5 - BrainMass
... 5.72) Using the values from Thermodynamics Quantities for Selected Substances at 298.15 K (25°C), calculate the value of ΔH° for each of the following reactions: a. N2O4 (g) + 4 H2 (g) N2 (g) + 4 H2O (g) b. 2 KOH(s) + CO2 (g) K2CO3(s) + H2O (g) c. SO2 (g) + 2 H2S (g) 3/8 S8(s) + 2 H2O (g) d. F ...
... 5.72) Using the values from Thermodynamics Quantities for Selected Substances at 298.15 K (25°C), calculate the value of ΔH° for each of the following reactions: a. N2O4 (g) + 4 H2 (g) N2 (g) + 4 H2O (g) b. 2 KOH(s) + CO2 (g) K2CO3(s) + H2O (g) c. SO2 (g) + 2 H2S (g) 3/8 S8(s) + 2 H2O (g) d. F ...
Ch. 3 - Chemical Reactions
... Describing Equations Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) • How many? • Of what? • In what state? ...
... Describing Equations Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) • How many? • Of what? • In what state? ...
CHEM102 Chemistry II Spring 11-12 Mid
... 28) Which of the following can serve as the solvent in a solution? 28) ______ A) a liquid B) a gas C) a solid D) a mixture of comingled liquids E) all of the above 29) If the concentration of H3O+ is 3.5 × 10-3 M, the concentration of OH- is ________ M. 29) ______ A) 3.5 × 10-11 B) 1.0 × 10-12 C) 2. ...
... 28) Which of the following can serve as the solvent in a solution? 28) ______ A) a liquid B) a gas C) a solid D) a mixture of comingled liquids E) all of the above 29) If the concentration of H3O+ is 3.5 × 10-3 M, the concentration of OH- is ________ M. 29) ______ A) 3.5 × 10-11 B) 1.0 × 10-12 C) 2. ...
Chapter 6A Chemical Reactions CHAPTER OUTLINE
... the transfer of hydrogen atoms produces energy in the cells. q For example, cellular respiration is an oxidationreduction process that transfers energy from the bonds in glucose to form ATP. C6H12O6 + 6 O2 ...
... the transfer of hydrogen atoms produces energy in the cells. q For example, cellular respiration is an oxidationreduction process that transfers energy from the bonds in glucose to form ATP. C6H12O6 + 6 O2 ...
chapter 4 review_package
... i. How many grams of aluminum oxide, Al2O3, would be expected to form in the reaction of 15.0g Al with 18.43g of oxygen gas? ...
... i. How many grams of aluminum oxide, Al2O3, would be expected to form in the reaction of 15.0g Al with 18.43g of oxygen gas? ...
FINAL EXAM Spring 2012
... 2C2H2 (g) + 5O2 (g) → 4CO2 (g) + 2H2O (l) The value of S° for this reaction is __________ J/K∙ mol. A) -432.4 B) -122.3 C) +689.3 D) +432.4 E) +122.3 28) For the reaction O2(g) 2O(g) , what conditions favor production of oxygen atoms? A) high temperature and low pressure B) low temperature and l ...
... 2C2H2 (g) + 5O2 (g) → 4CO2 (g) + 2H2O (l) The value of S° for this reaction is __________ J/K∙ mol. A) -432.4 B) -122.3 C) +689.3 D) +432.4 E) +122.3 28) For the reaction O2(g) 2O(g) , what conditions favor production of oxygen atoms? A) high temperature and low pressure B) low temperature and l ...
Chem Reactions (and Balancing Equations)
... Symbols used in equations (s) after the formula –solid Cu(s) (g) after the formula –gas H2 (g) (l) after the formula -liquid H2O(l) (aq) after the formula - dissolved in water, an aqueous solution. CaCl2 (aq) • used after a product indicates a gas (same as (g)) O2 • used after a product indic ...
