B.Sc Chemistry - Calicut University
... 2. Chemistry in everyday life 3. Plastics and rubbers in everyday life In the sixth semester there are five elective courses. An institution can chose any one of ...
... 2. Chemistry in everyday life 3. Plastics and rubbers in everyday life In the sixth semester there are five elective courses. An institution can chose any one of ...
STOICHIOMETRY via ChemLog - Small
... Avogadro’s number is the number of particles of substance in a mole. Just as 1 dozen means 12 of something, regardless of what it is - eggs, diamonds, molecules, 1 mole means 6.022 x 1023 of something. There are Avogardo’s number, 6.022 x 1023, of particles in every mole. That’s a lot! ...
... Avogadro’s number is the number of particles of substance in a mole. Just as 1 dozen means 12 of something, regardless of what it is - eggs, diamonds, molecules, 1 mole means 6.022 x 1023 of something. There are Avogardo’s number, 6.022 x 1023, of particles in every mole. That’s a lot! ...
chem 102 class notes - Louisiana Tech University
... be achieved from a given set of initial concentrations. The reaction quotient is used for predicting the net direction of equilibrium reactions. Reaction Quotient (Q) When the reactants and products of a given chemical reaction are mixed, it is useful to know whether the mixture is at equilibrium o ...
... be achieved from a given set of initial concentrations. The reaction quotient is used for predicting the net direction of equilibrium reactions. Reaction Quotient (Q) When the reactants and products of a given chemical reaction are mixed, it is useful to know whether the mixture is at equilibrium o ...
Chapter 6 Chemical Reactions
... (c) The amount of the diammine complex, Ag(NH3)2+(aq), increases with time as more and more AgCl(s) and NH3(aq) react. Problem 11.8. Molecules move faster at higher temperatures. (Their internal motions (vibrations and rotations) also gain more energy, so vibrations and rotations are more active.) M ...
... (c) The amount of the diammine complex, Ag(NH3)2+(aq), increases with time as more and more AgCl(s) and NH3(aq) react. Problem 11.8. Molecules move faster at higher temperatures. (Their internal motions (vibrations and rotations) also gain more energy, so vibrations and rotations are more active.) M ...
Cliffs Notes
... Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
... Technical Editor: Christopher Bushee Production Proofreader: Joel K. Draper Hungry Minds Indianapolis Production Services ...
Stoichiometry and the Mole - 2012 Book Archive
... Although the number of things in a mole is known to eight decimal places, it is usually fine to use only two or three decimal places in calculations. The numerical value of things in a mole is often called Avogadro’s number (NA), which is also known as the Avogadro constant, after Amadeo Avogadro, a ...
... Although the number of things in a mole is known to eight decimal places, it is usually fine to use only two or three decimal places in calculations. The numerical value of things in a mole is often called Avogadro’s number (NA), which is also known as the Avogadro constant, after Amadeo Avogadro, a ...
Chapter 16 Controlling the yield of reactions
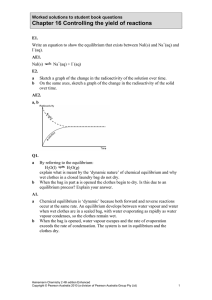
... is 48.8 at 455°C. An equilibrium mixture in a 2.0 L vessel at this temperature contains 0.220 mol of H2 and 0.110 mol of I2. a Calculate the concentration of HI in this mixture. b Another mixture was prepared by placing 4.0 mol of HI in a 2.0 L vessel at 330°C. At equilibrium 0.44 mol of H2 and 0.44 ...
... is 48.8 at 455°C. An equilibrium mixture in a 2.0 L vessel at this temperature contains 0.220 mol of H2 and 0.110 mol of I2. a Calculate the concentration of HI in this mixture. b Another mixture was prepared by placing 4.0 mol of HI in a 2.0 L vessel at 330°C. At equilibrium 0.44 mol of H2 and 0.44 ...
RIKEN Accelerator Progress Report
... ions were investigated. In addition to these atomic collision experiments, study of beam-foil spectroscopy has started this year and its first report is given in this volume. Some theoretical activities also began to work from this year mainly in the field of heavy particle collision, rearrangement ...
