Biol 1020 Ch. 4: organic molecules
... carbon is not a strongly electron seeking element, and it does not readily give up its electrons; therefore: ...
... carbon is not a strongly electron seeking element, and it does not readily give up its electrons; therefore: ...
Chemistry I Honors – Semester Exam Review – Fall 2000
... STRATEGY: Start by reading through your notes to refresh your memory on these topics. Then, use this review sheet as a starting point to identify the areas on which you need to spend more study time. For those areas, go back to homework assignments, quizzes, and reviews to practice more problems. I ...
... STRATEGY: Start by reading through your notes to refresh your memory on these topics. Then, use this review sheet as a starting point to identify the areas on which you need to spend more study time. For those areas, go back to homework assignments, quizzes, and reviews to practice more problems. I ...
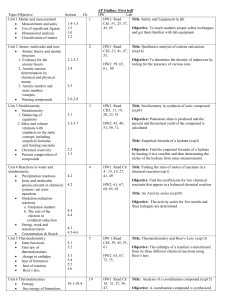
Topic/Objective - cloudfront.net
... c. Bond energies, enthalpy and chemical reactions Molecular models a. Lewis structures b. Valence bond: hybridization of orbitals, resonance, sigma and pi bonds c. VSEPR Unit 11 Gases Laws of ideal gases a. Equation of state for an ideal gas b. Partial pressures Kinetic molecular theory a. Int ...
... c. Bond energies, enthalpy and chemical reactions Molecular models a. Lewis structures b. Valence bond: hybridization of orbitals, resonance, sigma and pi bonds c. VSEPR Unit 11 Gases Laws of ideal gases a. Equation of state for an ideal gas b. Partial pressures Kinetic molecular theory a. Int ...
study guide first semester chemistry
... 1. Write the balanced equation for the following: (include the state of each reactant and product) a. magnesium reacts with nitrogen to produce magnesium nitride. (3Mg(s) + N2(g) Mg3N2(s) b. silver nitrate reacts with copper to form copper(II) nitrate and silver. ...
... 1. Write the balanced equation for the following: (include the state of each reactant and product) a. magnesium reacts with nitrogen to produce magnesium nitride. (3Mg(s) + N2(g) Mg3N2(s) b. silver nitrate reacts with copper to form copper(II) nitrate and silver. ...
Name - TeacherWeb
... In a sigma (σ) bond, the molecular orbital is symmetrical around the axis connecting two atomic nuclei. In a pi (π) bond, the orbitals are sausage-shaped regions above and below the bond axis. ...
... In a sigma (σ) bond, the molecular orbital is symmetrical around the axis connecting two atomic nuclei. In a pi (π) bond, the orbitals are sausage-shaped regions above and below the bond axis. ...
C2 Chemistry - Burton Borough School
... ATOMIC NUMBER (proton number/the small one) The number of outer shell electrons match the group the element is found in. E.g. Lithium 2,1 is a group 1 element. ...
... ATOMIC NUMBER (proton number/the small one) The number of outer shell electrons match the group the element is found in. E.g. Lithium 2,1 is a group 1 element. ...
Review Sheet for Chemistry* First Semester Final
... Write formulas for the following molecular compounds: ...
... Write formulas for the following molecular compounds: ...
Chemistry Outcomes - hrsbstaff.ednet.ns.ca
... Explain the hydrogen line spectrum in terms of Bohr Model of the atom State two differences between the Bohr model and the quantum mechanical model of the atom Draw an energy level diagram for a given atom Define valence shell and valence electrons Label the sublevels on an energy level diagram with ...
... Explain the hydrogen line spectrum in terms of Bohr Model of the atom State two differences between the Bohr model and the quantum mechanical model of the atom Draw an energy level diagram for a given atom Define valence shell and valence electrons Label the sublevels on an energy level diagram with ...
Gr - loyolascience2
... Why is it true that polar substances dissolve in polar solvents and non-polar substances dissolve in non-polar solvents? There are two important tendencies to consider. First, a substance will dissolve at all if the attraction between solute and solvent particles is stronger than the attraction betw ...
... Why is it true that polar substances dissolve in polar solvents and non-polar substances dissolve in non-polar solvents? There are two important tendencies to consider. First, a substance will dissolve at all if the attraction between solute and solvent particles is stronger than the attraction betw ...
JC2-Chemical-Bonding-Time-Trial-Soln
... The following lists the boiling points of fluorine and some fluoride compounds. By reference to their chemical structures and types of bonding, explain as fully as you can the differences in their boiling points. Boiling point / oC ...
