UNIT I: Introduction to Chemistry
... Use electronegativity values to determine whether a compound is ionic, polar or nonpolar covalent. Illustrate neutral atoms and ions using electron dot notation. Illustrate ionic and covalent bonds utilizing electron dot notation. Use appropriate materials to build adequate models of simple molecule ...
... Use electronegativity values to determine whether a compound is ionic, polar or nonpolar covalent. Illustrate neutral atoms and ions using electron dot notation. Illustrate ionic and covalent bonds utilizing electron dot notation. Use appropriate materials to build adequate models of simple molecule ...
ppt - Wits Structural Chemistry
... If the chemical formula of a substance is the same as its molecular formula, then the formula weight is also called the molecular weight. Molecular weight (Mr or MW) is the mass of a collection of atoms represented by a chemical formula for a molecule. What is the molecular formula for glucose? MW( ...
... If the chemical formula of a substance is the same as its molecular formula, then the formula weight is also called the molecular weight. Molecular weight (Mr or MW) is the mass of a collection of atoms represented by a chemical formula for a molecule. What is the molecular formula for glucose? MW( ...
SIDE GROUP ADDITION TO THE POLYCYCLIC AROMATIC
... Based on the evidence that aromatic compounds are commonly observed in interstellar space and in meteorites and should condense into interstellar grain mantles, we have investigated the energetic processing of PAHs in interstellar ice analogs to explore possible connections between interstellar and ...
... Based on the evidence that aromatic compounds are commonly observed in interstellar space and in meteorites and should condense into interstellar grain mantles, we have investigated the energetic processing of PAHs in interstellar ice analogs to explore possible connections between interstellar and ...
Chapter 11 Chemical Calculations
... than 2 elements choose the smallest overall and use that as the denominator for all calculations B. You will usually get nice whole numbers, but sometimes may end up with ratios like 1.5/1. In this case you need to multiply both the numerator and the denominator by a single number that will give you ...
... than 2 elements choose the smallest overall and use that as the denominator for all calculations B. You will usually get nice whole numbers, but sometimes may end up with ratios like 1.5/1. In this case you need to multiply both the numerator and the denominator by a single number that will give you ...
Two-Electron Reduction of a Vanadium(V) Nitride by CO to Release
... 150.6(1)°, on par with other isocyanate complexes.6 With 1-V(NCO) in hand, attention turned to probing the redox chemistry of the complex. When treated with 0.5% Na/Hg in the same manner as had been done for 1-Nb(NCO), 1-V(NCO) undergoes cyanate dissociation to give 1-V and sodium cyanate (Scheme 1B ...
... 150.6(1)°, on par with other isocyanate complexes.6 With 1-V(NCO) in hand, attention turned to probing the redox chemistry of the complex. When treated with 0.5% Na/Hg in the same manner as had been done for 1-Nb(NCO), 1-V(NCO) undergoes cyanate dissociation to give 1-V and sodium cyanate (Scheme 1B ...
8 theoretical problems 2 practical problems
... give the same osazone and therefore have identical stereochemistry at C-3, C-4, and C-5 (and C-6). A and B are also different from compound 1 (i.e. D-mannose) yet give the same osazone, and thus one of them must be the C-2 epimer of D-mannose (i.e. D-glucose) and the other must be the corresponding ...
... give the same osazone and therefore have identical stereochemistry at C-3, C-4, and C-5 (and C-6). A and B are also different from compound 1 (i.e. D-mannose) yet give the same osazone, and thus one of them must be the C-2 epimer of D-mannose (i.e. D-glucose) and the other must be the corresponding ...
CHEMICAL EQUATIONS - Clayton State University
... Ionic solid sodium chloride (Na+ and Cl- ions) formed from solid sodium and chlorine gas 2Na(s) + Cl2(g) → 2NaCl(s) The oxidation (rusting) of iron by reaction with moist air 4Fe(s) + 3O2(g) → 2Fe2O3(s) ...
