O 2 - Montville.net
... spread them across the United States of America, the country would be covered in popcorn to a depth of over 9 miles. If we were able to count atoms at the rate of 10 million per second, it would take about 2 billion years to count the atoms in one mole. Adopted from "Chemistry You Need to Know" by K ...
... spread them across the United States of America, the country would be covered in popcorn to a depth of over 9 miles. If we were able to count atoms at the rate of 10 million per second, it would take about 2 billion years to count the atoms in one mole. Adopted from "Chemistry You Need to Know" by K ...
Regents Chemistry - New York Science Teacher
... • In terms of energy and entropy, systems in nature tend to undergo ...
... • In terms of energy and entropy, systems in nature tend to undergo ...
Unit 2 Summary - A
... State the two main applications of fossil fuels:............................................ and .................................... State two significant disadvantages of being over-reliant on fossil fuels: ................................................................... and ................... ...
... State the two main applications of fossil fuels:............................................ and .................................... State two significant disadvantages of being over-reliant on fossil fuels: ................................................................... and ................... ...
Class XI Physical Chemistry Short note
... and protons. Since electrons have negligible mass, the entire mass of the atom was regarded as the mass of the proton only. Each proton has a mass of 1.67x 10-24 g which is taken as 1 unit mass. In 1920, Rutherford found that except for the hydrogen atom, the atomic masses of no other atom could be ...
... and protons. Since electrons have negligible mass, the entire mass of the atom was regarded as the mass of the proton only. Each proton has a mass of 1.67x 10-24 g which is taken as 1 unit mass. In 1920, Rutherford found that except for the hydrogen atom, the atomic masses of no other atom could be ...
Net ionic equation
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
Worked solutions to textbook questions 1 Chapter 14 From organic
... C. There is a double bond between two carbon atoms in alkenes. The first members of the other homologous series, methane, methanol and methanoic acid all contain 1 carbon atom. Q2. The systematic name for CH3CH2CH2CH(CH3)2 is: A 1,1-dimethylbutane B 2-methylpentane C 2-methylpentene D propyldimethyl ...
... C. There is a double bond between two carbon atoms in alkenes. The first members of the other homologous series, methane, methanol and methanoic acid all contain 1 carbon atom. Q2. The systematic name for CH3CH2CH2CH(CH3)2 is: A 1,1-dimethylbutane B 2-methylpentane C 2-methylpentene D propyldimethyl ...
No Slide Title
... Classification of Compounds as Electrolytes (in Water) We previously divided compounds into two general categories: Ionic compounds - Collections of cations (positive ions) and anions (negative ions) in a crystal structure. Usually form from a metal (or cation group) and a nonmetal (or anion group) ...
... Classification of Compounds as Electrolytes (in Water) We previously divided compounds into two general categories: Ionic compounds - Collections of cations (positive ions) and anions (negative ions) in a crystal structure. Usually form from a metal (or cation group) and a nonmetal (or anion group) ...
CHEMISTRY
... The contribution of 35Cl is: 34.96885 amu x 0.75771 = 26.496 amu The contribution of 37Cl is: 36.96590 amu x 0.24229 = 8.9565 amu The total average mass of a chlorine atom is the sum of the contributions of the individual isotopes: 26.496 amu + 8.9565 amu = 35.453 amu This number should be the same ...
... The contribution of 35Cl is: 34.96885 amu x 0.75771 = 26.496 amu The contribution of 37Cl is: 36.96590 amu x 0.24229 = 8.9565 amu The total average mass of a chlorine atom is the sum of the contributions of the individual isotopes: 26.496 amu + 8.9565 amu = 35.453 amu This number should be the same ...
Exam 961-1st Name___________________________________
... 9) Which element would have physical and chemical properties similar to chlorine? A) Ar B) Br C) P D) S ...
... 9) Which element would have physical and chemical properties similar to chlorine? A) Ar B) Br C) P D) S ...
