Chemistry – 5071
... This syllabus is designed to place less emphasis on factual material and greater emphasis on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need of students to develop skills that will be of long term value in an increasi ...
... This syllabus is designed to place less emphasis on factual material and greater emphasis on the understanding and application of scientific concepts and principles. This approach has been adopted in recognition of the need of students to develop skills that will be of long term value in an increasi ...
NICKEL(II) PINCER COMPLEXES SUPPORTED BY 2,6
... the metal environment, such as chirality or enhanced rigidity. Remote electronic modifications can be achieved by changing the R group, attached to the backbone of the ligand. Additionally, labile ligands and non-coordinating counter anions increase the efficiency of the metal centers. A general str ...
... the metal environment, such as chirality or enhanced rigidity. Remote electronic modifications can be achieved by changing the R group, attached to the backbone of the ligand. Additionally, labile ligands and non-coordinating counter anions increase the efficiency of the metal centers. A general str ...
File - UTeach Dallas Project
... The candidates should be able to demonstrate knowledge and understanding in relation to:(a) scientific phenomena, facts, concepts, theories and laws. (b) scientific terminology, use of symbols, quantities and units. (c) scientific apparatus and instruments and their safe operations. (d) scientific q ...
... The candidates should be able to demonstrate knowledge and understanding in relation to:(a) scientific phenomena, facts, concepts, theories and laws. (b) scientific terminology, use of symbols, quantities and units. (c) scientific apparatus and instruments and their safe operations. (d) scientific q ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Formula Weight/Mass (FW or FM) • A formula weight/Mass is the sum of the atomic weights/masses for the atoms in a chemical formula. • So, the FM or FM of calcium chloride, CaCl2, would be Ca: 1(40.1 amu) + Cl: 2(35.5 amu) 111.1 amu ...
... Formula Weight/Mass (FW or FM) • A formula weight/Mass is the sum of the atomic weights/masses for the atoms in a chemical formula. • So, the FM or FM of calcium chloride, CaCl2, would be Ca: 1(40.1 amu) + Cl: 2(35.5 amu) 111.1 amu ...
Stoichiometric relationships
... You will notice that often the letter or letters used are derived from the English name of the element, but in some cases they derive from other languages. For example, Hg for mercury comes from Latin, whereas W for tungsten has its origin in European dialects. Happily, these symbols are all accepte ...
... You will notice that often the letter or letters used are derived from the English name of the element, but in some cases they derive from other languages. For example, Hg for mercury comes from Latin, whereas W for tungsten has its origin in European dialects. Happily, these symbols are all accepte ...
Organic Chemistry - University of California, Riverside
... With the exception of the protonated amines and alcohols just mentioned, all of the organic molecules that we have considered have no ionic charge so they are electrically neutral. A molecule is electrically neutral because the total number of its electrons (-1 charge) is equal to the number of prot ...
... With the exception of the protonated amines and alcohols just mentioned, all of the organic molecules that we have considered have no ionic charge so they are electrically neutral. A molecule is electrically neutral because the total number of its electrons (-1 charge) is equal to the number of prot ...
C H
... CH 3 CH 3 CH 3 2,3,5-Trimethylhexane (not 2,4,5-Trimethylhexane) Suffixes of some classes of organic compounds ...
... CH 3 CH 3 CH 3 2,3,5-Trimethylhexane (not 2,4,5-Trimethylhexane) Suffixes of some classes of organic compounds ...
Summer Study Assignment – Honors Chem 2/AP Chemistry
... a. How many grams of iron (II) dichromate are required to produce 44.0 grams of carbon dioxide? b. How many grams of oxygen gas are required to produce 100.0 grams of ferric oxide? c. If 300.0 grams of iron (II) dichromate react, how many grams of oxygen gas will be consumed? d. How many grams of ir ...
... a. How many grams of iron (II) dichromate are required to produce 44.0 grams of carbon dioxide? b. How many grams of oxygen gas are required to produce 100.0 grams of ferric oxide? c. If 300.0 grams of iron (II) dichromate react, how many grams of oxygen gas will be consumed? d. How many grams of ir ...
Stoichiometry1
... AgNO3 (aq) + Cu(s) CuNO3(aq) + Ag(s) or the non-metal changes position with the ...
