aq) – aqueous (dissolved in water, exists as ions) ↓ - a precipitate has formed ...