qp2
... Planck by introducing a very important concept… quantization of energy. Planck said that black bodies contained microscopic oscillators that absorbed and emitted radiation. He also specified that oscillators of frequency (f) could only absorb and emit waves of energy at the same frequency. It was no ...
... Planck by introducing a very important concept… quantization of energy. Planck said that black bodies contained microscopic oscillators that absorbed and emitted radiation. He also specified that oscillators of frequency (f) could only absorb and emit waves of energy at the same frequency. It was no ...
Lecture6.QM.to.Lagrangian.Densities
... Just as there is no “derivation” of quantum mechanics from classical mechanics, there is no derivation of relativistic field theory from quantum mechanics. The “route” from one to the other is based on physically reasonable postulates and the imposition of Lorentz invariance and relativistic kinemat ...
... Just as there is no “derivation” of quantum mechanics from classical mechanics, there is no derivation of relativistic field theory from quantum mechanics. The “route” from one to the other is based on physically reasonable postulates and the imposition of Lorentz invariance and relativistic kinemat ...
wave function
... The photoelectric effect occurs when light incident on certain metallic surfaces causes electrons to be emitted from those surfaces Einstein extended Planck’s concept of quantization to electromagnetic waves All electromagnetic radiation can be considered a stream of quanta, now called photons A pho ...
... The photoelectric effect occurs when light incident on certain metallic surfaces causes electrons to be emitted from those surfaces Einstein extended Planck’s concept of quantization to electromagnetic waves All electromagnetic radiation can be considered a stream of quanta, now called photons A pho ...
Electromagnetic Waves
... An applet that displays the relative orientation of E, B and the direction of propagation of the wave can be found at this link. Use Firefox, select the applet “Electromagnetic Waves” (the forth from the top), download it and run it. E and B are perpendicular to each other and perpendicular to the d ...
... An applet that displays the relative orientation of E, B and the direction of propagation of the wave can be found at this link. Use Firefox, select the applet “Electromagnetic Waves” (the forth from the top), download it and run it. E and B are perpendicular to each other and perpendicular to the d ...
Quantum Mechanical Model of the Atom
... Distinguishes the shape of the orbitals • Magnetic Quantum Number (ml) distinguishes orientation in space • Spin Quantum Number (ms) gives the 2 possible locations of the spin ...
... Distinguishes the shape of the orbitals • Magnetic Quantum Number (ml) distinguishes orientation in space • Spin Quantum Number (ms) gives the 2 possible locations of the spin ...
CH4 PT1 Arrangement of Electrons
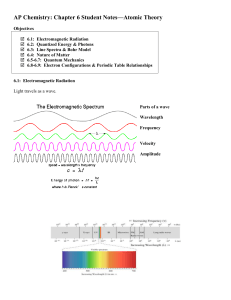
... point in a given amount of time. Units: Hertz, Hz or 1/s or s-1 ...
... point in a given amount of time. Units: Hertz, Hz or 1/s or s-1 ...
Introduction to Quantum Mechanic
... 1) Black-body radiation (1860-1901) 2) Atomic Spectroscopy (1888-) 3) Photoelectric Effect (1887-1905) C) Wave–particle duality 1) Compton Effect (1923). 2) Electron Diffraction Davisson and Germer (1925). 3) Young's Double Slit Experiment D) Louis de Broglie relation for a photon from relativity E) ...
... 1) Black-body radiation (1860-1901) 2) Atomic Spectroscopy (1888-) 3) Photoelectric Effect (1887-1905) C) Wave–particle duality 1) Compton Effect (1923). 2) Electron Diffraction Davisson and Germer (1925). 3) Young's Double Slit Experiment D) Louis de Broglie relation for a photon from relativity E) ...
Ch. 6 notes
... 6.5-6.7: Quantum Mechanics Developed by Werner Heisenberg (1901-1976), Louis De Broglie (1892-1987), Erwin Schrodinger (1887-1961) This answers the question: Where is the _____________ in the atom? The answer is complex. We can’t say exactly where the atom is. We can only say where we think it _____ ...
... 6.5-6.7: Quantum Mechanics Developed by Werner Heisenberg (1901-1976), Louis De Broglie (1892-1987), Erwin Schrodinger (1887-1961) This answers the question: Where is the _____________ in the atom? The answer is complex. We can’t say exactly where the atom is. We can only say where we think it _____ ...
Chapter 8 - Fayetteville State University
... B. is exclusively a particle phenomenon. C. in any particular event, exhibits either a wave nature or a particle nature, never both at the same time. D. has neither wave nor particle properties. 9. The de Broglie wavelength of an object A. is equal to Planck’s constant divided by the momentum of the ...
... B. is exclusively a particle phenomenon. C. in any particular event, exhibits either a wave nature or a particle nature, never both at the same time. D. has neither wave nor particle properties. 9. The de Broglie wavelength of an object A. is equal to Planck’s constant divided by the momentum of the ...
Chapter 27
... Particles had wave characteristics just like waves had particle characteristics (e.g. E&M wave photon) The de Broglie wavelength of a particle of mass, m, is ...
... Particles had wave characteristics just like waves had particle characteristics (e.g. E&M wave photon) The de Broglie wavelength of a particle of mass, m, is ...
Old Physics GRE Problems Based on content from Chapter 2 of your
... C. The charge of X is carried on its surface. D. X does not spin. E. X cannot be detected. 5. The measured index of refraction of x-rays in rock salt is less than one. This is consistent with the theory of relativity because A. relativity deals with light waves traveling in a vacuum only. B. x-rays ...
... C. The charge of X is carried on its surface. D. X does not spin. E. X cannot be detected. 5. The measured index of refraction of x-rays in rock salt is less than one. This is consistent with the theory of relativity because A. relativity deals with light waves traveling in a vacuum only. B. x-rays ...