Bonding in Atoms
... • States that an atom will lose or gain electrons in order to fill the outer sublevels (s and p) • Modeled by the Lewis Dot Diagram • Gain of electrons = anion • Loss of electrons = cations ...
... • States that an atom will lose or gain electrons in order to fill the outer sublevels (s and p) • Modeled by the Lewis Dot Diagram • Gain of electrons = anion • Loss of electrons = cations ...
The Chemical Basis of Life
... Chemical reactions involve making and breaking bonds. Making bonds requires the input of energy. Energy is stored in molecules in the chemical bonds. Breaking bonds usually releases energy. ...
... Chemical reactions involve making and breaking bonds. Making bonds requires the input of energy. Energy is stored in molecules in the chemical bonds. Breaking bonds usually releases energy. ...
Valence electrons and Lewis Dot Structures
... 1. Write the symbols down, metal first. 2. Determine the charge of each ion, either from its position on the PT or a chart like 7-8 on p. ...
... 1. Write the symbols down, metal first. 2. Determine the charge of each ion, either from its position on the PT or a chart like 7-8 on p. ...
Biochemistry I (CHE 418 / 5418)
... Covalent Bonds (Cont) • Some elements form double or triple covalent bonds by sharing two pairs or three pairs of electrons. – Draw Lewis dot structure and line structure for molecular oxygen (O2), which forms double bonds. – Draw Lewis dot structure and line structure for acetylene (C2H2), which c ...
... Covalent Bonds (Cont) • Some elements form double or triple covalent bonds by sharing two pairs or three pairs of electrons. – Draw Lewis dot structure and line structure for molecular oxygen (O2), which forms double bonds. – Draw Lewis dot structure and line structure for acetylene (C2H2), which c ...
Regents Review Packet B2 Answer Key
... The Bohr model of the atom was developed in the early part of the twentieth century. A diagram of the Bohr model for one atom, in the ground state, of a specific element, is shown below. The nucleus of this atom contains 4 protons and 5 neutrons. ...
... The Bohr model of the atom was developed in the early part of the twentieth century. A diagram of the Bohr model for one atom, in the ground state, of a specific element, is shown below. The nucleus of this atom contains 4 protons and 5 neutrons. ...
unit 2 - chemistry
... (valence) and bonding is the result 3.bonding – sharing, or giving/ receiving of valence electrons – chemical reaction B. Molecule – 2 or more atoms chemically combined C. Compound – combination of 2 or more elements D. Bonding 1. ionic – giving or receiving of e- (bond) transfer e- results in a c ...
... (valence) and bonding is the result 3.bonding – sharing, or giving/ receiving of valence electrons – chemical reaction B. Molecule – 2 or more atoms chemically combined C. Compound – combination of 2 or more elements D. Bonding 1. ionic – giving or receiving of e- (bond) transfer e- results in a c ...
1 - M*W
... b) Differ in electronegativity d) Have the same number of electrons 23) To draw a Lewis structure you do not need to know a) The number of valence electrons for each atom b) The types of atoms in the molecule c) The number of atoms in the molecule d) Bond energies 24) Neils Bohr’s contribution to mo ...
... b) Differ in electronegativity d) Have the same number of electrons 23) To draw a Lewis structure you do not need to know a) The number of valence electrons for each atom b) The types of atoms in the molecule c) The number of atoms in the molecule d) Bond energies 24) Neils Bohr’s contribution to mo ...
Medical Physics and Statistics
... The first prize of the Idaho Falls High School Science Fair was awarded on April 26 to a student of Eagle Rock High School. The student wanted to demonstrate the extent to which the public is manipulated by vague references to science in generating environmental concern. He prepared a proposal for ba ...
... The first prize of the Idaho Falls High School Science Fair was awarded on April 26 to a student of Eagle Rock High School. The student wanted to demonstrate the extent to which the public is manipulated by vague references to science in generating environmental concern. He prepared a proposal for ba ...
