Chemical reactions
... Reaction products = substances formed in a chemical reaction. They are written in the right term of the equation Because in a chemical reaction, the nature of atoms of the substances is not changed, the chemical equations are equalized so that the number of atoms of a certain element from the left t ...
... Reaction products = substances formed in a chemical reaction. They are written in the right term of the equation Because in a chemical reaction, the nature of atoms of the substances is not changed, the chemical equations are equalized so that the number of atoms of a certain element from the left t ...
classification of chemical reactions
... chemical reactions need a certain amount of energy to get started during a chemical reaction energy is either absorbed or released energy is needed to break chemical bonds broken bonds can form new chemical bond= new products ...
... chemical reactions need a certain amount of energy to get started during a chemical reaction energy is either absorbed or released energy is needed to break chemical bonds broken bonds can form new chemical bond= new products ...
The Nature of Chemical Reactions
... transferred from one reactant to another Rusting is an example of a redox reaction ...
... transferred from one reactant to another Rusting is an example of a redox reaction ...
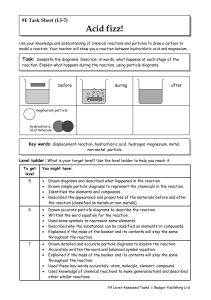
Year 9 Homework Task 9E-5 Reactions 5-7
... Use your knowledge and understanding of chemical reactions and particles to draw a cartoon to model a reaction. Your teacher will show you a reaction between hydrochloric acid and magnesium. ...
... Use your knowledge and understanding of chemical reactions and particles to draw a cartoon to model a reaction. Your teacher will show you a reaction between hydrochloric acid and magnesium. ...
Chapter 5 – Chemical Reactions
... If there are two compounds containing positive and negative groups, the reaction ...
... If there are two compounds containing positive and negative groups, the reaction ...
California Chemistry Standards Test
... 6. convert between C and K 7. absolute zero (0 K) Acids & Bases-(5) 1. properties of acids & bases and salt solutions 2. acids-hydrogen-ion donating 3. bases-hydrogen-ion accepting 4. strong and weak acids and bases 5. pH scale Solutions-(5) 1. solute and solvent 2. dissolving process at the molecul ...
... 6. convert between C and K 7. absolute zero (0 K) Acids & Bases-(5) 1. properties of acids & bases and salt solutions 2. acids-hydrogen-ion donating 3. bases-hydrogen-ion accepting 4. strong and weak acids and bases 5. pH scale Solutions-(5) 1. solute and solvent 2. dissolving process at the molecul ...
673 lab three
... reactions and give three example reactions and SHOW that they are balanced. Clearly indicate the role of COEFFICIENTS., B) DISCUSS CHEMICAL FORMULAS: Clearly define the meaning of SUBSCRIPTS and define the use of parenthesis with subscripts. Present the meaning of the subscripts in the compounds in ...
... reactions and give three example reactions and SHOW that they are balanced. Clearly indicate the role of COEFFICIENTS., B) DISCUSS CHEMICAL FORMULAS: Clearly define the meaning of SUBSCRIPTS and define the use of parenthesis with subscripts. Present the meaning of the subscripts in the compounds in ...
Chemical Equations and Reactions notes File
... Whole numbers in front of formula Distributes to numbers of atoms in formula specifies the number of moles in the reaction used to balance the equation ...
... Whole numbers in front of formula Distributes to numbers of atoms in formula specifies the number of moles in the reaction used to balance the equation ...
Title - Iowa State University
... 3. Which of the following statements about catalysts is false? a. A catalyst will speed up the rate of a reaction. b. Catalysts are used in very many commercially important chemical reactions. c. Catalytic converters are examples of heterogeneous catalysts. d. A catalyst can cause a nonspontaneous r ...
... 3. Which of the following statements about catalysts is false? a. A catalyst will speed up the rate of a reaction. b. Catalysts are used in very many commercially important chemical reactions. c. Catalytic converters are examples of heterogeneous catalysts. d. A catalyst can cause a nonspontaneous r ...
Lesson 6
... Turn to page 28 of the textbook and read "The Changes That Occur." 4. The substances present before a reaction are called the ________________. 5. The substances present after a reaction are called the _________________. 6. List the three main indicators that a chemical reaction has occurred. 7. Are ...
... Turn to page 28 of the textbook and read "The Changes That Occur." 4. The substances present before a reaction are called the ________________. 5. The substances present after a reaction are called the _________________. 6. List the three main indicators that a chemical reaction has occurred. 7. Are ...
Synthesis/Decomposition Reactions
... Name: _______________________________________________ Date: __________ Period: _________ ...
... Name: _______________________________________________ Date: __________ Period: _________ ...
Notes on Chapter 12 Chemical Equilibrium
... reaction (i.e. the energy required to break and form new bonds). ...
... reaction (i.e. the energy required to break and form new bonds). ...
Chemical reaction

A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive elements where both electronic and nuclear changes may occur.The substance (or substances) initially involved in a chemical reaction are called reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more products, which usually have properties different from the reactants. Reactions often consist of a sequence of individual sub-steps, the so-called elementary reactions, and the information on the precise course of action is part of the reaction mechanism. Chemical reactions are described with chemical equations, which symbolically present the starting materials, end products, and sometimes intermediate products and reaction conditions.Chemical reactions happen at a characteristic reaction rate at a given temperature and chemical concentration. Typically, reaction rates increase with increasing temperature because there is more thermal energy available to reach the activation energy necessary for breaking bonds between atoms.Reactions may proceed in the forward or reverse direction until they go to completion or reach equilibrium. Reactions that proceed in the forward direction to approach equilibrium are often described as spontaneous, requiring no input of free energy to go forward. Non-spontaneous reactions require input of free energy to go forward (examples include charging a battery by applying an external electrical power source, or photosynthesis driven by absorption of electromagnetic radiation in the form of sunlight).Different chemical reactions are used in combinations during chemical synthesis in order to obtain a desired product. In biochemistry, a consecutive series of chemical reactions (where the product of one reaction is the reactant of the next reaction) form metabolic pathways. These reactions are often catalyzed by protein enzymes. Enzymes increase the rates of biochemical reactions, so that metabolic syntheses and decompositions impossible under ordinary conditions can occur at the temperatures and concentrations present within a cell.The general concept of a chemical reaction has been extended to reactions between entities smaller than atoms, including nuclear reactions, radioactive decays, and reactions between elementary particles as described by quantum field theory.