... ions were investigated. In addition to these atomic collision experiments, study of beam-foil spectroscopy has started this year and its first report is given in this volume. Some theoretical activities also began to work from this year mainly in the field of heavy particle collision, rearrangement ...
Specification and sample assessment material - Edexcel
... The best way to develop practical and investigative skills is to embed practical activities in your teaching of theory. The development of knowledge and skills can happen together, leading to secure acquisition of knowledge and skills. There are some practicals in the specification content, which st ...
... The best way to develop practical and investigative skills is to embed practical activities in your teaching of theory. The development of knowledge and skills can happen together, leading to secure acquisition of knowledge and skills. There are some practicals in the specification content, which st ...
Experimental Chemistry I
... Purpose: A mixture of two different mole contents possesses different saturation characteristics. KClO4 contaminated with KMnO4 is dissolved in deionized water. In order to separate these two compounds, the mixture has to be deprived of impurities. These impurities are a reaction by-product, which f ...
... Purpose: A mixture of two different mole contents possesses different saturation characteristics. KClO4 contaminated with KMnO4 is dissolved in deionized water. In order to separate these two compounds, the mixture has to be deprived of impurities. These impurities are a reaction by-product, which f ...
AS Chemistry Teacher Handbook
... Candidates should know that 12C is used as the standard in comparing relative masses. Candidates should be able to use relative atomic masses to calculate relative formula masses. Candidates will not be expected to draw a diagram of ...
... Candidates should know that 12C is used as the standard in comparing relative masses. Candidates should be able to use relative atomic masses to calculate relative formula masses. Candidates will not be expected to draw a diagram of ...
KCl + O KClO 3 → However, this equation is not balanced, since
... Writing a balanced chemical equation: In order to write a chemical equation, it is essential to know what substances are reacting and what substances are formed. It is only through experiment that a knowledge of what occurs can be discovered ─ either your experiment or the experiments of others (whi ...
... Writing a balanced chemical equation: In order to write a chemical equation, it is essential to know what substances are reacting and what substances are formed. It is only through experiment that a knowledge of what occurs can be discovered ─ either your experiment or the experiments of others (whi ...
chemistry - The Aga Khan University
... 13.3.3.2 Formation of Peroxides on Heating with Oxygen 13.3.3.3 Formation of Nitrides on Heating in Air 13.3.4 Trends in Solubility of the Hydroxides, Sulphates and Carbonates 13.3.5 Trends in Thermal Stability of the Nitrates and Carbonates 13.3.6 How Beryllium Differs from other Members of its Gro ...
... 13.3.3.2 Formation of Peroxides on Heating with Oxygen 13.3.3.3 Formation of Nitrides on Heating in Air 13.3.4 Trends in Solubility of the Hydroxides, Sulphates and Carbonates 13.3.5 Trends in Thermal Stability of the Nitrates and Carbonates 13.3.6 How Beryllium Differs from other Members of its Gro ...
x - mrs. leinweber`s wiki
... hydrogen. Evidence indicates that this reaction establishes an equilibrium with only partial conversion of reactants to products. Initially, 2.00 mol of each reactant is placed in the vessel. Kc for this reaction is 4.20 at 900C. Calculate the concentration of each substance at equilibrium. ...
... hydrogen. Evidence indicates that this reaction establishes an equilibrium with only partial conversion of reactants to products. Initially, 2.00 mol of each reactant is placed in the vessel. Kc for this reaction is 4.20 at 900C. Calculate the concentration of each substance at equilibrium. ...
13 CHEMICAL EQUILIBRIUM W MODULE - 5
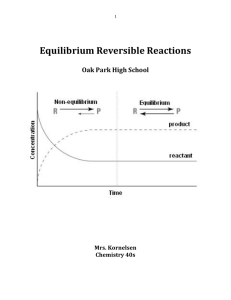
... When ethyl acetate and water are formed in the forward reaction the reverse reaction also starts in which ethanol and acetic acid are formed. After some time the concentrations of all the reactants and products become constant. This happens when the rates of forward and reverse reactions become equa ...
... When ethyl acetate and water are formed in the forward reaction the reverse reaction also starts in which ethanol and acetic acid are formed. After some time the concentrations of all the reactants and products become constant. This happens when the rates of forward and reverse reactions become equa ...