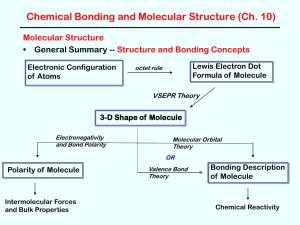
Chemical Bonding and Molecular Structure
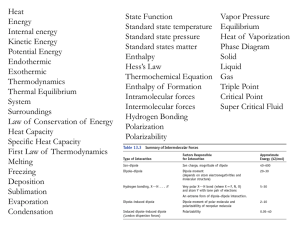
... molecule is composed of atoms that are bound together by sharing pairs of electrons using the atomic orbitals of the bound atoms. Electron pairs are assumed to be localized on a particular atom [lone pairs] or in the space between two atoms [bonding pairs]. Lewis Structures describe the valence elec ...
... molecule is composed of atoms that are bound together by sharing pairs of electrons using the atomic orbitals of the bound atoms. Electron pairs are assumed to be localized on a particular atom [lone pairs] or in the space between two atoms [bonding pairs]. Lewis Structures describe the valence elec ...
Bonding
... Answer the following questions using principles of chemical bonding and molecular structure. b.Consider the molecules CF4 and SF4. i. Draw the complete Lewis electron-dot structures for each molecule. ii.In terms of molecular geometry, account for the fact that the CF4 molecule is nonpolar, whereas ...
... Answer the following questions using principles of chemical bonding and molecular structure. b.Consider the molecules CF4 and SF4. i. Draw the complete Lewis electron-dot structures for each molecule. ii.In terms of molecular geometry, account for the fact that the CF4 molecule is nonpolar, whereas ...
Chem 101 notes review
... 1. list all the elements follow with an equal sign 2. follow with the number of atoms of that type in the molecule 1. follow with a multiplication sign 2. If the element is O follow with a -2 3. If the element is H follow with a +1 4. any other element enter a ? 5. follow with an = sign, do the math ...
... 1. list all the elements follow with an equal sign 2. follow with the number of atoms of that type in the molecule 1. follow with a multiplication sign 2. If the element is O follow with a -2 3. If the element is H follow with a +1 4. any other element enter a ? 5. follow with an = sign, do the math ...
Chapter1011
... • Polar or Non-Polar? – In very symmetrical structures (e.g. CO2 or CF4), the individual bond dipoles effectively cancel each other and the molecule is ...
... • Polar or Non-Polar? – In very symmetrical structures (e.g. CO2 or CF4), the individual bond dipoles effectively cancel each other and the molecule is ...
AP Chemistry 2013 Semester 1 Final Exam Review Problems
... b. What is the molecular formula of this substance? c. Draw the Lewis structure of the molecule using the fact that the Cl atoms bond to a single C atom, there is a C-C bond, and two C-O bonds in the compound. 17. Draw the Lewis structures for BH3 and NH3. a. What is the bond angle around the centra ...
... b. What is the molecular formula of this substance? c. Draw the Lewis structure of the molecule using the fact that the Cl atoms bond to a single C atom, there is a C-C bond, and two C-O bonds in the compound. 17. Draw the Lewis structures for BH3 and NH3. a. What is the bond angle around the centra ...
Note 1.1 Chemistry of Life
... the chemical and physical properties of the element. An atom is made up of three different sub atomic particles; neutrons (no charge), protons (positive charge), and electrons (negative charge). Atomic number is the number of protons found in the nucleus of the atom. It determines the particular ato ...
... the chemical and physical properties of the element. An atom is made up of three different sub atomic particles; neutrons (no charge), protons (positive charge), and electrons (negative charge). Atomic number is the number of protons found in the nucleus of the atom. It determines the particular ato ...
PPT - gserianne.com
... • atoms with the same atomic numbers but with different atomic weights • atoms with the same number of protons and electrons but a different number of neutrons • oxygen (atomic number 8) has the following isotopes (16O, 17O, 18O) • unstable isotopes (radioisotopes or radionuclides) are radioactive; ...
