Text S1.
... ccpVTZ basis set. The IEFPCM continuum solvent model was employed to imitate an organic solvent environment (= 4). Atomic charges were obtained by fitting the molecular electrostatic potential of dipeptides using the method Restraint Electrostatic Potential (RESP)3. The fitting phase consisted of t ...
... ccpVTZ basis set. The IEFPCM continuum solvent model was employed to imitate an organic solvent environment (= 4). Atomic charges were obtained by fitting the molecular electrostatic potential of dipeptides using the method Restraint Electrostatic Potential (RESP)3. The fitting phase consisted of t ...
The Language of Chemistry
... visually distinguishable. • When separated, the components of both types of mixtures yields pure substances. ...
... visually distinguishable. • When separated, the components of both types of mixtures yields pure substances. ...
Chapter 7 - Chemical Quantities
... All of the equations we’ve seen so far have assumed that 100% of product was formed. In reality, this is rarely the case. Side reactions or the equilibrium can prevent reactions from going to completion. When chemists perform reactions, there are two things they want to know; a) what is the theoreti ...
... All of the equations we’ve seen so far have assumed that 100% of product was formed. In reality, this is rarely the case. Side reactions or the equilibrium can prevent reactions from going to completion. When chemists perform reactions, there are two things they want to know; a) what is the theoreti ...
Unit 5 and 6 revsion - Deans Community High School
... Q14. The burning of fossil fuels is a major source of air pollution. (a) Name the acidic gas formed when air in a car engine is sparked. (b) Name the acidic gas formed when coal with a high sulphur content is burned. (c) Name the toxic gas formed when methane is burned in a limited supply of air. (d ...
... Q14. The burning of fossil fuels is a major source of air pollution. (a) Name the acidic gas formed when air in a car engine is sparked. (b) Name the acidic gas formed when coal with a high sulphur content is burned. (c) Name the toxic gas formed when methane is burned in a limited supply of air. (d ...
Chapter 5
... Calculation of number of moles and number of molecules (or formula units) Determination of empirical formulas from experimental data Finding the molecular formula from the empirical formula and molecular mass Naming rules for simple substances (see Handout 1) Organic compounds - definition; hydrocar ...
... Calculation of number of moles and number of molecules (or formula units) Determination of empirical formulas from experimental data Finding the molecular formula from the empirical formula and molecular mass Naming rules for simple substances (see Handout 1) Organic compounds - definition; hydrocar ...
Review for second exam:
... Calculation of number of moles and number of molecules (or formula units) Determination of empirical formulas from experimental data Finding the molecular formula from the empirical formula and molecular mass Naming rules for simple substances (see Handout 1) Organic compounds - definition; hydrocar ...
... Calculation of number of moles and number of molecules (or formula units) Determination of empirical formulas from experimental data Finding the molecular formula from the empirical formula and molecular mass Naming rules for simple substances (see Handout 1) Organic compounds - definition; hydrocar ...
Physical Properties
... Pure Substances • A pure substance has well defined physical and chemical properties. • Pure substances can be classified as elements or compounds. • Compounds can be further reduced into two or more elements. • Elements consist of only one type of atom. They cannot be decomposed or further simplifi ...
... Pure Substances • A pure substance has well defined physical and chemical properties. • Pure substances can be classified as elements or compounds. • Compounds can be further reduced into two or more elements. • Elements consist of only one type of atom. They cannot be decomposed or further simplifi ...
Raman Spectroscopy Case Study: The Dissolution of Amorphous
... • Then possible to image and record the spectral changes representative of subtle chemical differences such as the change in crystallinity with dissolution. ...
... • Then possible to image and record the spectral changes representative of subtle chemical differences such as the change in crystallinity with dissolution. ...
Study Guide for Exam 2_Sp12
... Periodic trends regarding atomic and ionic radii. What is meant by valence electrons? What is ionization energy? What is the octet rule? What are ions? How do they relate to the octet rule? How is charge balance related to writing formulas of ionic compounds? Write dot formulas for atoms, and show h ...
