Chapter 8
... Determine the Empirical Formula of Acetic Anhydride if its Percent Composition is 47% Carbon, 47% Oxygen and 6.0% Hydrogen 4. If any of the ratios is not a whole number, multiply all the ratios by a factor to make it a whole number – If ratio is ?.5 then multiply by 2; if ?.33 or ?.67 then multiply ...
... Determine the Empirical Formula of Acetic Anhydride if its Percent Composition is 47% Carbon, 47% Oxygen and 6.0% Hydrogen 4. If any of the ratios is not a whole number, multiply all the ratios by a factor to make it a whole number – If ratio is ?.5 then multiply by 2; if ?.33 or ?.67 then multiply ...
Alcohol responsive 2D coordination network of 3
... One of the most important objectives in the field of coordination networks or metal organic frameworks is the engineering of porous structures with a well defined chemical environment [1–2]. This is however very difficult to achieve in 3D structures due to catenation and interpenetration. Thus in re ...
... One of the most important objectives in the field of coordination networks or metal organic frameworks is the engineering of porous structures with a well defined chemical environment [1–2]. This is however very difficult to achieve in 3D structures due to catenation and interpenetration. Thus in re ...
Chapter 8
... • The molar mass is the mass in grams of one mole of a compound • The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu • 1 mole of H2O will weigh 18.02 g, therefore the molar mass of H2O is 18.02 g • 1 mole of H2O will contain 16.0 ...
... • The molar mass is the mass in grams of one mole of a compound • The relative weights of molecules can be calculated from atomic masses water = H2O = 2(1.008 amu) + 16.00 amu = 18.02 amu • 1 mole of H2O will weigh 18.02 g, therefore the molar mass of H2O is 18.02 g • 1 mole of H2O will contain 16.0 ...
Chapter 3 Molecules, Compounds, and Chemical Equations
... Chapter 3 Molecules, Compounds, and Chemical Equations ...
... Chapter 3 Molecules, Compounds, and Chemical Equations ...
Chapter 07 Notes - Mr. Julien`s Homepage
... Read Section 7.5 in your eText! I will determine the molecular formula of a substance from the empirical formula and molar mass. Do the Empirical and Molecular Formulas Tutorial and the Formula Mass Tutorial! A. Relating empirical and molecular formulas. 1. The molecular formula is the product of a ...
... Read Section 7.5 in your eText! I will determine the molecular formula of a substance from the empirical formula and molar mass. Do the Empirical and Molecular Formulas Tutorial and the Formula Mass Tutorial! A. Relating empirical and molecular formulas. 1. The molecular formula is the product of a ...
Ch3pdf.
... 2. use coefficients to indicate how many formula units are required to balance equation 3. balance those species that occur in the fewest formulas on each side. 4. reduce coefficients to smallest whole number values 5. when balancing reactions involving organic compounds, balance in the order: C, H, ...
... 2. use coefficients to indicate how many formula units are required to balance equation 3. balance those species that occur in the fewest formulas on each side. 4. reduce coefficients to smallest whole number values 5. when balancing reactions involving organic compounds, balance in the order: C, H, ...
AP Chemistry Summer Assignment
... 20. The hormone, thyroxine is secreted by the thyroid gland, and has the formula: C15H17NO4I4. How many milligrams of iodine can be extracted from 15.0 grams of thyroxine? ...
... 20. The hormone, thyroxine is secreted by the thyroid gland, and has the formula: C15H17NO4I4. How many milligrams of iodine can be extracted from 15.0 grams of thyroxine? ...
Calculating a substance`s gram formula mass
... Gay-Lussac (whose work we will study later this year). Avogadro’s Law states that, so long as temperature and pressure are equal, an equal volume of any gas will contain the same number of molecules. Avogadro's Law was ignored until 1858, when Stanislao Cannizaro proved it experimentally by using it ...
... Gay-Lussac (whose work we will study later this year). Avogadro’s Law states that, so long as temperature and pressure are equal, an equal volume of any gas will contain the same number of molecules. Avogadro's Law was ignored until 1858, when Stanislao Cannizaro proved it experimentally by using it ...
Name_______________________________________________
... c. An atom of any element is a unit that can stand on its own. d. The strength of attraction between charged particles depends on the distance between the atoms involved in the bond. 3. Noble-gas atoms are able to exist independently in nature because a. they are exceptions to the octet rule. b. the ...
... c. An atom of any element is a unit that can stand on its own. d. The strength of attraction between charged particles depends on the distance between the atoms involved in the bond. 3. Noble-gas atoms are able to exist independently in nature because a. they are exceptions to the octet rule. b. the ...
File - Meissnerscience.com
... a water solution and has _____ molecules incorporated into their crystal structure. Hydrates have a ________________ of water molecules ___________ bonded to each _________________. Compounds that have no water molecules incorporated into them are called _________________ to distinguish them from th ...
... a water solution and has _____ molecules incorporated into their crystal structure. Hydrates have a ________________ of water molecules ___________ bonded to each _________________. Compounds that have no water molecules incorporated into them are called _________________ to distinguish them from th ...
Chemical Formulas and Chemical Compounds
... 7. The oxidation numbers assigned to the atoms in some organic compounds have unexpected values. Assign oxidation numbers to each atom in the following compounds: (Note: Some oxidation numbers may not be whole numbers.) a. CO2 Carbon is ⫹4 and each oxygen is ⫺2. b. CH4 (methane) Carbon is ⫺4 and eac ...
... 7. The oxidation numbers assigned to the atoms in some organic compounds have unexpected values. Assign oxidation numbers to each atom in the following compounds: (Note: Some oxidation numbers may not be whole numbers.) a. CO2 Carbon is ⫹4 and each oxygen is ⫺2. b. CH4 (methane) Carbon is ⫺4 and eac ...