... Ionic solid sodium chloride (Na+ and Cl- ions) formed from solid sodium and chlorine gas 2Na(s) + Cl2(g) → 2NaCl(s) The oxidation (rusting) of iron by reaction with moist air 4Fe(s) + 3O2(g) → 2Fe2O3(s) ...
Moles
... One mole of seconds is about 19 quadrillion years, 4,240,666 times the age of the earth, or 954,150 times the age of the universe itself. A one liter bottle of water contains 55.5 moles of water. If you started with a mole of pennies and spent $1,000,000 every second for 100 years, you would still h ...
... One mole of seconds is about 19 quadrillion years, 4,240,666 times the age of the earth, or 954,150 times the age of the universe itself. A one liter bottle of water contains 55.5 moles of water. If you started with a mole of pennies and spent $1,000,000 every second for 100 years, you would still h ...
Chemistry - Sanskriti School
... Q14. An organic compound on analysis gave the following percentage composition; C=57.8%, H=3.6% and the rest is oxygen. The molecular mass of the compound was found to be 166. Find out the molecular formula of the compound. [C4H3O2] Q15. 250 ml of 0.5M Na2SO4 solution is added to an aqueous solution ...
... Q14. An organic compound on analysis gave the following percentage composition; C=57.8%, H=3.6% and the rest is oxygen. The molecular mass of the compound was found to be 166. Find out the molecular formula of the compound. [C4H3O2] Q15. 250 ml of 0.5M Na2SO4 solution is added to an aqueous solution ...
HEAd START TO A LEVEL CHEMISTRY WORKbOOK
... atoms and 16 g of oxygen since the relative atomic mass of hydrogen is 1 (x 2 because there two hydrogen atoms) and that of oxygen is 16. You should not learn large numbers of chemical formulae by heart. However, it is useful to know a few of them and when you do you should be able to work out the r ...
... atoms and 16 g of oxygen since the relative atomic mass of hydrogen is 1 (x 2 because there two hydrogen atoms) and that of oxygen is 16. You should not learn large numbers of chemical formulae by heart. However, it is useful to know a few of them and when you do you should be able to work out the r ...
Lecture 1 - Алтайский государственный технический
... sub-stances by chemical means. Compounds can be decomposed into two or more elements. Elements are the basic substances out of which all matter is composed. Everything in the world is made up from only 109 different elements. 90% of the human body is composed of only three elements: Oxygen, Carbon a ...
... sub-stances by chemical means. Compounds can be decomposed into two or more elements. Elements are the basic substances out of which all matter is composed. Everything in the world is made up from only 109 different elements. 90% of the human body is composed of only three elements: Oxygen, Carbon a ...
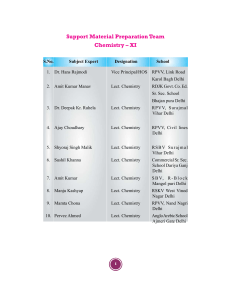
Support Material
... Law of Multiple Proportions (John Dalton) : When two elements combine to form two or more compounds, then the different masses of one element, which combine with a ®xed mass of the other, bear a simple ratio to one another. Gay Lussac’s Law : When gases combine or are produced in a chemical reac ...
... Law of Multiple Proportions (John Dalton) : When two elements combine to form two or more compounds, then the different masses of one element, which combine with a ®xed mass of the other, bear a simple ratio to one another. Gay Lussac’s Law : When gases combine or are produced in a chemical reac ...
chapter twenty-one transition metals and coordination chemistry
... The crystal field diagrams are different because the geometries of where the ligands point is different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 2 ...
... The crystal field diagrams are different because the geometries of where the ligands point is different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 2 ...
17.2 The Avogadro Number
... When counting atoms to balance an equation, remember that the coefficient applies to all atoms within the chemical formula for a compound. For example, 5CH4 means that 5 atoms of carbon and 20 atoms (5 × 4) of hydrogen are contributed to the chemical reaction by the compound methane. ...
