enthalpy changes
... The initial temperature of both solutions was 20°C. The highest temperature reached by the solution was 33°C. Calculate the Molar Enthalpy of Neutralisation. [The specific heat capacity (c) of water is 4.18 kJ oC -1 kg -1] ...
... The initial temperature of both solutions was 20°C. The highest temperature reached by the solution was 33°C. Calculate the Molar Enthalpy of Neutralisation. [The specific heat capacity (c) of water is 4.18 kJ oC -1 kg -1] ...
Chemical Reactivity and Biological Activity of Diketene
... The alkylating potential of diketene (4-methylene-2-oxetanone), the basic unit of many derivatives of pesticides, chemicals, pharmaceuticals, and dyestuffs, was investigated kinetically. The nucleophile 4-(pnitrobenzyl)pyridine (NBP), a trap for alkylating agents with nucleophilic characteristics si ...
... The alkylating potential of diketene (4-methylene-2-oxetanone), the basic unit of many derivatives of pesticides, chemicals, pharmaceuticals, and dyestuffs, was investigated kinetically. The nucleophile 4-(pnitrobenzyl)pyridine (NBP), a trap for alkylating agents with nucleophilic characteristics si ...
Ch16 - WordPress.com
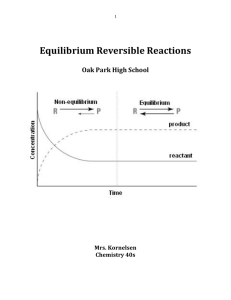
... An equilibrium mixture consists of the gases N2O4 and NO2: N2O4(g) 2NO2(g) The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: ...
... An equilibrium mixture consists of the gases N2O4 and NO2: N2O4(g) 2NO2(g) The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: ...
BSC HS CHEMISTRY SEMESTER I to VI CBCEGS
... a) Developing Habits of Independent and Fast Reading. Students will be required to read a prescribed prose anthology titled Selections from Modern English Prose (Ed. Haladhar Panda published by University Press, Hyderabad). The essays in the anthology will be read by students at home with the help o ...
... a) Developing Habits of Independent and Fast Reading. Students will be required to read a prescribed prose anthology titled Selections from Modern English Prose (Ed. Haladhar Panda published by University Press, Hyderabad). The essays in the anthology will be read by students at home with the help o ...
Ch16
... An equilibrium mixture consists of the gases N2O4 and NO2: N2O4(g) 2NO2(g) The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: ...
... An equilibrium mixture consists of the gases N2O4 and NO2: N2O4(g) 2NO2(g) The volume of the container was increased at constant temperature and a new equilbrium was established. Predict how each of the following quantities would change at the new equilibrium compared with the initial equilibrium: ...
Exam #3
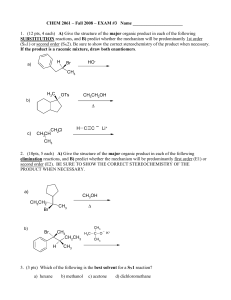
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
... (12 pts) Bromoetherification, the addition of the elements Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. Draw a stepwise mechanism for the following INTRAMOLECULAR bromoetherification reaction. Hint: the mechanism is analogous to that of bromohydrin f ...
Question Bank - Edudel.nic.in
... When 1 mole of NaCl is added to 1 litre water the boiling point increases. When 1 mole of CH3OH is added to 1 litre water, the boiling point decreases. Suggest reason. Can we separate water completely from HNO3 solution. Justify your answer. ...
... When 1 mole of NaCl is added to 1 litre water the boiling point increases. When 1 mole of CH3OH is added to 1 litre water, the boiling point decreases. Suggest reason. Can we separate water completely from HNO3 solution. Justify your answer. ...
Chapter 6
... The sign of ∆G indicates the direction in which a reaction moves to reach its equilibrium position. A reaction is thermodynamically favorable when its enthalpy, ∆H, decreases and its entropy, ∆S, increases. Substitut‑ ing the inequalities ∆H < 0 and ∆S > 0 into equation 6.2 shows that a reaction is ...
... The sign of ∆G indicates the direction in which a reaction moves to reach its equilibrium position. A reaction is thermodynamically favorable when its enthalpy, ∆H, decreases and its entropy, ∆S, increases. Substitut‑ ing the inequalities ∆H < 0 and ∆S > 0 into equation 6.2 shows that a reaction is ...
1 Bite Angle Effects of Diphosphines in Carbonylation Reactions
... systematic studies using diphosphines was by Unruh [23] who used substituted dppf. Both rate and selectivity increase when the w-value of the ligands increase. There are two possible reasons: electrons preference for linear alkyl complex formation when the p-back-donation to the phosphine increases ...
... systematic studies using diphosphines was by Unruh [23] who used substituted dppf. Both rate and selectivity increase when the w-value of the ligands increase. There are two possible reasons: electrons preference for linear alkyl complex formation when the p-back-donation to the phosphine increases ...