Chem 30A Final Exam
... 16. You are titrating an unknown quantity of sulfuric acid (H2SO4) with a 0.1950 M NaOH standard solution and you find it takes 32.50 mL to reach an endpoint (phenolpthalein color change). What is the amount of sulfuric acid present in moles? Hint: Write a balanced chemical reaction equation for th ...
... 16. You are titrating an unknown quantity of sulfuric acid (H2SO4) with a 0.1950 M NaOH standard solution and you find it takes 32.50 mL to reach an endpoint (phenolpthalein color change). What is the amount of sulfuric acid present in moles? Hint: Write a balanced chemical reaction equation for th ...
Mechanism and Elementary Reactions
... simplify the rate expressions for complex reaction networks. Without further redue, let’s get into the details! The important difference between a reaction with an observed stoichiometry and an elementary reaction is that the stoichiometry of an elementary reaction defines the concentration dependen ...
... simplify the rate expressions for complex reaction networks. Without further redue, let’s get into the details! The important difference between a reaction with an observed stoichiometry and an elementary reaction is that the stoichiometry of an elementary reaction defines the concentration dependen ...
Test 4 Review - Ralph C. Mahar
... fluoride is 7.9 x 10-10. What is the molar solubility of SrF2 at 25°C? SrF2 D Sr+2 + 2F[Sr2+][F-]2=7.9 x 10-10 [Sr2+]= x [F-]= 2x (x)(2x)2=7.9 x 10-10 x = 5.8 x 10-4M ...
... fluoride is 7.9 x 10-10. What is the molar solubility of SrF2 at 25°C? SrF2 D Sr+2 + 2F[Sr2+][F-]2=7.9 x 10-10 [Sr2+]= x [F-]= 2x (x)(2x)2=7.9 x 10-10 x = 5.8 x 10-4M ...
Free response review
... 1. A 50.0 gram sample of solid ammonium carbonate is placed in a closed evacuated 3.00L flask and heated to 400° C. It decomposed to produce ammonia, water, and carbon dioxide. The equilibrium constant, Kp, for the reaction is 0.295 at 400° C. a. Write the balanced equation. b. Write the Kp equilibr ...
... 1. A 50.0 gram sample of solid ammonium carbonate is placed in a closed evacuated 3.00L flask and heated to 400° C. It decomposed to produce ammonia, water, and carbon dioxide. The equilibrium constant, Kp, for the reaction is 0.295 at 400° C. a. Write the balanced equation. b. Write the Kp equilibr ...
State of Equilibrium
... Stable equilibrium is the most frequently met state in thermodynamics, and most systems exist in this state. Most of the theories of thermodynamics are based on stable equilibrium, which might be more correctly named thermostatics. The measurement of thermodynamic properties relies on the measuring ...
... Stable equilibrium is the most frequently met state in thermodynamics, and most systems exist in this state. Most of the theories of thermodynamics are based on stable equilibrium, which might be more correctly named thermostatics. The measurement of thermodynamic properties relies on the measuring ...
ACS Practice Test 1
... They lower the vapor pressure of water when dissolved in it. (E) They raise the boiling point of water when dissolved in it. 42. The addition of a catalyst in a chemical reaction (A) increases the concentration of products at equilibrium. (B) increases the fraction of reactant molecules with a given ...
... They lower the vapor pressure of water when dissolved in it. (E) They raise the boiling point of water when dissolved in it. 42. The addition of a catalyst in a chemical reaction (A) increases the concentration of products at equilibrium. (B) increases the fraction of reactant molecules with a given ...
Specification
... Labelled as: quantity / unit, e.g. c / mol L–1. Only values will then be written on the axes or in a table. ...
... Labelled as: quantity / unit, e.g. c / mol L–1. Only values will then be written on the axes or in a table. ...
List of Definitions for AS Chemistry
... increases when the matter or energy in the system becomes more random in its arrangement. A system that has a high degree of disorder/randomness is said to have a large entropy. Gases have the highest entropy followed by liquids and solids. ...
