Construction of Porous Solids from Hydrogen
... M3(BTC)2‚12H2O (M ) Co, Ni, and Zn). Identical synthetic procedures were used to prepare the three compounds, that of the cobalt compound is described here in detail. An aqueous mixture (15 mL) of cobalt(II) acetate tetrahydrate (0.41 g, 1.65 mmol) and the acid form of BTC (BTCH3) (0.20 g, 0.95 mmol ...
... M3(BTC)2‚12H2O (M ) Co, Ni, and Zn). Identical synthetic procedures were used to prepare the three compounds, that of the cobalt compound is described here in detail. An aqueous mixture (15 mL) of cobalt(II) acetate tetrahydrate (0.41 g, 1.65 mmol) and the acid form of BTC (BTCH3) (0.20 g, 0.95 mmol ...
Prep UK-intro.p65
... B(III), Al(III), Si(IV), P(V) S(IV), S(VI), O(II), F(I), Cl(I), Cl(III), Cl(V) and Cl(VIII) are normal oxidation states of 2nd and 3rd row elements in compounds with halogens and in oxoanions ...
... B(III), Al(III), Si(IV), P(V) S(IV), S(VI), O(II), F(I), Cl(I), Cl(III), Cl(V) and Cl(VIII) are normal oxidation states of 2nd and 3rd row elements in compounds with halogens and in oxoanions ...
HYBRID MULTIDENTATE PHOSPHINE
... synthetic chemists are continuously looking for new methodologies for the synthesis of these compounds. The principal focus of this thesis is the design of novel ligands for potential use in homogenous catalysis. ...
... synthetic chemists are continuously looking for new methodologies for the synthesis of these compounds. The principal focus of this thesis is the design of novel ligands for potential use in homogenous catalysis. ...
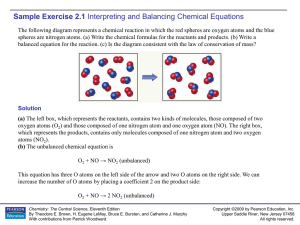
Sample Exercise 2.1
... Now there are two N atoms and four O atoms on the right. Placing the coefficient 2 in front of NO balances both the number of N atoms and O atoms: O2 + 2 NO → 2 NO2 (balanced) (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each ...
... Now there are two N atoms and four O atoms on the right. Placing the coefficient 2 in front of NO balances both the number of N atoms and O atoms: O2 + 2 NO → 2 NO2 (balanced) (c) The left box (reactants) contains four O2 molecules and eight NO molecules. Thus, the molecular ratio is one O2 for each ...
SCHOOL OF CHEMICAL SCIENCES
... To produce chemistry graduates who are knowledgeable, highly skilled, wellmannered and possess excellent work ethics suited for the requirements of the public and industrial sectors. To provide quality education and chemistry students. To instill awareness among chemistry students towards the welfar ...
... To produce chemistry graduates who are knowledgeable, highly skilled, wellmannered and possess excellent work ethics suited for the requirements of the public and industrial sectors. To provide quality education and chemistry students. To instill awareness among chemistry students towards the welfar ...
- Mendeley Data
... +4 and +5 are the most stable ones under aerobic conditions), (c) the Lewis acid character of the vanadium centers and (d) the high affinity of the metal toward oxygen [22-24]. Since epoxides are the key starting materials for a wide variety of products, the epoxidation of alkenes is considered to ...
... +4 and +5 are the most stable ones under aerobic conditions), (c) the Lewis acid character of the vanadium centers and (d) the high affinity of the metal toward oxygen [22-24]. Since epoxides are the key starting materials for a wide variety of products, the epoxidation of alkenes is considered to ...
CHEMISTRY 133 LECTURE / STUDY GUIDE FOR R.H. LANGLEY
... in the end. For example, you will not be able to do most of Chapter 6 without knowing the Factor-Label Method, and if you wait until then to learn it you will be impossibly far behind. Do not waste your time learning extra conversions. The time that these extras will save you is time lost from study ...
... in the end. For example, you will not be able to do most of Chapter 6 without knowing the Factor-Label Method, and if you wait until then to learn it you will be impossibly far behind. Do not waste your time learning extra conversions. The time that these extras will save you is time lost from study ...
To do List
... oxygen atoms, and 3.25 x 10 atoms of hydrogen. If the molecular weight of this compound is known to be 363 grams/mole, what its true molecular formula? ...
