![1 Solutions 4a (Chapter 4 problems) Chem151 [Kua]](http://s1.studyres.com/store/data/002731518_1-574ec10e88e667508364281b6325aeef-300x300.png)
1 Solutions 4a (Chapter 4 problems) Chem151 [Kua]
... Molecular pictures and limiting reactant calculations require a balanced chemical equation for the reaction under consideration. For this process, the balanced equation is CH4 + 2 H2O → CO2 + 4 H2. (a) There are 6 H2O molecules and 5 CH4 molecules in the picture. Divide number of ...
... Molecular pictures and limiting reactant calculations require a balanced chemical equation for the reaction under consideration. For this process, the balanced equation is CH4 + 2 H2O → CO2 + 4 H2. (a) There are 6 H2O molecules and 5 CH4 molecules in the picture. Divide number of ...
Moles 1 - pedagogics.ca
... This triangle is a useful shortcut for working out all the quantities involved in the equation. If any one of the sections of the triangle is covered up, the relationship between the other two quantities to give the covered quantity is revealed. For example, if mass of substance is covered, we are l ...
... This triangle is a useful shortcut for working out all the quantities involved in the equation. If any one of the sections of the triangle is covered up, the relationship between the other two quantities to give the covered quantity is revealed. For example, if mass of substance is covered, we are l ...
CHAPTER 9
... Ideal Stoichiometric Calculations A balanced chemical equation is the key step in all stoichiometric calculations, because the mole ratio is obtained directly from it. Solving any reaction stoichiometry problem must begin with a balanced equation. Chemical equations help us plan the amounts of react ...
... Ideal Stoichiometric Calculations A balanced chemical equation is the key step in all stoichiometric calculations, because the mole ratio is obtained directly from it. Solving any reaction stoichiometry problem must begin with a balanced equation. Chemical equations help us plan the amounts of react ...
chemistry - Textbooks Online
... Chemistry, a branch of science concerned with the properties, structures and composition of substances and their reactions with one another. Inorganic Chemistry studies the preparation, properties and reactions of all chemical elements and their compounds, except those of carbon. Organic Chemistry s ...
... Chemistry, a branch of science concerned with the properties, structures and composition of substances and their reactions with one another. Inorganic Chemistry studies the preparation, properties and reactions of all chemical elements and their compounds, except those of carbon. Organic Chemistry s ...
VOLUME 3 - ICHO 41-45 _opravené_
... the forty-five years of its existence. In the elaboration of this collection the editor had to face certain difficulties because the aim was not only to make use of past recordings but also to give them such a form that they may be used in practice and further chemical education. Consequently, it wa ...
... the forty-five years of its existence. In the elaboration of this collection the editor had to face certain difficulties because the aim was not only to make use of past recordings but also to give them such a form that they may be used in practice and further chemical education. Consequently, it wa ...
Concept based notes Chemistry Lab Manual
... Q. 73. Why, H2S gas passed in presence of NH4OH? Ans. When H2S gas is passed in alkaline medium or in presence of NH4OH, the H+ ions from the dissociation of H2S gas combine with hydroxyl ions (OH-) from the dissociation of NH4OH to from nearly unionised H2O. The removal of H+ ions from the solutio ...
... Q. 73. Why, H2S gas passed in presence of NH4OH? Ans. When H2S gas is passed in alkaline medium or in presence of NH4OH, the H+ ions from the dissociation of H2S gas combine with hydroxyl ions (OH-) from the dissociation of NH4OH to from nearly unionised H2O. The removal of H+ ions from the solutio ...
RUMPLE-DISSERTATION-2014 - SMARTech Home
... I would like to begin by thanking my advisors Dr. Charles Liotta and Dr. Charles Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty su ...
... I would like to begin by thanking my advisors Dr. Charles Liotta and Dr. Charles Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty su ...
Energetics
... a chemical reaction is independent of the route by which the chemical reaction takes place and depends only on the difference between the total enthalpy of the reactants and that of the products. ...
... a chemical reaction is independent of the route by which the chemical reaction takes place and depends only on the difference between the total enthalpy of the reactants and that of the products. ...