... Symbols used in equations (s) after the formula –solid Cu(s) (g) after the formula –gas H2 (g) (l) after the formula -liquid H2O(l) (aq) after the formula - dissolved in water, an aqueous solution. CaCl2 (aq) • used after a product indicates a gas (same as (g)) O2 • used after a product indic ...
2014
... PCl5 (g) ⇆ PCl3 (g) + Cl2 (g) PCl5 (g) decomposes into PCl3 (g) and Cl2 (g) according to the equation above. A sample of PCl5 (g) is placed into a rigid, evacuated 1.00 L container. The initial pressure of the PCl5 (g) is 1.00 atm. The temperature is held constant until the PCl5 (g) reaches equilibr ...
... PCl5 (g) ⇆ PCl3 (g) + Cl2 (g) PCl5 (g) decomposes into PCl3 (g) and Cl2 (g) according to the equation above. A sample of PCl5 (g) is placed into a rigid, evacuated 1.00 L container. The initial pressure of the PCl5 (g) is 1.00 atm. The temperature is held constant until the PCl5 (g) reaches equilibr ...
Project Advance Chemistry 106 Sample Questions
... respectively. Determine the value of the Eocell for a voltaic cell in which the overall reaction is ...
... respectively. Determine the value of the Eocell for a voltaic cell in which the overall reaction is ...
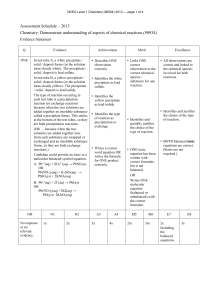
82KB - NZQA
... decomposing to form more than one substance when heated. Both are white solids and when strongly heated, they both form another white solid, calcium oxide. During heating, both of the solids break up because gas escapes from them. Contrast: The difference in these reactions is in the gases released. ...
... decomposing to form more than one substance when heated. Both are white solids and when strongly heated, they both form another white solid, calcium oxide. During heating, both of the solids break up because gas escapes from them. Contrast: The difference in these reactions is in the gases released. ...
CHM1 Review for Exam 9 Topics 1. Reaction Types a. Combustion
... (4) CH4 + 2 O2 CO2 + 2 H2O 8. One hundred grams of water is saturated with NH4Cl at 50°C. According to Table G, if the temperature is lowered to 10°C. what is the total amount of NH4Cl that will precipitate? (1) 5.0 g (2) 17 g ...
... (4) CH4 + 2 O2 CO2 + 2 H2O 8. One hundred grams of water is saturated with NH4Cl at 50°C. According to Table G, if the temperature is lowered to 10°C. what is the total amount of NH4Cl that will precipitate? (1) 5.0 g (2) 17 g ...
An Efficient Synthetic Route to Glycoamino Acid Building Blocks for
... [email protected] Received August 20, 2004 ...
... [email protected] Received August 20, 2004 ...
Rates of Reaction: Chemical Kinetics 50
... Dr. Goodall. The note is typed and faxed to Africa. The fax is placed in an envelope when it is received and given to a messenger who must travel a few kilometres by boat and a few hundred metres on foot before handing the message to Dr. Goodall. The messenger returns to the messenger office with th ...
... Dr. Goodall. The note is typed and faxed to Africa. The fax is placed in an envelope when it is received and given to a messenger who must travel a few kilometres by boat and a few hundred metres on foot before handing the message to Dr. Goodall. The messenger returns to the messenger office with th ...
1) In the reaction H2O + CH3COOH H3O+ + CH3COO
... zero, equal to zero or impossible to determine? ...
... zero, equal to zero or impossible to determine? ...
AP Chemistry Syllabus 2013 Mawhiney
... lectures but are also chosen to reflect the diversity of lab work generally completed in a first year course. Analysis of data from AP Chemistry examinees shows that increased laboratory time is correlated with higher AP grades. Depending on the particular lab, students will work individually or col ...
... lectures but are also chosen to reflect the diversity of lab work generally completed in a first year course. Analysis of data from AP Chemistry examinees shows that increased laboratory time is correlated with higher AP grades. Depending on the particular lab, students will work individually or col ...