Document
... There are seven base units, and each of these base units are nominally dimensionally independent. length(m), mass(kg), time(s), temperature(K), mole(mol), (electric current(A), Luminous intensity(cd)) From these seven base units, several other units are derived, which means the derived units can be ...
... There are seven base units, and each of these base units are nominally dimensionally independent. length(m), mass(kg), time(s), temperature(K), mole(mol), (electric current(A), Luminous intensity(cd)) From these seven base units, several other units are derived, which means the derived units can be ...
Lecture 6 – Thermochemistry
... State functions Always have the same value whenever the system is in the same state. ...
... State functions Always have the same value whenever the system is in the same state. ...
MC84 - Southchemistry.com
... (B) substance B is not involved in any of the steps in the mechanism of the reaction (C) substance B is not involved in the rate-determining step of the mechanism, but is involved in subsequent steps (D) substance B is probably a catalyst, and as such, its effect on the rate of the reaction does not ...
... (B) substance B is not involved in any of the steps in the mechanism of the reaction (C) substance B is not involved in the rate-determining step of the mechanism, but is involved in subsequent steps (D) substance B is probably a catalyst, and as such, its effect on the rate of the reaction does not ...
Example - cloudfront.net
... a) Balance elements that appear in more than one compound __________ (NH4)2CO3 NH3 + CO2 + H2O b) Balance __________________ as though they are one item as long as the ion stays together as a group on each side of the arrow. Al + CuSO4 Al2(SO4)3 + Cu c) If you can’t seem to get it balanced, ____ ...
... a) Balance elements that appear in more than one compound __________ (NH4)2CO3 NH3 + CO2 + H2O b) Balance __________________ as though they are one item as long as the ion stays together as a group on each side of the arrow. Al + CuSO4 Al2(SO4)3 + Cu c) If you can’t seem to get it balanced, ____ ...
C:\exams\June\June_06\chemistry\final\Chemistry 3202 June 2006
... 51.(c) At 1000 K, sulfur dioxide is converted into sulfur trioxide, as shown below. 2 SO2(g) + O2(g) ...
... 51.(c) At 1000 K, sulfur dioxide is converted into sulfur trioxide, as shown below. 2 SO2(g) + O2(g) ...
Final
... Perform molarity calculations and conversions Be able to develop a precipitation and acid/base neutralization reaction given the names of the starting materials Determine whether a material is soluble or insoluble Determine whether a precipitation and acid/base neutralization reaction occurs Write c ...
... Perform molarity calculations and conversions Be able to develop a precipitation and acid/base neutralization reaction given the names of the starting materials Determine whether a material is soluble or insoluble Determine whether a precipitation and acid/base neutralization reaction occurs Write c ...
Chemical Equations PowerPoint
... appearance of a product in a chemical reaction a) A chemical reaction that takes a long time to happen has a slow reaction rate. b) A chemical reaction that takes a short time to happen has a fast reaction rate. ...
... appearance of a product in a chemical reaction a) A chemical reaction that takes a long time to happen has a slow reaction rate. b) A chemical reaction that takes a short time to happen has a fast reaction rate. ...
Reading-Chem v Phys
... remains the same. A change in temperature may also bring about a physical change. The melting of ice, the freezing of water, the conversion of water to steam, and the condensation of steam to water are all examples of physical changes. We know that these physical properties of water are the same for ...
... remains the same. A change in temperature may also bring about a physical change. The melting of ice, the freezing of water, the conversion of water to steam, and the condensation of steam to water are all examples of physical changes. We know that these physical properties of water are the same for ...
Chemistry FINAL: CONTENT Review Packet
... 3. What is electronegativity? 4. What is the most electronegative element? _______________________ 5. Write whether an increase or decrease of a trend occurs as you go across the Periodic Table from left to right in a period, and when you go from top to bottom in a group. Across a Period ...
... 3. What is electronegativity? 4. What is the most electronegative element? _______________________ 5. Write whether an increase or decrease of a trend occurs as you go across the Periodic Table from left to right in a period, and when you go from top to bottom in a group. Across a Period ...
Exam #2
... 44. The metal calcium reacts with molecular hydrogen to form a compound. All of the following statements concerning this compound are true EXCEPT: (A) Its formula is CaH2. (B) It is ionic. (C) It is solid at room temperatur(E) (D) When added to water, it reacts to produce H2 gas. (E) When added to w ...
... 44. The metal calcium reacts with molecular hydrogen to form a compound. All of the following statements concerning this compound are true EXCEPT: (A) Its formula is CaH2. (B) It is ionic. (C) It is solid at room temperatur(E) (D) When added to water, it reacts to produce H2 gas. (E) When added to w ...
2008 Equilibrium -- without math (PowerPoint 13 MB)
... changes as time goes by. When a chemical reaction has reached the equilibrium state, the concentrations of reactants and products remain constant over time, and there are no visible changes in the system. Equilibrium 2007-2008 ...
... changes as time goes by. When a chemical reaction has reached the equilibrium state, the concentrations of reactants and products remain constant over time, and there are no visible changes in the system. Equilibrium 2007-2008 ...
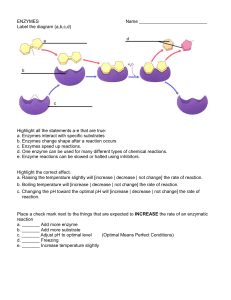
ENZYMES
... a. _______ Add more enzyme b. _______ Add more substrate c. _______ Adjust pH to optimal level (Optimal Means Perfect Conditions) d. _______ Freezing e. _______ Increase temperature slightly ...
... a. _______ Add more enzyme b. _______ Add more substrate c. _______ Adjust pH to optimal level (Optimal Means Perfect Conditions) d. _______ Freezing e. _______ Increase temperature slightly ...
Exam 4 - Chemistry Courses
... What mass of this substance must evaporate (at constant T) in order to freeze 2 moles of water at 20 °C to ice at 0 °C? The heat of vaporization of CCl2F2 is 289 J/g, the heat of fusion of water is 334 J/g , and the specific heat of water is 4.18 J/g-K. A. 52.0 g B. 26.0 g C. 15.0 g D. 3.01 g E. 2.8 ...
... What mass of this substance must evaporate (at constant T) in order to freeze 2 moles of water at 20 °C to ice at 0 °C? The heat of vaporization of CCl2F2 is 289 J/g, the heat of fusion of water is 334 J/g , and the specific heat of water is 4.18 J/g-K. A. 52.0 g B. 26.0 g C. 15.0 g D. 3.01 g E. 2.8 ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.