Name #_____
... A) The molecular formula of a compound can be the same as its empirical formula. B) The molecular formula of a compound can be some whole-number multiple of its empirical formula. C) Several compounds can have the same empirical formula, but have different molecular formulas. D) The empirical formul ...
... A) The molecular formula of a compound can be the same as its empirical formula. B) The molecular formula of a compound can be some whole-number multiple of its empirical formula. C) Several compounds can have the same empirical formula, but have different molecular formulas. D) The empirical formul ...
Slide 1
... Eg. Give a.)the total # of atoms and b.)the gram formula mass (gfm) for each of the following (round to whole #) 1.) NaCl ...
... Eg. Give a.)the total # of atoms and b.)the gram formula mass (gfm) for each of the following (round to whole #) 1.) NaCl ...
Unit 3: Bonding and Nomenclature Content Outline: Chemical
... 2. The attractive strengths vary between the atoms is due to the size and charges associated with the ions in the compound. a. These ionic compound strengths are measured using Lattice Energies b. This is the amount of energy released when one mole of an ionic crystalline compound is formed from gas ...
... 2. The attractive strengths vary between the atoms is due to the size and charges associated with the ions in the compound. a. These ionic compound strengths are measured using Lattice Energies b. This is the amount of energy released when one mole of an ionic crystalline compound is formed from gas ...
Chem A Naming Polyatomic Ions Name: Hour: ______ Page 1
... compound we named was an ionic compound. Ionic compounds are made of a metal and a nonmetal. The second type of compound we named was a covalent compound. Covalent compounds are made of two nonmetals. In this packet we will learned how to name compounds with more than two elements. In all the exampl ...
... compound we named was an ionic compound. Ionic compounds are made of a metal and a nonmetal. The second type of compound we named was a covalent compound. Covalent compounds are made of two nonmetals. In this packet we will learned how to name compounds with more than two elements. In all the exampl ...
Chapter 3
... What mass of N2 gas theoretically would result from the reaction of 150.0 g of hydrazine and 100.0 g of N2O4? If you actually obtain 66.4 g N2, what is percent yield? ...
... What mass of N2 gas theoretically would result from the reaction of 150.0 g of hydrazine and 100.0 g of N2O4? If you actually obtain 66.4 g N2, what is percent yield? ...
Writing Formulas Worksheet 1. sodium nitrate 16. aluminum sulfide
... hydrates. The number of moles of water present per mole of anhydrous salt (salt minus water of crystallization) is usually a whole number. One example is the hydrate of copper (II) sulfate. Its blue crystals look and feel dry. Yet each mole of hydrate contains 5 moles of water. Its formula is CuSO4 ...
... hydrates. The number of moles of water present per mole of anhydrous salt (salt minus water of crystallization) is usually a whole number. One example is the hydrate of copper (II) sulfate. Its blue crystals look and feel dry. Yet each mole of hydrate contains 5 moles of water. Its formula is CuSO4 ...
Chemistry 11 - Sardis Secondary
... - identifying limiting and excess reagents in a chemical reaction - calculating the amount of excess reactant - calculating the amount of product formed in a reaction using the limiting reactant C. Percent Yield (text pgs. 365-373) - calculating the efficiency of a chemical reaction from percent yie ...
... - identifying limiting and excess reagents in a chemical reaction - calculating the amount of excess reactant - calculating the amount of product formed in a reaction using the limiting reactant C. Percent Yield (text pgs. 365-373) - calculating the efficiency of a chemical reaction from percent yie ...
Elaine Teto
... relative amounts of each substance present. However, in using these coefficients we come to our next topic, balancing chemical reactions. One of the most important laws to remember, when considering balancing equations is the Law of Conservation of Mass, which states that matter can neither be creat ...
... relative amounts of each substance present. However, in using these coefficients we come to our next topic, balancing chemical reactions. One of the most important laws to remember, when considering balancing equations is the Law of Conservation of Mass, which states that matter can neither be creat ...
Chapter 3
... By definition, a molar mass is the mass of 1 mol of a substance (i.e., g/mol). The molar mass of an element is the mass number for the element that we find on the periodic table. The formula weight (in amu’s) will be the same number as the molar mass (in g/mol). ...
... By definition, a molar mass is the mass of 1 mol of a substance (i.e., g/mol). The molar mass of an element is the mass number for the element that we find on the periodic table. The formula weight (in amu’s) will be the same number as the molar mass (in g/mol). ...
Study Guide for Test 2: Chapters 3 & 4... This is NOT a complete list of what will be... Revised March 4, 2014
... 11) Still know Avogadro’s Number (Chapter 2) and be able to convert between number of items (atoms, molecules, ions, etc.) and moles of that item. Be able to combine this calculation with molar mass. (1 mole items = 6.022 x 1023 items) 12) Be able to convert between moles of a compound and moles of ...
... 11) Still know Avogadro’s Number (Chapter 2) and be able to convert between number of items (atoms, molecules, ions, etc.) and moles of that item. Be able to combine this calculation with molar mass. (1 mole items = 6.022 x 1023 items) 12) Be able to convert between moles of a compound and moles of ...
The Mole
... atoms, ions or molecules. Visible and measurable amounts contain huge numbers of these very tiny particles. Rather than thinking in terms of these individual small particles, chemists use a larger quantity of matter called a MOLE (symbol mol). A mole is just a quantity of material and is the primary ...
