* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Specific Heat
Insulated glazing wikipedia , lookup
Passive solar building design wikipedia , lookup
Thermal conductivity wikipedia , lookup
Radiator (engine cooling) wikipedia , lookup
Vapor-compression refrigeration wikipedia , lookup
Hypothermia wikipedia , lookup
Space Shuttle thermal protection system wikipedia , lookup
Dynamic insulation wikipedia , lookup
Building insulation materials wikipedia , lookup
Thermoregulation wikipedia , lookup
Solar air conditioning wikipedia , lookup
Solar water heating wikipedia , lookup
Heat exchanger wikipedia , lookup
Intercooler wikipedia , lookup
Heat equation wikipedia , lookup
R-value (insulation) wikipedia , lookup
Cogeneration wikipedia , lookup
Copper in heat exchangers wikipedia , lookup
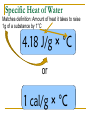
Thermochemistry Specific Heat Specific Heat Amount of heat it takes to raise 1g of How easy or hard an object a substance by 1°C is to heat up – how fast does it absorb heat energy Metals have a low specific heat Water has a high specific heat Learning Check 1. On a sunny day, the concrete deck around an outdoor swimming pool becomes hot, while the water stays cool. This is because… A. The deck has a higher specific heat than the water B. The deck has a lower specific heat then the water C. Both objects have the same specific heat Learning Check 2. Two objects are sitting next to each other in the sunlight. Object A gets hotter than object B. A. Object A has a lower specific heat than object B B. Object A has a higher specific heat than object B C. Both objects have the same specific heat Specific Heat of Water Matches definition: Amount of heat it takes to raise 1g of a substance by 1°C 4.18 J/g × °C or 1 cal/g × °C Formula for Specific Heat q = mc ΔT q = amount of heat – measured in cal or J m = mass – measured in g C = specific heat – units are J/g x °C or cal/g x °C ΔT = change in temp – measured in °C Specific Heat Problems: 1. Aluminum has a specific heat of 0.9020 J/g × °C. How much heat is lost when a piece of aluminum with a mass of 23.984 g cools from a temperature of 415.0°C to a temperature of 22.0°C? 2. 850 calories of heat are applied to a 250 g sample of liquid water with an initial temperature of 13.0 °C. Find: a) the change in temperature and b) the final temperature. (remember, the specific heat of liquid water, in calories, is 1.00 cal/g x °C.) Specific Heat Problems: 3. The temperature of a sample of water increases by 69.5 °C when 24,500 J are applied. The specific heat of liquid water is 4.18 J/g x °C. What is the mass of the sample of water? 4. When 34,700 J of heat are applied to a 350 g sample of an unknown material the temperature rises from 22.0°C to 173.0°C. What must be the specific heat of this material?