Chapter 3 Chemical Compounds
... molecules, which typically consist of two or more of nonmetal atoms held together by covalent bonds. A covalent bond, the most common kind of chemical bond, results when two atoms share electrons. Even some elements exit as molecules rather than as atoms. Hydrogen, nitrogen, oxygen, fluorine, chlori ...
... molecules, which typically consist of two or more of nonmetal atoms held together by covalent bonds. A covalent bond, the most common kind of chemical bond, results when two atoms share electrons. Even some elements exit as molecules rather than as atoms. Hydrogen, nitrogen, oxygen, fluorine, chlori ...
Unit 4 - Dorman High School
... V. Ionic Bonding and Structures of Ionic Compounds When an ionic compound is formed the bond is extremely strong. We write the formulas for these compounds, but they are empirical formulas because the compound is composed of a very tightly packed and ordered arrangement of ions. Ionic compounds can ...
... V. Ionic Bonding and Structures of Ionic Compounds When an ionic compound is formed the bond is extremely strong. We write the formulas for these compounds, but they are empirical formulas because the compound is composed of a very tightly packed and ordered arrangement of ions. Ionic compounds can ...
Chemical Equilbrium
... assumption, many times you will end up with a quadratic equation for an equilibrium constant expression. You can end up with a 3rd, 4th, 5th, etc. order polynomial, but I will not hold you responsible for being able to solve those as there is no simple formula for the solution. ...
... assumption, many times you will end up with a quadratic equation for an equilibrium constant expression. You can end up with a 3rd, 4th, 5th, etc. order polynomial, but I will not hold you responsible for being able to solve those as there is no simple formula for the solution. ...
Slide 1 - Herricks
... 4. Balance the elements one at a time by using coefficients. When no coefficient is written, it is assumed to be 1. Begin by balancing elements that appear only once on each side of the equation. Never balance an equation by changing the subscripts in a chemical formula. Each substance has only one ...
... 4. Balance the elements one at a time by using coefficients. When no coefficient is written, it is assumed to be 1. Begin by balancing elements that appear only once on each side of the equation. Never balance an equation by changing the subscripts in a chemical formula. Each substance has only one ...
Solutions - cloudfront.net
... In addition to dipole-dipole interactions, now you can have Hbonding. These additional interactions should make the interactions between CHCl3 and CH3COCH3 stronger than the self-self interactions. Note, normally H-bonds require that the H be bonded to an electronegative atom. C is not more electron ...
... In addition to dipole-dipole interactions, now you can have Hbonding. These additional interactions should make the interactions between CHCl3 and CH3COCH3 stronger than the self-self interactions. Note, normally H-bonds require that the H be bonded to an electronegative atom. C is not more electron ...
1. This is a question about trends in chemistry In
... take place in a single reactive encounter between the two molecules of the butadiene. a) ...
... take place in a single reactive encounter between the two molecules of the butadiene. a) ...
Final Exam Study Guide Page 1 Quiz
... a. Is completely used up in the reaction b. Will have some amount unchanged, or leftover, after the reaction c. Cannot be calculated without performing the reaction d. Has no effect in the amount of product formed ...
... a. Is completely used up in the reaction b. Will have some amount unchanged, or leftover, after the reaction c. Cannot be calculated without performing the reaction d. Has no effect in the amount of product formed ...
Slide 1
... of atom present Step 3: Divide the number of mole of each element by the smallest number of moles to convert the smallest number to 1. If all of the numbers so obtained are integers, these are the subscripts in the empirical formula. If one or more of these number are not integers, go on to step 4 ...
... of atom present Step 3: Divide the number of mole of each element by the smallest number of moles to convert the smallest number to 1. If all of the numbers so obtained are integers, these are the subscripts in the empirical formula. If one or more of these number are not integers, go on to step 4 ...
Chapter 12 Chemical Quantities
... Stoichiometry is the study of quantitative relationships between amounts of reactants used and products formed by a chemical reaction. ...
... Stoichiometry is the study of quantitative relationships between amounts of reactants used and products formed by a chemical reaction. ...
objectives chm 1025 - Miami Dade College
... a. Showing how to obtain an element’s average atomic mass and atomic number from the periodic table. b. Using the structure of the periodic table to classify elements (e.g., metal, nonmetal, metalloid, noble gas, representative element, transition element, inner transition element, alkali metal, alk ...
... a. Showing how to obtain an element’s average atomic mass and atomic number from the periodic table. b. Using the structure of the periodic table to classify elements (e.g., metal, nonmetal, metalloid, noble gas, representative element, transition element, inner transition element, alkali metal, alk ...
Review of Moles and Stoichiometry
... 15.) A compound was analyzed in a lab to determine its empirical formula. Decomposition of the compound at standard temperature and pressure produced 9.00 g carbon, 16.8 L hydrogen, and 2.80 L oxygen. a.) What is the empirical formula for this compound? ...