... atoms, ions or molecules. Visible and measurable amounts contain huge numbers of these very tiny particles. Rather than thinking in terms of these individual small particles, chemists use a larger quantity of matter called a MOLE (symbol mol). A mole is just a quantity of material and is the primary ...
chapter 2
... a. Alkali Metals – most reactive metals, react violently with water b. Alkaline Earth Metals – reactive metals but less so than alkali c. Halogens – most reactive non-metals, most are poisonous gases d. Noble Gases – do not react 3. If a noble gas could form a +1 ion, which of the noble gases would ...
... a. Alkali Metals – most reactive metals, react violently with water b. Alkaline Earth Metals – reactive metals but less so than alkali c. Halogens – most reactive non-metals, most are poisonous gases d. Noble Gases – do not react 3. If a noble gas could form a +1 ion, which of the noble gases would ...
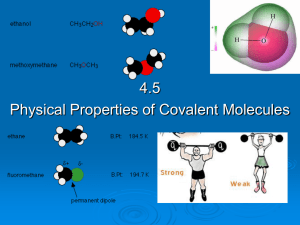
4.5 Physical properties of molecular covalent
... Covalent molecules are not charged because they are overall neutral and therefore do not conduct electricity. • Some covalent molecules can react with water and produce free ions which can carry an electrical current. • E.g. ammonia, NH3 NH3 (l) + H2O (l) ↔ NH4+ (aq) + OH- (aq) ...
... Covalent molecules are not charged because they are overall neutral and therefore do not conduct electricity. • Some covalent molecules can react with water and produce free ions which can carry an electrical current. • E.g. ammonia, NH3 NH3 (l) + H2O (l) ↔ NH4+ (aq) + OH- (aq) ...
Transition Metals - Ligand Stability and Chelation
... case. When looking carefully, you are only actually breaking and making bonds around the central metal. In each case, you break and make the similar (or the same) bonds. This is irrespective of what ligands are attached to the central metal. For example: ...
... case. When looking carefully, you are only actually breaking and making bonds around the central metal. In each case, you break and make the similar (or the same) bonds. This is irrespective of what ligands are attached to the central metal. For example: ...
Hints for Names and Formulas (Ch. 4 in Zumdahl Chemistry)
... ◘ examples: Co(OH) 3 is cobalt(III) hydroxide and Al2(SO3) 3 is aluminum sulfite (7) The sum of the positive and negative charges must total zero for any ionic compound formula ● this fact enables you to predict the subscripts of the ionic formula ◘ in aluminum sulfate Al3+ ion combines with sulfate ...
... ◘ examples: Co(OH) 3 is cobalt(III) hydroxide and Al2(SO3) 3 is aluminum sulfite (7) The sum of the positive and negative charges must total zero for any ionic compound formula ● this fact enables you to predict the subscripts of the ionic formula ◘ in aluminum sulfate Al3+ ion combines with sulfate ...
Chemical Formulas and Chemical Compounds
... a. What is the mass in grams of 3.00 mol of butane? ...
... a. What is the mass in grams of 3.00 mol of butane? ...
Limiting reactant - Dr. Gregory Chemistry
... relationships between the amounts of reactants used and amounts of products formed in a chemical reaction. It is based on the law of conservation of mass. ...
... relationships between the amounts of reactants used and amounts of products formed in a chemical reaction. It is based on the law of conservation of mass. ...
Correct Answer is 2
... A hydrated salt is a solid that includes water molecules within its crystal structure. A student heated a 9.10-gram sample of a hydrated salt to a constant mass of 5.41 grams. What percent by mass of water did the salt contain? ...
... A hydrated salt is a solid that includes water molecules within its crystal structure. A student heated a 9.10-gram sample of a hydrated salt to a constant mass of 5.41 grams. What percent by mass of water did the salt contain? ...
General Chemistry
... •O has a mass of 16 amu – but we can’t weigh out anything in amu •If we want to keep the number “16” for the mass of oxygen in some real units (like grams) then we are dealing with a whole bunch of atoms (in 16 g of oxygen). •That bunch of atoms is called a mole. ...
... •O has a mass of 16 amu – but we can’t weigh out anything in amu •If we want to keep the number “16” for the mass of oxygen in some real units (like grams) then we are dealing with a whole bunch of atoms (in 16 g of oxygen). •That bunch of atoms is called a mole. ...
General Chemistry
... •O has a mass of 16 amu – but we can’t weigh out anything in amu •If we want to keep the number “16” for the mass of oxygen in some real units (like grams) then we are dealing with a whole bunch of atoms (in 16 g of oxygen). •That bunch of atoms is called a mole. ...
... •O has a mass of 16 amu – but we can’t weigh out anything in amu •If we want to keep the number “16” for the mass of oxygen in some real units (like grams) then we are dealing with a whole bunch of atoms (in 16 g of oxygen). •That bunch of atoms is called a mole. ...
Ch.5
... The number of grams of a substance equivalent to the sum of all its average atomic mass units (amu) is known as the molar mass. ...
... The number of grams of a substance equivalent to the sum of all its average atomic mass units (amu) is known as the molar mass. ...
Lecture3_Module_19
... representing a snap shot in time, with the volume weighted average concentrations found in the literature ...
... representing a snap shot in time, with the volume weighted average concentrations found in the literature ...
Chapter 7_honors
... equal to ZERO. (ex. Na, O2, P4, S8) 2. The more electronegative element in a binary molecular compound is assigned the # equal to the negative charge it would have as an anion. Likewise the less electronegative element is assigned the positive charge as the cation. ...
... equal to ZERO. (ex. Na, O2, P4, S8) 2. The more electronegative element in a binary molecular compound is assigned the # equal to the negative charge it would have as an anion. Likewise the less electronegative element is assigned the positive charge as the cation. ...
Host–guest chemistry

In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through noncovalent bonding. Noncovalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. There are four commonly mentioned types of non-covalent interactions: hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.