Level shifts of rubidium Rydberg states due to binary interactions
... data in the calculation, we only include two-particle couplings with 兩具¯兩Vdd兩 ¯ 典兩 ⬎ 10−4n*4 / R3, where n* = n − ␦ᐉ is the effective principal quantum number. It is noted that all elements of Vdd are calculated before the small ones are discarded. Furthermore, in the sum in Eq. 共3兲 we only include ...
... data in the calculation, we only include two-particle couplings with 兩具¯兩Vdd兩 ¯ 典兩 ⬎ 10−4n*4 / R3, where n* = n − ␦ᐉ is the effective principal quantum number. It is noted that all elements of Vdd are calculated before the small ones are discarded. Furthermore, in the sum in Eq. 共3兲 we only include ...
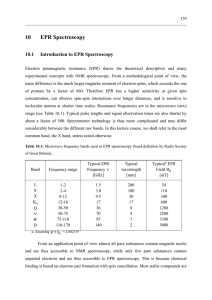
10 EPR Spectroscopy
... These defects determine the electronic and optical properties of these materials. EPR ...
... These defects determine the electronic and optical properties of these materials. EPR ...
Ground-state stability and criticality of two
... Our results show that the ground-state diagram of twoelectron atoms interacting via Yukawa potentials does not present a 2e− − 0e− line. That is, the systems always undergoes a 2e− − 1e− transition before losing both electrons as the screening grows. Even the numerical results are not accurate enoug ...
... Our results show that the ground-state diagram of twoelectron atoms interacting via Yukawa potentials does not present a 2e− − 0e− line. That is, the systems always undergoes a 2e− − 1e− transition before losing both electrons as the screening grows. Even the numerical results are not accurate enoug ...
Curriculum Vitae - Université Paris-Sud
... In particular, various metal ions were used widely as radical scavengers and redox indicators in the reduction or oxidation processes induced indirectly by the short-lived primary radiolytic species, allowing their identification and the calibration of their formation yield.6,7,8 Metal ions such as ...
... In particular, various metal ions were used widely as radical scavengers and redox indicators in the reduction or oxidation processes induced indirectly by the short-lived primary radiolytic species, allowing their identification and the calibration of their formation yield.6,7,8 Metal ions such as ...
Three-Level L-Type Atomic System Localized by the Parameters of
... 1 and ρij∗ = ρ ji . The effect of SGC is very sensiThe above equations are constrained by ρ11 + ρ 22 + ρ33 = tive to the orientations of the atomic dipole moments µ13 and µ12 . Here, the parameter p denotes the alignment of the two dipole moments and is defined as p = µ13 ⋅ µ12 µ13 ⋅ µ12 = cos θ wit ...
... 1 and ρij∗ = ρ ji . The effect of SGC is very sensiThe above equations are constrained by ρ11 + ρ 22 + ρ33 = tive to the orientations of the atomic dipole moments µ13 and µ12 . Here, the parameter p denotes the alignment of the two dipole moments and is defined as p = µ13 ⋅ µ12 µ13 ⋅ µ12 = cos θ wit ...
Document
... • A sample of hydrogen gas was collected over water at 21C and 0.9 atm. The volume of the container was 7.80 L. Calculate the mass of H2(g) collected. (Vapor pressure of water = 0.025 atm at 21C.) • (A) 0.283 g • (B) 435 g • (C) 0.571 g • (D) 7.14 g ...
... • A sample of hydrogen gas was collected over water at 21C and 0.9 atm. The volume of the container was 7.80 L. Calculate the mass of H2(g) collected. (Vapor pressure of water = 0.025 atm at 21C.) • (A) 0.283 g • (B) 435 g • (C) 0.571 g • (D) 7.14 g ...
chapter twenty-one transition metals and coordination chemistry
... The crystal field diagrams are different because the geometries of where the ligands point is different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 2 ...
... The crystal field diagrams are different because the geometries of where the ligands point is different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 2 ...
Document
... A. The transmission coefficient through the barrier depends on E, V and a B. The transmission coefficient increases when a decreases for a given E and V C. The transmission coefficient increases when V decreases for a given E and a D. The transmission coefficient increases when E decreases for a giv ...
... A. The transmission coefficient through the barrier depends on E, V and a B. The transmission coefficient increases when a decreases for a given E and V C. The transmission coefficient increases when V decreases for a given E and a D. The transmission coefficient increases when E decreases for a giv ...
Ionization

Ionization is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons to form ions, often in conjunction with other chemical changes. Ionization can result from the loss of an electron after collisions with sub atomic particles, collisions with other atoms, molecules and ions, or through the interaction with light. Heterolytic bond cleavage and heterolytic substitution reactions can result in the formation of ion pairs. Ionization can occur through radioactive decay by the internal conversion process, in which an excited nucleus transfers its energy to one of the inner-shell electrons causing it to be ejected.