... 15.) A compound was analyzed in a lab to determine its empirical formula. Decomposition of the compound at standard temperature and pressure produced 9.00 g carbon, 16.8 L hydrogen, and 2.80 L oxygen. a.) What is the empirical formula for this compound? ...
Biomaterials Based on Polymers, Fibers, and Textiles
... ionic bonds = electrostatic attractions between the positive and negative charges of the ions. In aqueous solutions, all cations and anions are surrounded by a shell of water molecules bound . Increasing the salt (eg NaCl ) concentration weakens the ionic bonds . Binding energy about 787 kJ / mol ( ...
... ionic bonds = electrostatic attractions between the positive and negative charges of the ions. In aqueous solutions, all cations and anions are surrounded by a shell of water molecules bound . Increasing the salt (eg NaCl ) concentration weakens the ionic bonds . Binding energy about 787 kJ / mol ( ...
Chapter 4 The Structure of Matter
... • b. usually low melting points except for compounds that form network structures like SiO2 ...
... • b. usually low melting points except for compounds that form network structures like SiO2 ...
For H 2 O
... and carbon dioxide Incomplete combustion produces CO and C in addition to CO2 and H2O due to decreased amounts of O2 ...
... and carbon dioxide Incomplete combustion produces CO and C in addition to CO2 and H2O due to decreased amounts of O2 ...
Nomenclature and chemical reactions PPT
... and carbon dioxide Incomplete combustion produces CO and C in addition to CO2 and H2O due to decreased amounts of O2 ...
... and carbon dioxide Incomplete combustion produces CO and C in addition to CO2 and H2O due to decreased amounts of O2 ...
Summary from Organic Chemistry Packet:
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
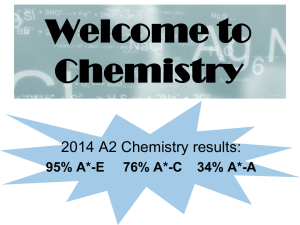
Welcome to Chemistry
... AS can be sat as a stand alone qualification over 1 year, exams are sat at the end of Y12. 2 written exams each 1 hour and 30 minutes. A level is the full 2 year qualification with all the exams at the end of Y13. 3 written papers each 2 hours. ...
... AS can be sat as a stand alone qualification over 1 year, exams are sat at the end of Y12. 2 written exams each 1 hour and 30 minutes. A level is the full 2 year qualification with all the exams at the end of Y13. 3 written papers each 2 hours. ...
Balancing Chemical Equation Practice.docx
... Reading adapted from Sarquis’s Modern Chemistry Introduction A chemical reaction is the process by which one or more substances are changed into one or more different substances. In any chemical reaction, the original substances are known as the reactants, and the resulting substances are known as t ...
... Reading adapted from Sarquis’s Modern Chemistry Introduction A chemical reaction is the process by which one or more substances are changed into one or more different substances. In any chemical reaction, the original substances are known as the reactants, and the resulting substances are known as t ...
Curriculum Plan
... relationships and conversions Understand and agree to lab safety rules; identify and know how to use safety equipment Understand: Definitions, the Laws of Conservation of Energy and of Conservation of Matter, Temperature scales and conversions, Chemical and physical changes, Classifying Matter, Chem ...
... relationships and conversions Understand and agree to lab safety rules; identify and know how to use safety equipment Understand: Definitions, the Laws of Conservation of Energy and of Conservation of Matter, Temperature scales and conversions, Chemical and physical changes, Classifying Matter, Chem ...
Weiguang Zhang 1,* Yun Zhong 1, Minyu Tan 2,*, Ning Tang 2 and
... In general, coordination of a ligand to a metal ion causes an increase in the electron density on the metal, resulting in a decrease in the metal ion’s electron binding energy [12]. The Zn(2p)3/2 chemical shift, which are the differences between the Zn(2p)3/2 binding energies of the complex and ZnCl ...
... In general, coordination of a ligand to a metal ion causes an increase in the electron density on the metal, resulting in a decrease in the metal ion’s electron binding energy [12]. The Zn(2p)3/2 chemical shift, which are the differences between the Zn(2p)3/2 binding energies of the complex and ZnCl ...
zinc(II): Zn 2 [(n
... been assigned to the carbon in the alkyl and the latter to the CSS. Its N(1s) spectrum showed a single full-width peak, but the S(2p) spectrum showed a broad peak with a shoulder suggesting two types of sulfur atoms. The two peaks indicated by the broken lines, which were obtained by a graphical res ...
... been assigned to the carbon in the alkyl and the latter to the CSS. Its N(1s) spectrum showed a single full-width peak, but the S(2p) spectrum showed a broad peak with a shoulder suggesting two types of sulfur atoms. The two peaks indicated by the broken lines, which were obtained by a graphical res ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.