Annex 1
... Corrections of the text of an application should be done by reprinting the whole page. Proof correction marks -as for example specified in the International Standard ISO 5776- are not accepted. Making corrections by means of white correcting fluid, self adhesive strips of paper, erasure or strikethr ...
... Corrections of the text of an application should be done by reprinting the whole page. Proof correction marks -as for example specified in the International Standard ISO 5776- are not accepted. Making corrections by means of white correcting fluid, self adhesive strips of paper, erasure or strikethr ...
The Mole
... Here is how the International Union of Pure and Applied Chemistry (IUPAC) defines "mole:" • The mole is the amount of substance of a system that contains as many elementary entities as there are atoms in 0.012 kilogram of carbon-12. When the mole is used, the elementary entities must be specified a ...
... Here is how the International Union of Pure and Applied Chemistry (IUPAC) defines "mole:" • The mole is the amount of substance of a system that contains as many elementary entities as there are atoms in 0.012 kilogram of carbon-12. When the mole is used, the elementary entities must be specified a ...
CHM203 - National Open University of Nigeria
... divisions of chemistry, viz: Physical, Inorganic and Organic chemistry. While inorganic and physical chemistry are detailed elsewhere, organic chemistry which deals with hydrocarbons, their numerous derivatives in addition to their physical and chemical properties will be studied in this text. This ...
... divisions of chemistry, viz: Physical, Inorganic and Organic chemistry. While inorganic and physical chemistry are detailed elsewhere, organic chemistry which deals with hydrocarbons, their numerous derivatives in addition to their physical and chemical properties will be studied in this text. This ...
Unit 3 Answer Key
... 2. A mole is a unit used to measure the amount of a substance. One mole contains the same number of particles as the number of atoms in 1 g of carbon-12. 3. You would have two times the Avogadro constant of hydrogen atoms. Rounded off, the number would be 2(6.02 × 1023) = 1.20 × 1024 hydrogen at ...
... 2. A mole is a unit used to measure the amount of a substance. One mole contains the same number of particles as the number of atoms in 1 g of carbon-12. 3. You would have two times the Avogadro constant of hydrogen atoms. Rounded off, the number would be 2(6.02 × 1023) = 1.20 × 1024 hydrogen at ...
College Grossmont 115
... that our measurement is precise to within one tenth of a gram. A value of 2.9 g indicates that the actual mass lies within the range of 2.8 to 3.0 grams. In this case, we record the ...
... that our measurement is precise to within one tenth of a gram. A value of 2.9 g indicates that the actual mass lies within the range of 2.8 to 3.0 grams. In this case, we record the ...
Lecture 2
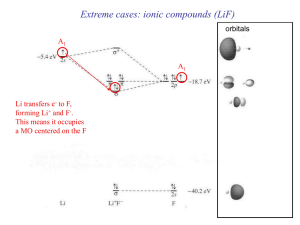
... that do not change =1; and orbitals that remain in the same position but change sign = -1) 4. Find the irreducible representations (they correspond to the symmetry of group orbitals, also called Symmetry Adapted Linear Combinations SALC’s of the orbitals). 5. Find AO’s in central atom with the same ...
... that do not change =1; and orbitals that remain in the same position but change sign = -1) 4. Find the irreducible representations (they correspond to the symmetry of group orbitals, also called Symmetry Adapted Linear Combinations SALC’s of the orbitals). 5. Find AO’s in central atom with the same ...
Personal Tutoring Help on Questions and Problems
... flowing powder. Calculate the percent composition (d) NH 4NO 2 ¡ N 2 ⫹ H 2O of CaSiO3. (e) NaHCO 3 ¡ Na2CO 3 ⫹ H 2O ⫹ CO 2 3.52 The empirical formula of a compound is CH. If the (f) P4O 10 ⫹ H 2O ¡ H 3PO 4 molar mass of this compound is about 78 g, what is its (g) HCl ⫹ CaCO 3 ¡ CaCl2 ⫹ H 2O ⫹ CO 2 m ...
