THERMODYNAMICS. Elements of Physical Chemistry. By P. Atkins
... Since work done by system = pex∆V System at equilibrium when pex = pint (mechanical equilibrium) Change either pressure to get reversible work i.e. pex > pint or pint > pex at constant temperature by an infinitesimal ...
... Since work done by system = pex∆V System at equilibrium when pex = pint (mechanical equilibrium) Change either pressure to get reversible work i.e. pex > pint or pint > pex at constant temperature by an infinitesimal ...
200 ways to pass the regents
... 108. Molarity is a way to measure the concentration of a solution. Molarity is equal to the number of moles of solute divided by the number of liters of solution. The formula is on the back of the reference tables. 109. Percent by mass = mass of the part / mass of the whole x 100% 110. Parts per mil ...
... 108. Molarity is a way to measure the concentration of a solution. Molarity is equal to the number of moles of solute divided by the number of liters of solution. The formula is on the back of the reference tables. 109. Percent by mass = mass of the part / mass of the whole x 100% 110. Parts per mil ...
(activity) of hydrogen ions
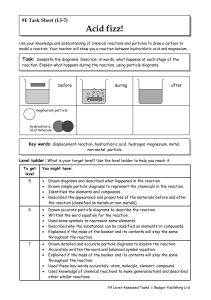
... metal oxide, hydroxide or carbonate, often of a transition metal like iron, zinc or copper). (1)The metal, oxide, hydroxide or carbonate is stirred with the acid and maybe heated to speed up the reaction (2). When no more of the solid dissolves the solution can be partly evaporated with further heat ...
... metal oxide, hydroxide or carbonate, often of a transition metal like iron, zinc or copper). (1)The metal, oxide, hydroxide or carbonate is stirred with the acid and maybe heated to speed up the reaction (2). When no more of the solid dissolves the solution can be partly evaporated with further heat ...
Handout - UNT Chemistry
... The equilibrium concentrations (or pressures) will be at the extent of reaction at which the Gibbs function of the system will be at a minimum. The equilibrium may lie: (1) Close to pure A: The reaction "doesn't go" (2) Close to pure B: The reaction "proceeds to completion" (3) At a point where ther ...
... The equilibrium concentrations (or pressures) will be at the extent of reaction at which the Gibbs function of the system will be at a minimum. The equilibrium may lie: (1) Close to pure A: The reaction "doesn't go" (2) Close to pure B: The reaction "proceeds to completion" (3) At a point where ther ...
Thermochemistry (Energy Relationships in Chemical Reactions
... It is not possible to measure the internal energy of any system. However, we can measure the changes in the internal energy. ...
... It is not possible to measure the internal energy of any system. However, we can measure the changes in the internal energy. ...
Follow Along Notes - Jackson County School System
... b. The same equilibrium condition would result if we started with only pure ZnCO3(s) in a closed container as if we started with only pure ZnO(s) in a closed container. c. Introducing 1.0 atm pressure of N2(g) into the system at equilibrium in a closed container would result in more ZnCO3(s) being f ...
... b. The same equilibrium condition would result if we started with only pure ZnCO3(s) in a closed container as if we started with only pure ZnO(s) in a closed container. c. Introducing 1.0 atm pressure of N2(g) into the system at equilibrium in a closed container would result in more ZnCO3(s) being f ...
AP `99 Multiple Choice
... (NH4)2SO4(s) (E) Sr(NO3)2(s) 66. When solid ammonium chloride, NH4Cl(s), is added to water at 25_C it dissolves and the temperature of the solution decreases. Which of the following is true for the values of _H and _S for the dissolving process? (A) Positive ...
... (NH4)2SO4(s) (E) Sr(NO3)2(s) 66. When solid ammonium chloride, NH4Cl(s), is added to water at 25_C it dissolves and the temperature of the solution decreases. Which of the following is true for the values of _H and _S for the dissolving process? (A) Positive ...
Balancing Chemical Equations Using Algebra
... Step 1. Assign a variable to each unknown coefficient. ...
... Step 1. Assign a variable to each unknown coefficient. ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.