... AgNO3 (aq) + Cu(s) CuNO3(aq) + Ag(s) or the non-metal changes position with the ...
Atomic Polar Tensor Transferabllity and Atomic Charges kr the
... in ref 1. (RtY)represents the center of charge of the h brid orbital (pv),where p and v indicate orbitals of atom A, and R,,YB represents the bonding center of charge since p and v belong to different atoms, A and B, whether chemically bonded or not. These contributions in expression 1 are known, re ...
... in ref 1. (RtY)represents the center of charge of the h brid orbital (pv),where p and v indicate orbitals of atom A, and R,,YB represents the bonding center of charge since p and v belong to different atoms, A and B, whether chemically bonded or not. These contributions in expression 1 are known, re ...
Discussion Questions
... Justify your choice. For those you did not choose, explain why they are incorrect. 13. What is the difference between the empirical and molecular formulas of a compound? Can they ever be the same? Explain. 14. Atoms of three different elements are represented by , , and . Which compound i ...
... Justify your choice. For those you did not choose, explain why they are incorrect. 13. What is the difference between the empirical and molecular formulas of a compound? Can they ever be the same? Explain. 14. Atoms of three different elements are represented by , , and . Which compound i ...
Conversion Problems
... since 181/30.01 = about 6, the molecule is made up of 6 empirical units giving us a molecular formula = 6(CH2O) = C6H12O6 (c) Cadaverine is a molecular compound formed in decomposing fish. It has a molar mass of 102.18 and is composed solely of C, H, and N. When 0.03560 g of cadaverine is combusted ...
... since 181/30.01 = about 6, the molecule is made up of 6 empirical units giving us a molecular formula = 6(CH2O) = C6H12O6 (c) Cadaverine is a molecular compound formed in decomposing fish. It has a molar mass of 102.18 and is composed solely of C, H, and N. When 0.03560 g of cadaverine is combusted ...
Chemistry
... Chemistry is about the study of matter, its interactions and transformations. At a macroscopic level, we observe matter and its interactions everywhere in our daily life. The microscopic level looks at the structure of matter that gives rise to these interactions. At O-Level, students have been intr ...
... Chemistry is about the study of matter, its interactions and transformations. At a macroscopic level, we observe matter and its interactions everywhere in our daily life. The microscopic level looks at the structure of matter that gives rise to these interactions. At O-Level, students have been intr ...
Calculations with Chemical Formulas and Equations
... • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
... • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
DEPARTMENT OF CHEMISTRY, CFS, IIUM
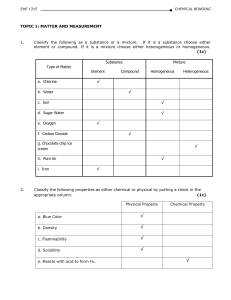
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
... variety of matter is recognized is called a property. A characteristic that depends upon the amount of matter in the sample is called an extensive property. A characteristic that does not depend upon the amount of matter is called an intensive property. A characteristic that can be observed without ...
Appendix N CONCENTRATION UNITS
... Your first task in mastering chemical formulas and nomenclature will be to learn the names and symbols of the most common elements. At this stage of the game, you should concentrate on learning the names and symbols of the elements in the first three rows of the periodic table. Most of the symbols a ...
... Your first task in mastering chemical formulas and nomenclature will be to learn the names and symbols of the most common elements. At this stage of the game, you should concentrate on learning the names and symbols of the elements in the first three rows of the periodic table. Most of the symbols a ...
File
... hydrate is heated. The remaining salt is known as the anhydrous salt. The general reaction for heating a hydrate is: CoCl2·6H2O + heat ...
... hydrate is heated. The remaining salt is known as the anhydrous salt. The general reaction for heating a hydrate is: CoCl2·6H2O + heat ...
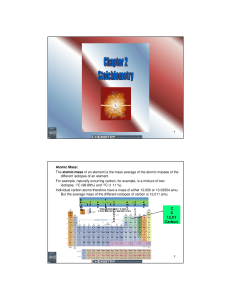
Atomic Mass: The atomic mass of an element is the mass average of
... To calculate the number of particles (atoms, molecules, shoes….etc) use the following formula: Number of particles = number of moles x Avogadro’s number Calculate the number of atoms in 2 mole of hydrogen? Number of hydrogen atoms = 2 moles of H x 6.02214 x 1023 H atom / mole Number of hydrogen atom ...