Chemical Transport Model - Technical Description
... CSIRO and the Bureau of Meteorology advise that the information contained in this publication comprises general statements based on scientific research. The reader is advised and needs to be aware that such information may be incomplete or unable to be used in any specific situation. No reliance or ...
... CSIRO and the Bureau of Meteorology advise that the information contained in this publication comprises general statements based on scientific research. The reader is advised and needs to be aware that such information may be incomplete or unable to be used in any specific situation. No reliance or ...
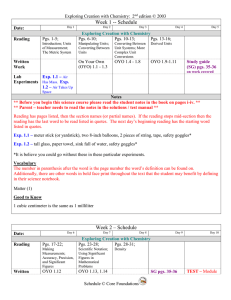
Week 1 -- Schedule
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
... ** Before you begin this science course please read the student notes in the book on pages i-iv. ** ** Parent – teacher needs to read the notes in the solutions / test manual ** Reading has pages listed, then the section names (or partial names). If the reading stops mid-section then the reading has ...
ELECTROCHEMICAL STUDY OF CORROSION PROCESSES IN
... Figure 6.7: Contributions of activation, degree of dissociation, and compressibility to corrosion rate of Type 304 SS as a function of pressure at 450 oC in deaerated 0.01 M HCl…………………………………………. 137 Figure 6.8: Electrochemical corrosion rate (standard deviation of the current noise) of Type 304 SS a ...
... Figure 6.7: Contributions of activation, degree of dissociation, and compressibility to corrosion rate of Type 304 SS as a function of pressure at 450 oC in deaerated 0.01 M HCl…………………………………………. 137 Figure 6.8: Electrochemical corrosion rate (standard deviation of the current noise) of Type 304 SS a ...
UNIT 1. SOME BASIC CONCEPTS OF CHEMISTRY Concept
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
... Q3- What is a chemical equation? What are its essential features? (L-2) Ans. the qualitative and quantitative representation of a chemical reaction in short form in terms of symbols and formulae is called chemical equation. For example, on heating calcium carbonate, it gives Caco3 →Ca0 + CO2 Essenti ...
Lab Manual (Eng. Medium)
... top (zero) calibration mark and then drained into a separate container until the calibration mark for the desired volume is reached. The remaining liquid is either discarded or returned to its original container. The maximum indicated capacity of some graduated pipettes is delivered by draining to a ...
... top (zero) calibration mark and then drained into a separate container until the calibration mark for the desired volume is reached. The remaining liquid is either discarded or returned to its original container. The maximum indicated capacity of some graduated pipettes is delivered by draining to a ...
PART 6-ICHO-26-30
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
THE DIFFUSION MECHANISM OF HYDROCARBONS IN... Jirong Xiao B.S., East China Institute of Chemical Technology
... processes such as air separation, linear paraffin separation, drying of cracked gas, pressure swing H 2 purification, xyleneseparation, and removal of organics from water (Ruthven 1984). ...
... processes such as air separation, linear paraffin separation, drying of cracked gas, pressure swing H 2 purification, xyleneseparation, and removal of organics from water (Ruthven 1984). ...
study guide spring 2012
... d. number of products. A chemical equation is balanced when the a. coefficients of the reactants equal the coefficients of the products. b. same number of each kind of atom appears in the reactants and in the products. c. products and reactants are the same chemicals. d. subscripts of the reactants ...
... d. number of products. A chemical equation is balanced when the a. coefficients of the reactants equal the coefficients of the products. b. same number of each kind of atom appears in the reactants and in the products. c. products and reactants are the same chemicals. d. subscripts of the reactants ...
Ternary nucleation of inorganic acids, ammonia, and water
... Figure 3 shows the dependence of nucleation rate on ammonia vapor concentration for the three systems at T ⫽298.15 K and RH⫽50%. The acid concentration is adjusted so that nucleation rate is 1 cm⫺3 s⫺1 when ammonia concentration is 109 molecules/cm3 . In this figure and for the rest of the paper, th ...
... Figure 3 shows the dependence of nucleation rate on ammonia vapor concentration for the three systems at T ⫽298.15 K and RH⫽50%. The acid concentration is adjusted so that nucleation rate is 1 cm⫺3 s⫺1 when ammonia concentration is 109 molecules/cm3 . In this figure and for the rest of the paper, th ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.