... Periodic trends regarding atomic and ionic radii. What is meant by valence electrons? What is ionization energy? What is the octet rule? What are ions? How do they relate to the octet rule? How is charge balance related to writing formulas of ionic compounds? Write dot formulas for atoms, and show h ...
Chapter 7
... 2. The amount of a substance that contains the same number of particles as the number of atoms in exactly 12 g of carbon-12 ...
... 2. The amount of a substance that contains the same number of particles as the number of atoms in exactly 12 g of carbon-12 ...
groups (families) vs rows
... aspirin, dyes and disinfectants. One industrial method of preparing chlorobenzene is to react benzene, C6H6 , with chlorine, which is represented by the following equation: C6H6 (l) + Cl2 (g) → C6H5Cl (s) + HCl (g) When 36.8g of C6H6 react with an excess of Cl2, the actual yield of C6H5Cl is 38.8g W ...
... aspirin, dyes and disinfectants. One industrial method of preparing chlorobenzene is to react benzene, C6H6 , with chlorine, which is represented by the following equation: C6H6 (l) + Cl2 (g) → C6H5Cl (s) + HCl (g) When 36.8g of C6H6 react with an excess of Cl2, the actual yield of C6H5Cl is 38.8g W ...
Exam 1 Review Sheet
... solution keys and work to a known answer, but remember that you won’t have the answers on exam day. Be honest with yourself: do you really know how to work a problem? Do you truly understand the concepts behind it? 5. If the answers to the questions posed in #4 are ‘no’ or ‘maybe not,’ you have a nu ...
... solution keys and work to a known answer, but remember that you won’t have the answers on exam day. Be honest with yourself: do you really know how to work a problem? Do you truly understand the concepts behind it? 5. If the answers to the questions posed in #4 are ‘no’ or ‘maybe not,’ you have a nu ...
Chapter 12 - "Chemical Formulas and Equations"
... • Units of measurement used with equations. – We use a mole concept to bring together the concepts of counting numbers and atomic weights of elements. – The mole is derived from the following information. • Atomic weights are an average of the relative masses of all of the isotopes of the given ele ...
... • Units of measurement used with equations. – We use a mole concept to bring together the concepts of counting numbers and atomic weights of elements. – The mole is derived from the following information. • Atomic weights are an average of the relative masses of all of the isotopes of the given ele ...
CHAPTER 25 - CARBON AND ITS COMPOUNDS
... Molecules with single bonds can rotate about the single bond. These are the same compound. They have the same melting point, the same boiling points and the same chemistry. The higher the temperature the faster they change back and forth from one form to the other. Example: cyclohexane (chair and bo ...
... Molecules with single bonds can rotate about the single bond. These are the same compound. They have the same melting point, the same boiling points and the same chemistry. The higher the temperature the faster they change back and forth from one form to the other. Example: cyclohexane (chair and bo ...
Chemistry 11 Review Sheet
... a) write and balance the equation for this reaction. b) what volume (in liters) of oxygen will be used? c) what mass of carbon dioxide would be produced? ...
... a) write and balance the equation for this reaction. b) what volume (in liters) of oxygen will be used? c) what mass of carbon dioxide would be produced? ...
Gilad Haran - Laboratoire Léon Brillouin
... Fluorescence spectroscopy on the single-molecule level can provide a unique view of the heterogeneous dynamics of folding proteins. We are using a custom-developed set of methodologies, based on intra-molecular fluorescence resonance energy transfer (FRET), to study folding in real time. In particul ...
... Fluorescence spectroscopy on the single-molecule level can provide a unique view of the heterogeneous dynamics of folding proteins. We are using a custom-developed set of methodologies, based on intra-molecular fluorescence resonance energy transfer (FRET), to study folding in real time. In particul ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... 2 C6H12(l) + 5 O2(g) → 2 H2C6H8O4(l) + 2 H2O(g) (a) Assume that you carry out this reaction starting with 25.0 g of cyclohexane and that cyclohexane is the limiting reactant. What is the theoretical yield of adipic acid? (b) If you obtain 33.5 g of adipic acid from your reaction, what is the percent ...