... The following lists the boiling points of fluorine and some fluoride compounds. By reference to their chemical structures and types of bonding, explain as fully as you can the differences in their boiling points. Boiling point / oC ...
Dehydration of 2-methylcyclohexanol
... the 2o carbocation can undergo a rearrangement forming a more stable 3 o carbocation. This carbocation can form two products methylenecyclohexane and 1-methylcyclohexene. The product distribution can be under either kinetic or ...
... the 2o carbocation can undergo a rearrangement forming a more stable 3 o carbocation. This carbocation can form two products methylenecyclohexane and 1-methylcyclohexene. The product distribution can be under either kinetic or ...
Examination questions Division of Condensed Matter Physics
... 18. Quantum dots, quantum wires, quantum wells: fabrication, electronic states in low ...
... 18. Quantum dots, quantum wires, quantum wells: fabrication, electronic states in low ...
8872 Chemistry H1 syllabus for 2016
... describe, interpret and/or predict the effect of different types of bonding (ionic bonding; covalent bonding; hydrogen bonding; other intermolecular interactions; metallic bonding) on the physical properties of substances ...
... describe, interpret and/or predict the effect of different types of bonding (ionic bonding; covalent bonding; hydrogen bonding; other intermolecular interactions; metallic bonding) on the physical properties of substances ...
atom a very small particle that makes up most kinds of matters and
... two or more atoms of the same element that have different numbers of neutrons in the nuclei (same number of protons) states that mass is neither created nor destroyed and as a result the mass of the substances before a physical or chemical change is equal to the mass of the substances present after ...
... two or more atoms of the same element that have different numbers of neutrons in the nuclei (same number of protons) states that mass is neither created nor destroyed and as a result the mass of the substances before a physical or chemical change is equal to the mass of the substances present after ...
Visualizing the Transition State and
... to the discussion of collision theory, they would immediately shout “from molecular collisions!” At this point in the demonstration, I have two students participate in colliding two Switch Pitches together. I have the rest of the class predict under what conditions reactants will most efficiently co ...
... to the discussion of collision theory, they would immediately shout “from molecular collisions!” At this point in the demonstration, I have two students participate in colliding two Switch Pitches together. I have the rest of the class predict under what conditions reactants will most efficiently co ...
Year 10 Chemistry Exam June 2011 Multiple Choice Section A
... 1. An aqueous solution is obtained when: a. a substance dissolves in any liquid b. a substance is dissolved in water c. when a substance is mixed with water and doesn’t dissolve d. water is removed from a substance 2. The graph shows the relative amount of chemical substances which can be taken up b ...
... 1. An aqueous solution is obtained when: a. a substance dissolves in any liquid b. a substance is dissolved in water c. when a substance is mixed with water and doesn’t dissolve d. water is removed from a substance 2. The graph shows the relative amount of chemical substances which can be taken up b ...
Carbene Singlets, Triplets, and the Physics that
... (molecular, in the likely event of two molecules interacting) orbitals, resulting in a mixing of pure orbitals. This mixing can either yield a bonding or an anti-bonding orbital, as seen in the interaction diagram (see figure 3). Orbital mixing diagrams like these are often qualitative, and while kn ...
... (molecular, in the likely event of two molecules interacting) orbitals, resulting in a mixing of pure orbitals. This mixing can either yield a bonding or an anti-bonding orbital, as seen in the interaction diagram (see figure 3). Orbital mixing diagrams like these are often qualitative, and while kn ...
Semester Exam Review - Teach-n-Learn-Chem
... STRATEGY: Start by reading through your notes to refresh your memory on these topics. Then, use this review sheet as a starting point to identify the areas on which you need to spend more study time. For those areas, go back to homework assignments, quizzes, and reviews to practice more problems. I ...
... STRATEGY: Start by reading through your notes to refresh your memory on these topics. Then, use this review sheet as a starting point to identify the areas on which you need to spend more study time. For those areas, go back to homework assignments, quizzes, and reviews to practice more problems. I ...
Ch03macromolecules - Environmental
... All of life is built on carbon Cells ~72% H2O ~25% carbon compounds ...
... All of life is built on carbon Cells ~72% H2O ~25% carbon compounds ...
Honors-Final-Review-2014
... a. A solution that keeps a constant neutral pH when small amounts of acid or base are added b. Solution of known concentration c. Acid contains one H d. Acid contains three or more H’s e. The point at which the indicator changes color f. Any substance that accepts a proton g. Any substance that dona ...
... a. A solution that keeps a constant neutral pH when small amounts of acid or base are added b. Solution of known concentration c. Acid contains one H d. Acid contains three or more H’s e. The point at which the indicator changes color f. Any substance that accepts a proton g. Any substance that dona ...