... increases when the matter or energy in the system becomes more random in its arrangement. A system that has a high degree of disorder/randomness is said to have a large entropy. Gases have the highest entropy followed by liquids and solids. ...
Chapter 4 The Study of Chemical Reactions
... Thermodynamics is the branch of chemistry that deals with the energy changes accompanying chemical and physical transformations. These energy changes are most useful for describing the properties of systems at equilibrium. The equilibrium concentrations of reactants and products are governed by the ...
... Thermodynamics is the branch of chemistry that deals with the energy changes accompanying chemical and physical transformations. These energy changes are most useful for describing the properties of systems at equilibrium. The equilibrium concentrations of reactants and products are governed by the ...
Avogadro`s Law is relation between
... 7- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–8 M c. 3.0 × 10–4 M b. 10–10 M d. 2.5 × 10–11 M 8- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–1 M c. 2.6 × 10–7 M b. 10–13 M d. 1.2 × 10–12 ...
... 7- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–8 M c. 3.0 × 10–4 M b. 10–10 M d. 2.5 × 10–11 M 8- Calculate the value of [–OH] from the given [H3O+] and label the solution as acidic or basic. a. 10–1 M c. 2.6 × 10–7 M b. 10–13 M d. 1.2 × 10–12 ...
Unit 5 Practice Problems (with answers at end) - H
... 14. Calculate Go for the reaction: CH4(g) + O2(g) CO2(g) + H2O(ℓ) ,(not balanced) using the thermodynamic tables in your study guide. Is this reaction spontaneous at room temperature? 15. Calculate So for the reaction above. What does this value suggest? Why is the sign what it is? 16. Assuming t ...
... 14. Calculate Go for the reaction: CH4(g) + O2(g) CO2(g) + H2O(ℓ) ,(not balanced) using the thermodynamic tables in your study guide. Is this reaction spontaneous at room temperature? 15. Calculate So for the reaction above. What does this value suggest? Why is the sign what it is? 16. Assuming t ...
Test 9 Review - Evan`s Chemistry Corner
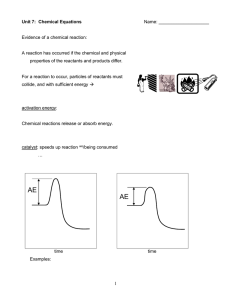
... Collision theory. In order for a reaction to occur, particles of the reactant must collide. Not all collisions cause reactions. An effective collision is one in which the colliding particles approach each other at the proper angle and with the proper amount of energy to cause a reaction. The greater ...
... Collision theory. In order for a reaction to occur, particles of the reactant must collide. Not all collisions cause reactions. An effective collision is one in which the colliding particles approach each other at the proper angle and with the proper amount of energy to cause a reaction. The greater ...
classical notions of heterogeneous freezing
... It is seen from Fig. 3 that for solids or liquids, the change in volume with pressure is small, so V can be treated as a constant and the integral can be considered approximately as ≈ V∆p. Since at normal pressure the value of V∆p is small, we can suppose that the Gibbs energies of solids and liquid ...
... It is seen from Fig. 3 that for solids or liquids, the change in volume with pressure is small, so V can be treated as a constant and the integral can be considered approximately as ≈ V∆p. Since at normal pressure the value of V∆p is small, we can suppose that the Gibbs energies of solids and liquid ...
402 - Sydenham High School
... to distinguish between strength and concentration. This takes mental effort The fact that sometimes a chemical can act as an acid in one reaction and as a base in another reaction is interesting. I now see how important empirical evidence is to the study of chemistry. The ultimate way of knowing in ...
... to distinguish between strength and concentration. This takes mental effort The fact that sometimes a chemical can act as an acid in one reaction and as a base in another reaction is interesting. I now see how important empirical evidence is to the study of chemistry. The ultimate way of knowing in ...
Unit 2: Chemical Reactions
... the total mass of the products • Equations must balance on both sides of the equation ...
... the total mass of the products • Equations must balance on both sides of the equation ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.