... 2 C6H12(l) + 5 O2(g) → 2 H2C6H8O4(l) + 2 H2O(g) (a) Assume that you carry out this reaction starting with 25.0 g of cyclohexane and that cyclohexane is the limiting reactant. What is the theoretical yield of adipic acid? (b) If you obtain 33.5 g of adipic acid from your reaction, what is the percent ...
"Supramolecular chemistry is the chemistry of the intermolecular
... interactions, i.e. on the association of two or more building blocks, which are held together by intermolecular bonds" F. Vögtle "Supramolecular chemistry is defined as chemistry "beyond the molecule", as chemistry of tailor-shaped intermolecular interaction. In 'supramolecules' information is store ...
... interactions, i.e. on the association of two or more building blocks, which are held together by intermolecular bonds" F. Vögtle "Supramolecular chemistry is defined as chemistry "beyond the molecule", as chemistry of tailor-shaped intermolecular interaction. In 'supramolecules' information is store ...
Chemical Nomenclature, Formulas, and Equations
... The rules for naming oxoanions, anions of oxoacids, are as follows: 1. When all the H ions are removed from the “-ic” acid, the anion’s name ends with “-ate.” 2. When all the H ions are removed from the “-ous” acid, the anion’s name ends with “-ite.” 3. The names of anions in which one or more but ...
... The rules for naming oxoanions, anions of oxoacids, are as follows: 1. When all the H ions are removed from the “-ic” acid, the anion’s name ends with “-ate.” 2. When all the H ions are removed from the “-ous” acid, the anion’s name ends with “-ite.” 3. The names of anions in which one or more but ...
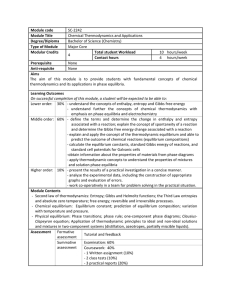
Module code SC-2242 Module Title Chemical Thermodynamics and
... Lower order: 30% - understand the concepts of enthalpy, entropy and Gibbs free energy - understand further the concepts of chemical thermodynamics with emphasis on phase equilibria and electrochemistry Middle order: 60% - define the terms and determine the change in enthalpy ...
... Lower order: 30% - understand the concepts of enthalpy, entropy and Gibbs free energy - understand further the concepts of chemical thermodynamics with emphasis on phase equilibria and electrochemistry Middle order: 60% - define the terms and determine the change in enthalpy ...
Document
... 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. Mono- is OPTIONAL on the ...
... 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. Mono- is OPTIONAL on the ...
Introduction to Chemical Reactions and Equations Study Guide
... 2. The numbers written to the bottom right of element symbols in a compound are called ___subscripts________. These numbers tell you the ratio of __atoms______ in the compound. 3. Sometimes the names of ionic compounds include roman numerals written in parenthesis after the cation’s name. What do th ...
... 2. The numbers written to the bottom right of element symbols in a compound are called ___subscripts________. These numbers tell you the ratio of __atoms______ in the compound. 3. Sometimes the names of ionic compounds include roman numerals written in parenthesis after the cation’s name. What do th ...
Chapter 3 - Bruder Chemistry
... The quantitative nature of chemical formulas and reactions is called stoichiometry. Lavoisier observed that mass is conserved in a chemical reaction. • This observation is known as the law of conservation of mass. Chemical equations give a description of a chemical reaction. There are two parts to a ...
... The quantitative nature of chemical formulas and reactions is called stoichiometry. Lavoisier observed that mass is conserved in a chemical reaction. • This observation is known as the law of conservation of mass. Chemical equations give a description of a chemical reaction. There are two parts to a ...
Notes - Ch 2
... 2-10 Determination of Molecular Formulas A) These are the formulas written as the molecule actually occurs. To determine them you need to know the molar mass (or be able to calculate it) B) Example: Many compounds have the empirical formula of CH2O. Here are the molecular weights of three. Determin ...
... 2-10 Determination of Molecular Formulas A) These are the formulas written as the molecule actually occurs. To determine them you need to know the molar mass (or be able to calculate it) B) Example: Many compounds have the empirical formula of CH2O. Here are the molecular weights of three. Determin ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.