Chapter 3
... that you change the coefficient for a particular reactant or product, only to have to change it again later in the process. In general, it will facilitate the balancing process if you do the following: 1. Change the coefficients of compounds (e.g., CO2) before changing the coefficients of elements ( ...
... that you change the coefficient for a particular reactant or product, only to have to change it again later in the process. In general, it will facilitate the balancing process if you do the following: 1. Change the coefficients of compounds (e.g., CO2) before changing the coefficients of elements ( ...
AQA A-level Chemistry
... A reaction profile is a diagram of the enthalpy levels of the reactants and products in a chemical reaction. The vertical (y) axis is enthalpy but not ∆H. The horizontal (x) axis is progress of reaction, reaction coordinate or extent of reaction. Two horizontal lines are drawn and labelled with the ...
... A reaction profile is a diagram of the enthalpy levels of the reactants and products in a chemical reaction. The vertical (y) axis is enthalpy but not ∆H. The horizontal (x) axis is progress of reaction, reaction coordinate or extent of reaction. Two horizontal lines are drawn and labelled with the ...
Chemical Redox Agents for Organometallic
... in the absence of resistance effects) make CPE ideal for forcing the uptake or release of one or more electrons from a substrate. The literature of CPE is well-developed for organic, inorganic, and organometallic systems.5,6 When CPE is coupled with voltammetric methods such as cyclic voltammetry, p ...
... in the absence of resistance effects) make CPE ideal for forcing the uptake or release of one or more electrons from a substrate. The literature of CPE is well-developed for organic, inorganic, and organometallic systems.5,6 When CPE is coupled with voltammetric methods such as cyclic voltammetry, p ...
Chemical Redox Agents for Organometallic
... in the absence of resistance effects) make CPE ideal for forcing the uptake or release of one or more electrons from a substrate. The literature of CPE is well-developed for organic, inorganic, and organometallic systems.5,6 When CPE is coupled with voltammetric methods such as cyclic voltammetry, p ...
... in the absence of resistance effects) make CPE ideal for forcing the uptake or release of one or more electrons from a substrate. The literature of CPE is well-developed for organic, inorganic, and organometallic systems.5,6 When CPE is coupled with voltammetric methods such as cyclic voltammetry, p ...
The Proline Effect Revisited - Department of Chemistry, York University
... formed from a peptide that comprises residues containing only alkyl side chains, has an oxazolone structure. The b ion can further dissociate to produce the a ion, an iminium ion. The y ion has been proposed to be a protonated truncated peptide or amino acid;16-19 earlier MS/MS8 and MS/MS/MS10 exper ...
... formed from a peptide that comprises residues containing only alkyl side chains, has an oxazolone structure. The b ion can further dissociate to produce the a ion, an iminium ion. The y ion has been proposed to be a protonated truncated peptide or amino acid;16-19 earlier MS/MS8 and MS/MS/MS10 exper ...
Flotation behaviour and surface characteristics of the artificial
... (Zhang et al., 2011). Therefore, the upgrading of titanium slag is an urgent need in the titanium industry. Flotation can be implemented to upgrade titanium slag, as it has long served the mineral processing field as a mature technology (Bahri et al., 2016; Albrecht et al., 2016). The primary titani ...
... (Zhang et al., 2011). Therefore, the upgrading of titanium slag is an urgent need in the titanium industry. Flotation can be implemented to upgrade titanium slag, as it has long served the mineral processing field as a mature technology (Bahri et al., 2016; Albrecht et al., 2016). The primary titani ...
Electrochemistry
Electrochemistry is the branch of physical chemistry that studies chemical reactions which take place at the interface of an electrode, usually a solid metal or a semiconductor, and an ionic conductor, the electrolyte. These reactions involve electric charges moving between the electrodes and the electrolyte (or ionic species in a solution). Thus electrochemistry deals with the interaction between electrical energy and chemical change.When a chemical reaction is caused by an externally supplied current, as in electrolysis, or if an electric current is produced by a spontaneous chemical reaction as in a battery, it is called an electrochemical reaction. Chemical reactions where electrons are transferred directly between molecules and/or atoms are called oxidation-reduction or (redox) reactions. In general, electrochemistry describes the overall reactions when individual redox reactions are separate but connected by an external electric circuit and an intervening electrolyte.