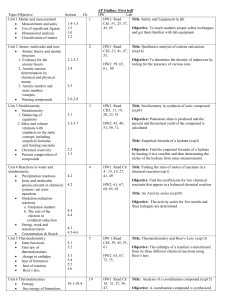
Kémiai technológia I
... 1. 1 The properties of gases: perfect gas, non-ideal gases. The perfect gas equations. The ideal gas law. Non-ideal gases: the compression factor, the virial equation, the van der Waals equation and its parameters, critical point. Fundamentals of the kinetic theory of gases. The molecular origin of ...
... 1. 1 The properties of gases: perfect gas, non-ideal gases. The perfect gas equations. The ideal gas law. Non-ideal gases: the compression factor, the virial equation, the van der Waals equation and its parameters, critical point. Fundamentals of the kinetic theory of gases. The molecular origin of ...
Conjugate Acids and Bases
... They proceed to a state of chemical equilibrium in which the ratio of concentrations of reactants and products is constant. Equilibrium-constant expressions are algebraic equations that describe the concentration relationships among reactants and products at equilibrium. The Equilibrium State The fi ...
... They proceed to a state of chemical equilibrium in which the ratio of concentrations of reactants and products is constant. Equilibrium-constant expressions are algebraic equations that describe the concentration relationships among reactants and products at equilibrium. The Equilibrium State The fi ...
The retrospect of the science and the thermodynamics
... Calculating the work requirement and heat transfer to a series of chemical engineering process, a plants. Prediction or estimation the composition in each phase at equilibria states. Phase equilibrium problems ...
... Calculating the work requirement and heat transfer to a series of chemical engineering process, a plants. Prediction or estimation the composition in each phase at equilibria states. Phase equilibrium problems ...
Basics of thermodynamics
... where the bar indicates an imperfect differential (i.e., it is not the differential of some hypothetical ¯ = −P dV . function Q). Remember that the mechanical work done is given by dW We shall generally consider closed systems but ones that are composite of two or more simple systems. Constraints th ...
... where the bar indicates an imperfect differential (i.e., it is not the differential of some hypothetical ¯ = −P dV . function Q). Remember that the mechanical work done is given by dW We shall generally consider closed systems but ones that are composite of two or more simple systems. Constraints th ...
11 Thermodynamics and Thermochemistry
... initial state to the final state. The state functions we will be working with this chapter are listed below: P, pressure; ...
... initial state to the final state. The state functions we will be working with this chapter are listed below: P, pressure; ...
document
... energy in the reactants will be released as heat or the solution can supply heat to allow formation of a product with a higher potential energy ii) The solution will absorb or release energy during the reaction. We will see this as a temperature change ...
... energy in the reactants will be released as heat or the solution can supply heat to allow formation of a product with a higher potential energy ii) The solution will absorb or release energy during the reaction. We will see this as a temperature change ...
2 - Yale University
... of Chem 125 students at Yale and may not be copied or distributed further. It is not readily understood without reference to notes or the wiki from the lecture. ...
... of Chem 125 students at Yale and may not be copied or distributed further. It is not readily understood without reference to notes or the wiki from the lecture. ...
Weak Acids and Bases Practice -- Chemistry 121A
... yield. It runs from 0% (all reactants) to 100% (all products). This is simply more convenient than Q, which runs from 0 to infinity. The way I look at it is this for a weak acid: %ionization = [A−]eq/[HA]o x 100% That is, the “final” concentration of A− at equilibrium divided by the “initial” concen ...
... yield. It runs from 0% (all reactants) to 100% (all products). This is simply more convenient than Q, which runs from 0 to infinity. The way I look at it is this for a weak acid: %ionization = [A−]eq/[HA]o x 100% That is, the “final” concentration of A− at equilibrium divided by the “initial” concen ...
Chapter 5 Chemical Equilibrium 1 State whether each of the
... matter what path is taken from reactants to products. Some examples are S, H, G, p, V, T. ...
... matter what path is taken from reactants to products. Some examples are S, H, G, p, V, T. ...
Name - cloudfront.net
... 43. What would likely happen (how would it feel) if you were to touch the flask in which an endothermic reaction were occurring? 44. Standard conditions of temperature and pressure for a thermochemical equation are __ and __kPa. 45. If heat is released by a chemical system, an equal amount of heat w ...
... 43. What would likely happen (how would it feel) if you were to touch the flask in which an endothermic reaction were occurring? 44. Standard conditions of temperature and pressure for a thermochemical equation are __ and __kPa. 45. If heat is released by a chemical system, an equal amount of heat w ...
Lecture 7. Fundamentals of atmospheric chemistry: Part 2 1
... These constants are available from the tables. Therefore equilibrium constants can be used to find the concentrations of reactants and products for reactions that have reached equilibrium. ...
... These constants are available from the tables. Therefore equilibrium constants can be used to find the concentrations of reactants and products for reactions that have reached equilibrium. ...
Acc
... Count atoms on both sides (polyatomics can be counted as one if they are on both sides) Balance using coefficients (save H & O for last if they’re present) Check balancing and make sure coefficients are in the lowest ratio ...
... Count atoms on both sides (polyatomics can be counted as one if they are on both sides) Balance using coefficients (save H & O for last if they’re present) Check balancing and make sure coefficients are in the lowest ratio ...
Kinetics of metamorphic reactions
... A kinetic approach to studying metamorphic rocks is different from an “equilibrium approach”, and may in many cases be the more realistic approach to studying metamorphism. Nevertheless, careful mineralogical and textural analysis of metamorphic rocks often validates the use of an equilibrium approa ...
... A kinetic approach to studying metamorphic rocks is different from an “equilibrium approach”, and may in many cases be the more realistic approach to studying metamorphism. Nevertheless, careful mineralogical and textural analysis of metamorphic rocks often validates the use of an equilibrium approa ...
Topic/Objective - cloudfront.net
... Title: Molecular mass of a volatile liquid (exp3) Objective: A small amount of a volatile liquid inside a small test tube and then heated in boiling water until all the liquid vaporizes and fills the tube as excess water escapes. After the gas is cooled, the mass, volume, and pressure is measured to ...
... Title: Molecular mass of a volatile liquid (exp3) Objective: A small amount of a volatile liquid inside a small test tube and then heated in boiling water until all the liquid vaporizes and fills the tube as excess water escapes. After the gas is cooled, the mass, volume, and pressure is measured to ...
Balancing Single Replacement Reactions - Kossmann
... Name: __________________________ Class: ____________________ Date: _____________ ...
... Name: __________________________ Class: ____________________ Date: _____________ ...
KINETICS AND EQUILIBRIUM
... in water before being reacted together. When an ionic substance is dissolved in water the crystal lattice is broken and ions are free to move about, collide and react. Meaning no bonds need to be broken for the reaction to occur. 2. Concentration of Reactants a. An increase in the concentration (amo ...
... in water before being reacted together. When an ionic substance is dissolved in water the crystal lattice is broken and ions are free to move about, collide and react. Meaning no bonds need to be broken for the reaction to occur. 2. Concentration of Reactants a. An increase in the concentration (amo ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.