... When counting atoms to balance an equation, remember that the coefficient applies to all atoms within the chemical formula for a compound. For example, 5CH4 means that 5 atoms of carbon and 20 atoms (5 × 4) of hydrogen are contributed to the chemical reaction by the compound methane. ...
Ch.1-Matter and Change
... Pure substances are either compounds or elements. A pure substance differs from a mixture in the following ways: Every sample of a given pure substance has exactly the same characteristic properties. Every sample of a given pure substance has exactly the same composition. Water is always 11.2% hydro ...
... Pure substances are either compounds or elements. A pure substance differs from a mixture in the following ways: Every sample of a given pure substance has exactly the same characteristic properties. Every sample of a given pure substance has exactly the same composition. Water is always 11.2% hydro ...
1.6 Energy changes in chemical reactions
... nitrogen oxides on a street-by-street basis. These high-resolution measurements help city planners to decide better policies for preventing health damage by pollution. On a global scale, an understanding of the chemical reactions that take place in the atmosphere is needed to tackle the environmenta ...
... nitrogen oxides on a street-by-street basis. These high-resolution measurements help city planners to decide better policies for preventing health damage by pollution. On a global scale, an understanding of the chemical reactions that take place in the atmosphere is needed to tackle the environmenta ...
Descriptive Chemistry of Elements p
... Formation of [AlH4] ion can be seen as coordination of H ion to the electron deficient AlH3 molecule. AlH3 + H [AlH4] Lithium aluminum hydride Li[AlH4] is a useful reducing agent. It reduces organic molecules such as aldehydes, ketones and carboxylic acids to alcohols. ...
... Formation of [AlH4] ion can be seen as coordination of H ion to the electron deficient AlH3 molecule. AlH3 + H [AlH4] Lithium aluminum hydride Li[AlH4] is a useful reducing agent. It reduces organic molecules such as aldehydes, ketones and carboxylic acids to alcohols. ...
coordination compounds
... 7. The nomenclature of complex compounds The name of the complex compounds are composed as follows: Name the ligands first, in alphabetical order, then the central atom or ion. Name of the complex is written in one word. Neutral ligands called without changes; in the names of negatively charged liga ...
... 7. The nomenclature of complex compounds The name of the complex compounds are composed as follows: Name the ligands first, in alphabetical order, then the central atom or ion. Name of the complex is written in one word. Neutral ligands called without changes; in the names of negatively charged liga ...
Chemistry
... aspects of general chemistry. Chemistry is mastered when students make the right connections in three key areas: topics that are related, conceptual reasoning with quantitative work, and the different modes of communicating information. McMurry/Fay’s Chemistry, Sixth Edition breaks through the tradi ...
... aspects of general chemistry. Chemistry is mastered when students make the right connections in three key areas: topics that are related, conceptual reasoning with quantitative work, and the different modes of communicating information. McMurry/Fay’s Chemistry, Sixth Edition breaks through the tradi ...
Final Exam Review Packet
... 8. What are the products of complete combustion reactions? 9. What is a binary compound? 10. Complete the equation Al + H2SO4 => ? 11. What is ionic bonding? 12. What is covalent bonding? 13. Name of Cr2O7-2? 14. What is a noble gas? 15. What is an oxide? 16. Balance: NH3 + O2 => H2O + NO2? 17. What ...
... 8. What are the products of complete combustion reactions? 9. What is a binary compound? 10. Complete the equation Al + H2SO4 => ? 11. What is ionic bonding? 12. What is covalent bonding? 13. Name of Cr2O7-2? 14. What is a noble gas? 15. What is an oxide? 16. Balance: NH3 + O2 => H2O + NO2? 17. What ...
The Mole
... all the water is removed. You are left with just a white powder. Hydrated describes the crystalline compound which contains water molecules in its structure. Anhydrous describes the compound when all the water of crystallisation has been removed. When compounds are crystallised from water, they freq ...
... all the water is removed. You are left with just a white powder. Hydrated describes the crystalline compound which contains water molecules in its structure. Anhydrous describes the compound when all the water of crystallisation has been removed. When compounds are crystallised from water, they freq ...