... flowing powder. Calculate the percent composition (d) NH 4NO 2 ¡ N 2 ⫹ H 2O of CaSiO3. (e) NaHCO 3 ¡ Na2CO 3 ⫹ H 2O ⫹ CO 2 3.52 The empirical formula of a compound is CH. If the (f) P4O 10 ⫹ H 2O ¡ H 3PO 4 molar mass of this compound is about 78 g, what is its (g) HCl ⫹ CaCO 3 ¡ CaCl2 ⫹ H 2O ⫹ CO 2 m ...
Textbook sample chapter
... mass = Ar × number of moles To convert the mass of a substance consisting of molecules to moles, simply substitute Mr for Ar. ...
... mass = Ar × number of moles To convert the mass of a substance consisting of molecules to moles, simply substitute Mr for Ar. ...
Chapter -
... – Mole ratio (divide all moles by the smallest number of moles) – The numbers represent subscripts. • If the numbers are not whole numbers, multiply by some factor to make them whole. ...
... – Mole ratio (divide all moles by the smallest number of moles) – The numbers represent subscripts. • If the numbers are not whole numbers, multiply by some factor to make them whole. ...
Integrated Physics and Chemistry
... between ionic, covalent, and metallic bonds; Compare the properties of substances with different types of bonds Name simple ionic and covalent compounds; Predict the charge of a transition metal cation in an ionic compound; Write chemical formulas for simple ionic compounds; Distinguish a covalent c ...
... between ionic, covalent, and metallic bonds; Compare the properties of substances with different types of bonds Name simple ionic and covalent compounds; Predict the charge of a transition metal cation in an ionic compound; Write chemical formulas for simple ionic compounds; Distinguish a covalent c ...
4-Pres-Feb-08
... Chemical Equation • C2H5OH + 3 O2 2 CO2 + 3 H2O The equation is balanced and the reaction can be completely stated as: ...
... Chemical Equation • C2H5OH + 3 O2 2 CO2 + 3 H2O The equation is balanced and the reaction can be completely stated as: ...
Document
... Now there are two N atoms and four O atoms on the right. Placing the coefficient 2 in front of NO balances both the number of N atoms and O atoms: O2 + 2 NO → 2 NO2 (balanced) (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each ...
... Now there are two N atoms and four O atoms on the right. Placing the coefficient 2 in front of NO balances both the number of N atoms and O atoms: O2 + 2 NO → 2 NO2 (balanced) (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each ...
Document
... CO2 (37.8 g). This is reasonable because each carbon in CO2 has two oxygen atoms associated with it, while in C6H12O6 each carbon has only one oxygen atom associated with it and two hydrogen atoms, which are much lighter than oxygen. Therefore the mass of glucose produced should be less than the mas ...
... CO2 (37.8 g). This is reasonable because each carbon in CO2 has two oxygen atoms associated with it, while in C6H12O6 each carbon has only one oxygen atom associated with it and two hydrogen atoms, which are much lighter than oxygen. Therefore the mass of glucose produced should be less than the mas ...
(NH 3 ) 2 - GZ @ Science Class Online
... Carry out procedures to identify ions involves collecting primary data and using these observations to identify ions in a solution using a procedure provided. Identification of ions must be supported by experimental observations and identification of all precipitates formed. Ions to be identified wi ...
... Carry out procedures to identify ions involves collecting primary data and using these observations to identify ions in a solution using a procedure provided. Identification of ions must be supported by experimental observations and identification of all precipitates formed. Ions to be identified wi ...
Chem expo 12
... Unit 3 Areas of study Chemical analysis, which looks at the various analytical techniques available to the chemist. The food we eat, the chemicals and fertilisers used to grow this food, the fuels we use for transport and energy and the wide range of medications that we use to prolong and enhance ou ...
... Unit 3 Areas of study Chemical analysis, which looks at the various analytical techniques available to the chemist. The food we eat, the chemicals and fertilisers used to grow this food, the fuels we use for transport and energy and the wide range of medications that we use to prolong and enhance ou ...