Chapter 12 Stoichiometry - Conejo Valley Unified School
... • Stoichiometry is the part of chemistry that studies amounts of reactants and products that are involved in reactions. ...
... • Stoichiometry is the part of chemistry that studies amounts of reactants and products that are involved in reactions. ...
Topic 1 Review - Capital High School
... 13. 6.0 mol of aluminium reacts with oxygen to form aluminium oxide. What is the amount of oxygen, in mol, needed for complete reaction? 4Al(s) + 3O2(g) → 2Al2O3(s) 14. What is the total number of nitrogen atoms in two mol of NH4NO3? 15. On analysis, a compound with molar mass 60 g mol-1 was found t ...
... 13. 6.0 mol of aluminium reacts with oxygen to form aluminium oxide. What is the amount of oxygen, in mol, needed for complete reaction? 4Al(s) + 3O2(g) → 2Al2O3(s) 14. What is the total number of nitrogen atoms in two mol of NH4NO3? 15. On analysis, a compound with molar mass 60 g mol-1 was found t ...
IB Chemistry Review. Unit I. Topics 2
... 13. 6.0 mol of aluminium reacts with oxygen to form aluminium oxide. What is the amount of oxygen, in mol, needed for complete reaction? 4Al(s) + 3O2(g) → 2Al2O3(s) 14. What is the total number of nitrogen atoms in two mol of NH4NO3? 15. On analysis, a compound with molar mass 60 g mol-1 was found t ...
... 13. 6.0 mol of aluminium reacts with oxygen to form aluminium oxide. What is the amount of oxygen, in mol, needed for complete reaction? 4Al(s) + 3O2(g) → 2Al2O3(s) 14. What is the total number of nitrogen atoms in two mol of NH4NO3? 15. On analysis, a compound with molar mass 60 g mol-1 was found t ...
E:\My Documents\sch3u\SCH3Ureview.wpd
... a) How many Nitrogen molecules would you have? b) How many Nitrogen atoms would you have? c) Why are these two numbers not the same? 7) What is the percent composition by mass of the compounds: a) H2O b) CO2 c) HCN d) Al2(CO3)3 8) A compound is found to have the following percentage composition by m ...
... a) How many Nitrogen molecules would you have? b) How many Nitrogen atoms would you have? c) Why are these two numbers not the same? 7) What is the percent composition by mass of the compounds: a) H2O b) CO2 c) HCN d) Al2(CO3)3 8) A compound is found to have the following percentage composition by m ...
Double-Replacement Reactions - Fort Thomas Independent Schools
... • The classification scheme described in this section provides an introduction to five basic types of reactions: • Synthesis (Combination) • Decomposition • Single-displacement (replacement) • Double-displacement (replacement) • Combustion reactions ...
... • The classification scheme described in this section provides an introduction to five basic types of reactions: • Synthesis (Combination) • Decomposition • Single-displacement (replacement) • Double-displacement (replacement) • Combustion reactions ...
Determination of K of Weak Acids
... 3. Measure out a small quantity (0.15–0.20 g) of the unknown into each weighing dish. Note: It is not necessary to know the exact mass of each sample. 4. Using a graduated cylinder, precisely measure 50.0 mL of distilled water into a 150-mL beaker. 5. Transfer sample #1 to the water in the beaker an ...
... 3. Measure out a small quantity (0.15–0.20 g) of the unknown into each weighing dish. Note: It is not necessary to know the exact mass of each sample. 4. Using a graduated cylinder, precisely measure 50.0 mL of distilled water into a 150-mL beaker. 5. Transfer sample #1 to the water in the beaker an ...
EXAM 3 NAME____KEY –Answers highlighted______(1 pt.) Dr
... a) the quantity of heat needed to change 1.00 g of a substance by 1.00 K. b) the mass of a substance that 1.00 J of energy will heat by 1.00 K. c) the mass of a substance that 1.00 cal of energy will heat by 1.00 K. d) the temperature change undergone when 1.00 g of a substance absorbs 4.184 J. e) t ...
... a) the quantity of heat needed to change 1.00 g of a substance by 1.00 K. b) the mass of a substance that 1.00 J of energy will heat by 1.00 K. c) the mass of a substance that 1.00 cal of energy will heat by 1.00 K. d) the temperature change undergone when 1.00 g of a substance absorbs 4.184 J. e) t ...
Chapter 8 Thermochemistry: Chemical Energy
... Standard Heats of Formation Standard Heat of Formation (DHof ): The enthalpy change for the formation of 1 mol of a substance in its standard state from its constituent elements in their ...
... Standard Heats of Formation Standard Heat of Formation (DHof ): The enthalpy change for the formation of 1 mol of a substance in its standard state from its constituent elements in their ...
Review Package
... Types of atoms involved Types of bond (ionic or covalent) Electrons (shared or transferred) Dissolve in water (yes or no) Conducts electricity Example: ...
... Types of atoms involved Types of bond (ionic or covalent) Electrons (shared or transferred) Dissolve in water (yes or no) Conducts electricity Example: ...
chemistry 11 exam review
... 5. What is the new volume of 750.0 mL of gas at 700.0 torr when the pressure is changed to 800.0 torr? (656.3 torr) 6. 45 mL of gas at 15C and 790 torr is changed to 23C and 810 torr. What is the new volume? (45 mL) 7. 175 mL of gas at –30.0C and 2.57 atm is changed to standard conditions. What i ...
... 5. What is the new volume of 750.0 mL of gas at 700.0 torr when the pressure is changed to 800.0 torr? (656.3 torr) 6. 45 mL of gas at 15C and 790 torr is changed to 23C and 810 torr. What is the new volume? (45 mL) 7. 175 mL of gas at –30.0C and 2.57 atm is changed to standard conditions. What i ...
Fractional Composition
... and B, instead of HA and A• Equation 3 gives the fraction in the form BH+ • Equation 4 gives the fraction in the form B • Ka is then the acid dissociation constant for BH+ (which is Kw/Kb) ...
... and B, instead of HA and A• Equation 3 gives the fraction in the form BH+ • Equation 4 gives the fraction in the form B • Ka is then the acid dissociation constant for BH+ (which is Kw/Kb) ...
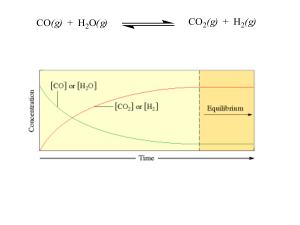
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.