Heat Work
... STP (point a). Points b and c are on an isothermal. If the engine produces positive work, a) is the cycle clockwise or counter clockwise, b) what is the efficiency of the cycle? ...
... STP (point a). Points b and c are on an isothermal. If the engine produces positive work, a) is the cycle clockwise or counter clockwise, b) what is the efficiency of the cycle? ...
Ch.19 (section 1 only)
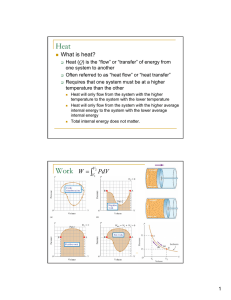
... Device that uses heat to perform work Hot Reservoir (e.g. steam) Cool Reservoir (e.g. pool of water) Efficiency is work done per unit of input heat (e = W/QH) • Ex. A heat engine does 100J of work when given 300J from the hot reservoir. The efficiency is 100J/300J = 0.33 = ...
... Device that uses heat to perform work Hot Reservoir (e.g. steam) Cool Reservoir (e.g. pool of water) Efficiency is work done per unit of input heat (e = W/QH) • Ex. A heat engine does 100J of work when given 300J from the hot reservoir. The efficiency is 100J/300J = 0.33 = ...
Teacher`s notes 22 Specific Heat Capacity of a solid
... this reason it is best to purchase a block and heater that are matched. Ideally several blocks are required, aluminium, brass and iron. Glycerol is mentioned as the heat conductive medium to surround the heater this reduces the risk or fire. Oil with a very high boiling point and flash point can be ...
... this reason it is best to purchase a block and heater that are matched. Ideally several blocks are required, aluminium, brass and iron. Glycerol is mentioned as the heat conductive medium to surround the heater this reduces the risk or fire. Oil with a very high boiling point and flash point can be ...
Heat Transfer - cloudfront.net
... • Explanation: Sun is heating up the ground more quickly than it heats the air, especially if the surface of the ground is a dark color. The heated air rises and bends light waves as it passes through them. Making the objects on the other side shimmer. ...
... • Explanation: Sun is heating up the ground more quickly than it heats the air, especially if the surface of the ground is a dark color. The heated air rises and bends light waves as it passes through them. Making the objects on the other side shimmer. ...
EML 6154 - UFL MAE - University of Florida
... to understand advanced heat transfer solution techniques coupled with a strong foundation and appreciation for the physics and mathematics of conduction heat transfer. Micro-scale heat transfer, including energy carriers, carrier length scales, and micro-scale heat transfer regimes is also covered a ...
... to understand advanced heat transfer solution techniques coupled with a strong foundation and appreciation for the physics and mathematics of conduction heat transfer. Micro-scale heat transfer, including energy carriers, carrier length scales, and micro-scale heat transfer regimes is also covered a ...
Conceptual Physics. Tenth Edition
... is 37 C. When the surrounding of a person is colder than 37 C, heat transfer from the body to it’s surrounding will take place and will not stop until the body and it’s surrounding have a common temperature (thermal equilibrium) See figure 2. (Hewitt, 2007): “Whenever heat flows into or out of a sys ...
... is 37 C. When the surrounding of a person is colder than 37 C, heat transfer from the body to it’s surrounding will take place and will not stop until the body and it’s surrounding have a common temperature (thermal equilibrium) See figure 2. (Hewitt, 2007): “Whenever heat flows into or out of a sys ...
Thermodynamics-d2
... If 200cm3 of tea (c=4186J/kg°C) at 95°C is poured into a 150-g glass cup (c=840J/kg°C) initially at 25°C, what will be the final temperature T of the mixture when equilibrium is reached, assuming no heat flows to the surroundings? ...
... If 200cm3 of tea (c=4186J/kg°C) at 95°C is poured into a 150-g glass cup (c=840J/kg°C) initially at 25°C, what will be the final temperature T of the mixture when equilibrium is reached, assuming no heat flows to the surroundings? ...
HEAT ENERGY
... HOT it is Measured in degrees Celsius - °C It is NOT the same as heat energy although the two quantities are related. e.g. a beaker of water at 60 °C is hotter than a bath of water at 40 °C BUT the bath contains more joules of heat energy ...
... HOT it is Measured in degrees Celsius - °C It is NOT the same as heat energy although the two quantities are related. e.g. a beaker of water at 60 °C is hotter than a bath of water at 40 °C BUT the bath contains more joules of heat energy ...
Tutorial 3
... thick plastic cover whose thermal conductivity is k = 0.15 W/m · °C. Electrical measurements indicate that a current of 10 A passes through the wire and there is a voltage drop of 8 V along the wire. If the insulated wire is exposed to a medium at T = 30°C with a heat transfer coefficient of h = 12 ...
... thick plastic cover whose thermal conductivity is k = 0.15 W/m · °C. Electrical measurements indicate that a current of 10 A passes through the wire and there is a voltage drop of 8 V along the wire. If the insulated wire is exposed to a medium at T = 30°C with a heat transfer coefficient of h = 12 ...
Condensation and the Nusselt`s Film Theory
... Condensation occurs if a vapor is cooled below its (pressure dependent) saturation temperature. The heat of evaporation which is released during condensation must be removed by heat transfer, e.g. at a cooled wall. Figure 1 shows how saturated vapor at temperature Ts is condensing on a vertical wall ...
... Condensation occurs if a vapor is cooled below its (pressure dependent) saturation temperature. The heat of evaporation which is released during condensation must be removed by heat transfer, e.g. at a cooled wall. Figure 1 shows how saturated vapor at temperature Ts is condensing on a vertical wall ...
Water is able to absorb a high amount of heat before
... because many organisms are mainly composed of water, the property of high heat capacity allows highly regulated internal body temperatures. For example, the temperature of your body does not drastically drop to the same temperature as the outside temperature while you are skiing or playing in the sn ...
... because many organisms are mainly composed of water, the property of high heat capacity allows highly regulated internal body temperatures. For example, the temperature of your body does not drastically drop to the same temperature as the outside temperature while you are skiing or playing in the sn ...
Heat sink

A heat sink is a passive heat exchanger that transfers the heat generated by an electronic or a mechanical device into a coolant fluid in motion. Then-transferred heat leaves the device with the fluid in motion, therefore allowing the regulation of the device temperature at physically feasible levels. In computers, heat sinks are used to cool central processing units or graphics processors. Heat sinks are used with high-power semiconductor devices such as power transistors and optoelectronics such as lasers and light emitting diodes (LEDs), where the heat dissipation ability of the basic device is insufficient to moderate its temperature.A heat sink is designed to maximize its surface area in contact with the cooling medium surrounding it, such as the air. Air velocity, choice of material, protrusion design and surface treatment are factors that affect the performance of a heat sink. Heat sink attachment methods and thermal interface materials also affect the die temperature of the integrated circuit. Thermal adhesive or thermal grease improve the heat sink's performance by filling air gaps between the heat sink and the heat spreader on the device.