... oxygen atoms, and 3.25 x 10 atoms of hydrogen. If the molecular weight of this compound is known to be 363 grams/mole, what its true molecular formula? ...
Chemical Reactions and Equations - 2012 Book Archive
... Unfortunately, it is also an incomplete chemical equation. The law of conservation of matter says that matter cannot be created or destroyed. In chemical equations, the number of atoms of each element in the reactants must be the same as the number of atoms of each element in the products. If we cou ...
... Unfortunately, it is also an incomplete chemical equation. The law of conservation of matter says that matter cannot be created or destroyed. In chemical equations, the number of atoms of each element in the reactants must be the same as the number of atoms of each element in the products. If we cou ...
chemistry - Textbooks Online
... Alchemy was a mixture of scientific investigation and mystical quest, with strands of philosophy from Greece, China, Egypt and Arabia mixed in. The main aims of alchemy that emerged with time were the quest for the elixir of life (the drinking of which would endue the alchemist with immortality), an ...
... Alchemy was a mixture of scientific investigation and mystical quest, with strands of philosophy from Greece, China, Egypt and Arabia mixed in. The main aims of alchemy that emerged with time were the quest for the elixir of life (the drinking of which would endue the alchemist with immortality), an ...
BS Chemistry - Government College University Faisalabad
... of resonance; hypercohjugation; tautomerism; hydrogen bonding. Nomenclature of organic compounds Common and trivial name of organic compounds; and introduction to the systematic nomenclature of mono and bi-bunfctional organic compounds by IUPAC rules. Hydrocarbons Open Chain preparation, properties ...
... of resonance; hypercohjugation; tautomerism; hydrogen bonding. Nomenclature of organic compounds Common and trivial name of organic compounds; and introduction to the systematic nomenclature of mono and bi-bunfctional organic compounds by IUPAC rules. Hydrocarbons Open Chain preparation, properties ...
Chapter 8 Concepts of Chemical Bonding
... charges, the greater the energy and (b) the farther apart the ions are, the lower the energy. Solve NaF consists of Na+ and F− ions, CsI of Cs+ and I− ions, and CaO of Ca2+ and O2− ions. Because the product Q1Q2 appears in the numerator of Equation 8.4, the lattice energy increases dramatically when ...
... charges, the greater the energy and (b) the farther apart the ions are, the lower the energy. Solve NaF consists of Na+ and F− ions, CsI of Cs+ and I− ions, and CaO of Ca2+ and O2− ions. Because the product Q1Q2 appears in the numerator of Equation 8.4, the lattice energy increases dramatically when ...
Table of Contents - slccscience`s Home Page
... and its compounds. Organic chemistry is the study of carbon and its compounds. Since there are 117 known elements, it often seems odd that an entire branch of chemistry is devoted to a single element and its compounds while the other 116 elements and their compounds are all lumped together in a sepa ...
... and its compounds. Organic chemistry is the study of carbon and its compounds. Since there are 117 known elements, it often seems odd that an entire branch of chemistry is devoted to a single element and its compounds while the other 116 elements and their compounds are all lumped together in a sepa ...
Matter Flashcards 5 - Henrico County Public Schools
... No, the properties of the compound are different from those of the elements that make it up. For instance, Sodium (Na) is an explosive substance and Chlorine (Cl) is a poisonous gas, but combined together, they make NaCl, or table salt, which is not poisonous or explosive. SOL 5.4 ...
... No, the properties of the compound are different from those of the elements that make it up. For instance, Sodium (Na) is an explosive substance and Chlorine (Cl) is a poisonous gas, but combined together, they make NaCl, or table salt, which is not poisonous or explosive. SOL 5.4 ...
Net ionic equation
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
... The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is because the water molecules have a par ...
CHAPTER 21 NONMETALLIC ELEMENTS AND THEIR COMPOUNDS
... The density of a gas depends on temperature, pressure, and the molar mass of the substance. When two gases are at the same pressure and temperature, the ratio of their densities should be the same as the ratio of their molar masses. The molar mass of ammonium chloride is 53.5 g/mol, and the ratio of ...
... The density of a gas depends on temperature, pressure, and the molar mass of the substance. When two gases are at the same pressure and temperature, the ratio of their densities should be the same as the ratio of their molar masses. The molar mass of ammonium chloride is 53.5 g/mol, and the ratio of ...
Stoichiometry - HCC Learning Web
... • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles. • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound. Stoichiometry © 2015 Pearson Education, Inc. ...
... • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles. • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound. Stoichiometry © 2015 Pearson Education, Inc. ...
Chapter 03 - KFUPM Faculty List
... Factors like 3/2 are ok, but usually not very much liked. So we multiply what we have (especially the 3/2) by 2: 2 NH3 + 2 O2 2 NO + 3 H2O: now N and H ok, but there are 4O atoms on the left and 5O atoms on the right so when we use (5/2) O2, then we have 5O atoms on the left and on the right side, ...
... Factors like 3/2 are ok, but usually not very much liked. So we multiply what we have (especially the 3/2) by 2: 2 NH3 + 2 O2 2 NO + 3 H2O: now N and H ok, but there are 4O atoms on the left and 5O atoms on the right so when we use (5/2) O2, then we have 5O atoms on the left and on the right side, ...
Has the Periodic Table Been Successfully Axiomatized?
... which they make following their axiomatization of the Periodic Table. If there is any sense in which one may distinguish between a naive and a sophisticated version of the periodic table, it lies with the use of atomic weight and later the property of atomic number to order the elements. Whereas ato ...
... which they make following their axiomatization of the Periodic Table. If there is any sense in which one may distinguish between a naive and a sophisticated version of the periodic table, it lies with the use of atomic weight and later the property of atomic number to order the elements. Whereas ato ...
chem - CBSE Guess
... Rancidity: The oily and fatty food oxidizes and give bad smell and test is called rancidity.Preventatioin:By adding antioxidant which slow down the process of oxidation.2. Vaccum packing,3Flusing N2 gas in chips packets.3.Refrigeration. Q.Explain the various types of reactions with one example of ea ...
... Rancidity: The oily and fatty food oxidizes and give bad smell and test is called rancidity.Preventatioin:By adding antioxidant which slow down the process of oxidation.2. Vaccum packing,3Flusing N2 gas in chips packets.3.Refrigeration. Q.Explain the various types of reactions with one example of ea ...
mole - hrsbstaff.ednet.ns.ca
... This reaction requires two molecules of hydrogen and one molecule of oxygen. To carry out this chemical reaction a chemist must mix hydrogen and oxygen together in the correct ratio (2:1). How does the chemist know when the correct ratio is present? These molecules are too small to see so we cannot ...
... This reaction requires two molecules of hydrogen and one molecule of oxygen. To carry out this chemical reaction a chemist must mix hydrogen and oxygen together in the correct ratio (2:1). How does the chemist know when the correct ratio is present? These molecules are too small to see so we cannot ...
6 theoretical problems 2 practical problems
... 3.2 Write molecular formulas for all possible ionic compounds which comply with the following conditions: each compound has 1) empirical formula Pt(NH3)2Cl2, 2) an anion and a cation and is composed of discrete, monomeric square planar platinum(II) complex, 3) only one type of cation and one type of ...
... 3.2 Write molecular formulas for all possible ionic compounds which comply with the following conditions: each compound has 1) empirical formula Pt(NH3)2Cl2, 2) an anion and a cation and is composed of discrete, monomeric square planar platinum(II) complex, 3) only one type of cation and one type of ...
Exemplar Paper
... 2. When dilute hydrochloric acid is added to a solution of sodium thiosulfate in a beaker, solid sulfur forms in the solution. Na2S2O3(aq) + 2HCl(aq) → 2NaCl(aq) + SO2(aq) + S(s) + H2O(ℓ) The effect of concentration on the rate of reaction can be studied by varying the sodium thiosulfate concentra ...
... 2. When dilute hydrochloric acid is added to a solution of sodium thiosulfate in a beaker, solid sulfur forms in the solution. Na2S2O3(aq) + 2HCl(aq) → 2NaCl(aq) + SO2(aq) + S(s) + H2O(ℓ) The effect of concentration on the rate of reaction can be studied by varying the sodium thiosulfate concentra ...
ordinary level chemistry syllabus
... In line with efforts to improve the quality of education, the government of Rwanda emphasises the importance of aligning the syllabus, teaching and learning and assessment approaches in order to ensure that the system is producing the kind of citizens the country needs. Many factors influence what c ...
... In line with efforts to improve the quality of education, the government of Rwanda emphasises the importance of aligning the syllabus, teaching and learning and assessment approaches in order to ensure that the system is producing the kind of citizens the country needs. Many factors influence what c ...