Midterm 2 from Summer 2012
... Hydrazine is an extremely combustible compound composed of Nitrogen and Hydrogen that is used as a fuel for ICBM's. This compound has a composition of 87.42% Nitrogen and 12.58% Hydrogen. What is the Empirical Formula of ...
... Hydrazine is an extremely combustible compound composed of Nitrogen and Hydrogen that is used as a fuel for ICBM's. This compound has a composition of 87.42% Nitrogen and 12.58% Hydrogen. What is the Empirical Formula of ...
6. NaF
... molecule of chlorine trifluoride, ClF3 contains 1 atom of chlorine and 3 atoms of fluorine. Rule 1. The element with the lower group number is written first in the name; the element with the higher group number is written second in the name. Rule 2. If both elements are in the same group, the elemen ...
... molecule of chlorine trifluoride, ClF3 contains 1 atom of chlorine and 3 atoms of fluorine. Rule 1. The element with the lower group number is written first in the name; the element with the higher group number is written second in the name. Rule 2. If both elements are in the same group, the elemen ...
Search for the Electron Electric Dipole Moment Using PbO
... • electronically excited molecules decay to hot free atoms or to ground-state molecules • Production of polar molecules requires assembly from two different atomic species • molecules can be formed in single rotational state, at translational temperature of atoms (100 K routine, 1 K possible) BUT ...
... • electronically excited molecules decay to hot free atoms or to ground-state molecules • Production of polar molecules requires assembly from two different atomic species • molecules can be formed in single rotational state, at translational temperature of atoms (100 K routine, 1 K possible) BUT ...
(chemical reaction).
... unit of the compound. The molecular weight of a substance is the sum of the atomic weights of all the atoms in a molecule of the substance. Note: the formula weight is applicable to both molecular and ionic compounds. ...
... unit of the compound. The molecular weight of a substance is the sum of the atomic weights of all the atoms in a molecule of the substance. Note: the formula weight is applicable to both molecular and ionic compounds. ...
Stoichiometry 2
... Step 3. Fill in the ladder grid with as much information as possible – this is typically supplied gram weights and molar mass data. Step 4. Convert g moles by ‘climbing’ down ladder(s) (g / M = moles). Remember: Molar masses are calculated for ONE molecular formula only. I.E. ignore any balancing ...
... Step 3. Fill in the ladder grid with as much information as possible – this is typically supplied gram weights and molar mass data. Step 4. Convert g moles by ‘climbing’ down ladder(s) (g / M = moles). Remember: Molar masses are calculated for ONE molecular formula only. I.E. ignore any balancing ...
Gen Chem--Chapter 3 lecture notes.ppt (Read
... ignored when writing organic formulas (molecules containing carbon, hydrogen, and maybe other elements) in order to give a better idea of how the atoms are connected: C2H6O is the molecular formula for ethanol, but nobody ever writes it this way—instead the formula is written C2H5OH to indicate one ...
... ignored when writing organic formulas (molecules containing carbon, hydrogen, and maybe other elements) in order to give a better idea of how the atoms are connected: C2H6O is the molecular formula for ethanol, but nobody ever writes it this way—instead the formula is written C2H5OH to indicate one ...
Stoichiometry …like a beautiful sunset on a serene lake – NOT!
... and 2 jars of peanut butter. • During the sandwich-making process (the “reaction”) which “reactant” will you run out of first (bread, jelly, or pb?) • The “reactant” that you run out of 1st will stop the sandwich-making process. • And, 300 boy scouts are going to tie you up in the knots they just le ...
... and 2 jars of peanut butter. • During the sandwich-making process (the “reaction”) which “reactant” will you run out of first (bread, jelly, or pb?) • The “reactant” that you run out of 1st will stop the sandwich-making process. • And, 300 boy scouts are going to tie you up in the knots they just le ...
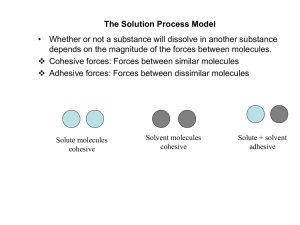
The Solution Process Model
... If Ct is very small (Ct >0.1 Cs), then dissolution rate is directly proportional to the saturation solubility (Cs). This assumes that S is constant which is only true at the very beginning of the experiment when only a small amount (>0.5%) of the solid drug has dissolved. When these two requirements ...
... If Ct is very small (Ct >0.1 Cs), then dissolution rate is directly proportional to the saturation solubility (Cs). This assumes that S is constant which is only true at the very beginning of the experiment when only a small amount (>0.5%) of the solid drug has dissolved. When these two requirements ...
2014
... PCl5 (g) ⇆ PCl3 (g) + Cl2 (g) PCl5 (g) decomposes into PCl3 (g) and Cl2 (g) according to the equation above. A sample of PCl5 (g) is placed into a rigid, evacuated 1.00 L container. The initial pressure of the PCl5 (g) is 1.00 atm. The temperature is held constant until the PCl5 (g) reaches equilibr ...
... PCl5 (g) ⇆ PCl3 (g) + Cl2 (g) PCl5 (g) decomposes into PCl3 (g) and Cl2 (g) according to the equation above. A sample of PCl5 (g) is placed into a rigid, evacuated 1.00 L container. The initial pressure of the PCl5 (g) is 1.00 atm. The temperature is held constant until the PCl5 (g) reaches equilibr ...
3.1 Balancing Chemical Equations
... Mi Vi = Mf Vf Rearrangement of the equation allows for the calculation of the molarity of the diluted (Mf) solution: ...
... Mi Vi = Mf Vf Rearrangement of the equation allows for the calculation of the molarity of the diluted (Mf) solution: ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.