... • atoms with the same atomic numbers but with different atomic weights • atoms with the same number of protons and electrons but a different number of neutrons • oxygen (atomic number 8) has the following isotopes (16O, 17O, 18O) • unstable isotopes (radioisotopes or radionuclides) are radioactive; ...
Chemical Bonds
... electrons) attract each other. These forces pull atoms together to form compounds An atom is chemically stable when it has a complete outer energy level ...
... electrons) attract each other. These forces pull atoms together to form compounds An atom is chemically stable when it has a complete outer energy level ...
Chemical Bonding
... • The subatomic particles that make up atoms are protons, neutrons, and electrons. • Protons=Positive charge • Neutrons=Neutral charge • Electrons=Negative charge ...
... • The subatomic particles that make up atoms are protons, neutrons, and electrons. • Protons=Positive charge • Neutrons=Neutral charge • Electrons=Negative charge ...
Chemistry of Life
... • Large molecule built by amino acids • Amino Acids- There are _________ different proteins • Proteins have many different functions such as enzymes, structure, antibodies, hemoglobin(blood flow) ...
... • Large molecule built by amino acids • Amino Acids- There are _________ different proteins • Proteins have many different functions such as enzymes, structure, antibodies, hemoglobin(blood flow) ...
Honors Midterm Review – 2015-16
... _________ responsible for the uncertainty principle which states that it is impossible to know (with any great degree of certainty) both the location and velocity of an electron) _________ responsible for the planetary model of the atom, where electrons traveled in distinct paths around the nucleus ...
... _________ responsible for the uncertainty principle which states that it is impossible to know (with any great degree of certainty) both the location and velocity of an electron) _________ responsible for the planetary model of the atom, where electrons traveled in distinct paths around the nucleus ...
Small Business Success on the Web
... chemical behavior of an atom depends on its electron arrangement depends on the number of electrons in its outermost shell, the ...
... chemical behavior of an atom depends on its electron arrangement depends on the number of electrons in its outermost shell, the ...
05 Chemistry Basics with Flips 2011
... chemical behavior of an atom depends on its electron arrangement depends on the number of electrons in its outermost shell, the ...
... chemical behavior of an atom depends on its electron arrangement depends on the number of electrons in its outermost shell, the ...
Topics for Final
... Amplitude, wavelength, frequency Electromagnetic radiation, atomic emission spectrum Planck’s Constant = h (E= hv) Photons, photoelectric effect, ground state ...
... Amplitude, wavelength, frequency Electromagnetic radiation, atomic emission spectrum Planck’s Constant = h (E= hv) Photons, photoelectric effect, ground state ...
Chemistry for Bio 11
... Elements combine in chemical reactions to form compounds • Molecules- 2 or more atoms combined in a specific way • Compounds- different elements in a molecule, in exact, whole-number ratios, joined by a chemical bond • 2 major means of intramolecular chemical bonding: Covalent (incl. polar and nonp ...
... Elements combine in chemical reactions to form compounds • Molecules- 2 or more atoms combined in a specific way • Compounds- different elements in a molecule, in exact, whole-number ratios, joined by a chemical bond • 2 major means of intramolecular chemical bonding: Covalent (incl. polar and nonp ...
Resonance (chemistry)
In chemistry, resonance or mesomerism is a way of describing delocalized electrons within certain molecules or polyatomic ions where the bonding cannot be expressed by one single Lewis formula. A molecule or ion with such delocalized electrons is represented by several contributing structures (also called resonance structures or canonical forms).Each contributing structure can be represented by a Lewis structure, with only an integer number of covalent bonds between each pair of atoms within the structure. Several Lewis structures are used collectively to describe the actual molecular structure, which is an approximate intermediate between the canonical forms called a resonance hybrid. Contributing structures differ only in the position of electrons, not in the position of nuclei.Electron delocalization lowers the potential energy of the substance and thus makes it more stable than any of the contributing structures. The difference between the potential energy of the actual structure and that of the contributing structure with the lowest potential energy is called the resonance energy or delocalization energy.Resonance is distinguished from tautomerism and conformational isomerism, which involve the formation of isomers, thus the rearrangement of the nuclear positions.