... To calculate the number of particles (atoms, molecules, shoes….etc) use the following formula: Number of particles = number of moles x Avogadro’s number Calculate the number of atoms in 2 mole of hydrogen? Number of hydrogen atoms = 2 moles of H x 6.02214 x 1023 H atom / mole Number of hydrogen atom ...
Chemical Reactions
... Convert this to a formula equation Solid iron (III) sulfide reacts with gaseous hydrogen chloride to form iron (II) chloride and hydrogen ...
... Convert this to a formula equation Solid iron (III) sulfide reacts with gaseous hydrogen chloride to form iron (II) chloride and hydrogen ...
Common Curriculum Map Discipline: Science Course: Chemistry
... 2. Differentiate between cations and anions. 3. List some common properties of ionic compounds. 4. State the octet rule. 5. Apply the octet rule to determine the type of ion each element is most likely to form. 6. Draw a Lewis Dot structure for a given atom. 7. Draw a Lewis Dot diagram to show the e ...
... 2. Differentiate between cations and anions. 3. List some common properties of ionic compounds. 4. State the octet rule. 5. Apply the octet rule to determine the type of ion each element is most likely to form. 6. Draw a Lewis Dot structure for a given atom. 7. Draw a Lewis Dot diagram to show the e ...
Li K-edge XANES and Li(1s) XPS Spectra of Lithium Compounds
... though they have still some difficulties, for example, X-ray source for XANES and low sensitivities for both methods. In this study, Li K-edge XANES and Li(1s) XPS spectra of various lithium compounds and complexes have been measured to get directly the information on electronic states of lithium io ...
... though they have still some difficulties, for example, X-ray source for XANES and low sensitivities for both methods. In this study, Li K-edge XANES and Li(1s) XPS spectra of various lithium compounds and complexes have been measured to get directly the information on electronic states of lithium io ...
Learning Outcomes
... (e) deduce from the given melting point and boiling point the identities of substances and their purity ......................................................................................................................................... 13 (f) explain that the measurement of purity in substance ...
... (e) deduce from the given melting point and boiling point the identities of substances and their purity ......................................................................................................................................... 13 (f) explain that the measurement of purity in substance ...
semester i - Pt. Ravishankar Shukla University
... 1) Basic Radicals : Ag, Pb, Hg, Bi, Cu, Cd, As, Sb, Sn, Fe, Al, Cr, Zn, Mn, Co, Ni, Ba, Sr, Ca, Mg, Na, K, Ce, Th, Zr, W, Te, Ti, Mo, U, V, Be, Li, Au, Pt. 2) Acid Radicals : Carbonate, Sulphite, Sulphide, Nitrite, Nitrate, Acetate, Flouride. Chloride, Bromide, Iodide, Sulphate, Borate, Oxalate, Pho ...
... 1) Basic Radicals : Ag, Pb, Hg, Bi, Cu, Cd, As, Sb, Sn, Fe, Al, Cr, Zn, Mn, Co, Ni, Ba, Sr, Ca, Mg, Na, K, Ce, Th, Zr, W, Te, Ti, Mo, U, V, Be, Li, Au, Pt. 2) Acid Radicals : Carbonate, Sulphite, Sulphide, Nitrite, Nitrate, Acetate, Flouride. Chloride, Bromide, Iodide, Sulphate, Borate, Oxalate, Pho ...
Metal Complexes Containing Natural and Artificial Radioactive
... compounds. Actinides form various Anm+ (m = 2–4) and AnO2m+ (m = 1, 2) ions containing only f electrons. The shielding by f electrons causes the contraction of the An3+ ions and the magnitude of the actinide contraction along the series to be parallel to that of the lanthanide contraction. Differenc ...
... compounds. Actinides form various Anm+ (m = 2–4) and AnO2m+ (m = 1, 2) ions containing only f electrons. The shielding by f electrons causes the contraction of the An3+ ions and the magnitude of the actinide contraction along the series to be parallel to that of the lanthanide contraction. Differenc ...