File
... 37. The hydrides formed by the transfer of electrons from electropositive metals to hydrogen are called __________. (Ionic hydrides, covalent hydrides, Complex hydrides, Interstitial hydrides) 38. NaH is an example of __________. (Ionic hydrides, covalent hydrides, Complex hydrides, Interstitial hyd ...
... 37. The hydrides formed by the transfer of electrons from electropositive metals to hydrogen are called __________. (Ionic hydrides, covalent hydrides, Complex hydrides, Interstitial hydrides) 38. NaH is an example of __________. (Ionic hydrides, covalent hydrides, Complex hydrides, Interstitial hyd ...
Photogeneration of Hydride Donors and Their Use Toward CO2
... 1. Fujita, E.; Brunschwig, B. S.; Creutz, C.; Muckerman, J. T.; Sutin, N.; Szalda, D. J.; van Eldik, R. “Transition State Characterization for the Reversible Binding of Dihydrogen to Bis(2,2'-bipyridine)rhodium(I) from Temperature- and Pressure-Dependent Experimental and Theoretical Studies” Inorg. ...
... 1. Fujita, E.; Brunschwig, B. S.; Creutz, C.; Muckerman, J. T.; Sutin, N.; Szalda, D. J.; van Eldik, R. “Transition State Characterization for the Reversible Binding of Dihydrogen to Bis(2,2'-bipyridine)rhodium(I) from Temperature- and Pressure-Dependent Experimental and Theoretical Studies” Inorg. ...
Unit 2 Powerpoint Notes
... • The final step in writing correct chemical equations is to make sure the law of conservation of mass is satisfied. – The numbers and types of atoms on both sides of the equation must be the same – this is called balancing an equation. – Equations are balanced by inserting coefficients - whole numb ...
... • The final step in writing correct chemical equations is to make sure the law of conservation of mass is satisfied. – The numbers and types of atoms on both sides of the equation must be the same – this is called balancing an equation. – Equations are balanced by inserting coefficients - whole numb ...
REDOX EQUILIBRIA SL - chemistryatdulwich
... The oxidation number or the oxidation state of an element in a compound indicates the number of electrons over which an atom/ion of an element has gained or lost control during a reaction. Oxidation numbers are used to keep track of how many electrons are lost or gained by each atom or ion. They are ...
... The oxidation number or the oxidation state of an element in a compound indicates the number of electrons over which an atom/ion of an element has gained or lost control during a reaction. Oxidation numbers are used to keep track of how many electrons are lost or gained by each atom or ion. They are ...
the Main-Group Metals - McQuarrie General Chemistry
... The alkaline-earth metals—beryllium, magnesium, calcium, strontium, barium, and radium—occur in Group 2 in the periodic table (Figure I.1). Beryllium is a relatively rare element but occurs as localized surface deposits in the mineral beryl (Figure I.2). Essentially unlimited quantities of magnesium ...
... The alkaline-earth metals—beryllium, magnesium, calcium, strontium, barium, and radium—occur in Group 2 in the periodic table (Figure I.1). Beryllium is a relatively rare element but occurs as localized surface deposits in the mineral beryl (Figure I.2). Essentially unlimited quantities of magnesium ...
SCH 4U REVIEW Notes
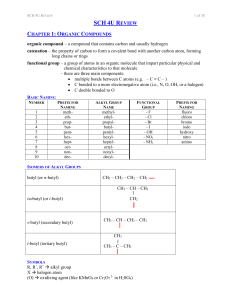
... PRIORITY FOR NAMING (FROM HIGHEST TO LOWEST) OH hydroxyl NH2 amino F, Cl, Br, I fluoro, chloro, bromo, iodo CH2CH2CH3 propyl CH2CH3 ethyl CH3 methyl ...
... PRIORITY FOR NAMING (FROM HIGHEST TO LOWEST) OH hydroxyl NH2 amino F, Cl, Br, I fluoro, chloro, bromo, iodo CH2CH2CH3 propyl CH2CH3 ethyl CH3 methyl ...
CHEM 121 Chp 5 Spaulding
... Thus, a redox reaction always has two components, one that is oxidized and one that is reduced A redox reaction involves the transfer of electrons from one element to another. ...
... Thus, a redox reaction always has two components, one that is oxidized and one that is reduced A redox reaction involves the transfer of electrons from one element to another. ...
Topic 1: Quantitative chemistry
... Strictly speaking we can not use the term molecular formula when dealing with giant structures as they do not consist of separate molecules; in that case any formula is always an empirical formula indicating the most simple ratio of ions or atoms e.g. in the case of silicon dioxide, SiO2, this formu ...
... Strictly speaking we can not use the term molecular formula when dealing with giant structures as they do not consist of separate molecules; in that case any formula is always an empirical formula indicating the most simple ratio of ions or atoms e.g. in the case of silicon dioxide, SiO2, this formu ...
Chapter 5: Calculations and the Chemical Equation
... determining the composition. For example, dihydrogen monoxide (water, H2O) is a compound composed of two hydrogen atoms for every oxygen atom. The Chemical Formula A chemical formula (also called molecular formula) is a concise way of expressing information about the atoms that constitute a particul ...
... determining the composition. For example, dihydrogen monoxide (water, H2O) is a compound composed of two hydrogen atoms for every oxygen atom. The Chemical Formula A chemical formula (also called molecular formula) is a concise way of expressing information about the atoms that constitute a particul ...
1.2 Calculations
... DEFINITION: The mole is the amount of substance in grams that has the same number of particles as there are atoms in 12 grams of carbon-12. DEFINITION: Relative atomic mass is the average mass of one atom compared to one twelfth of the mass of one atom of carbon-12 DEFINITION: Relative molecular mas ...
... DEFINITION: The mole is the amount of substance in grams that has the same number of particles as there are atoms in 12 grams of carbon-12. DEFINITION: Relative atomic mass is the average mass of one atom compared to one twelfth of the mass of one atom of carbon-12 DEFINITION: Relative molecular mas ...
Stoichiometry
... • When balancing an equation, ONLY the coefficients can be changed. • NEVER change the subscripts. • For example: 3H2O 3 is the coefficient. 2 and 1 are the subscripts. • Changing the subscripts changes the compound. H2O2 is not water but hydrogen peroxide. ...
... • When balancing an equation, ONLY the coefficients can be changed. • NEVER change the subscripts. • For example: 3H2O 3 is the coefficient. 2 and 1 are the subscripts. • Changing the subscripts changes the compound. H2O2 is not water but hydrogen peroxide. ...
12 U Chem Review
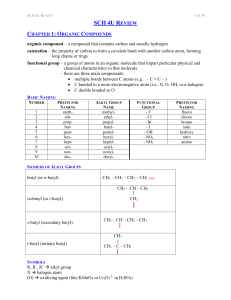
... PRIORITY FOR NAMING (FROM HIGHEST TO LOWEST) OH hydroxyl NH2 amino F, Cl, Br, I fluoro, chloro, bromo, iodo CH2CH2CH3 propyl CH2CH3 ethyl CH3 methyl ...
... PRIORITY FOR NAMING (FROM HIGHEST TO LOWEST) OH hydroxyl NH2 amino F, Cl, Br, I fluoro, chloro, bromo, iodo CH2CH2CH3 propyl CH2CH3 ethyl CH3 methyl ...
sch4ureview
... PRIORITY FOR NAMING (FROM HIGHEST TO LOWEST) OH hydroxyl NH2 amino F, Cl, Br, I fluoro, chloro, bromo, iodo CH2CH2CH3 propyl CH2CH3 ethyl CH3 methyl ...
... PRIORITY FOR NAMING (FROM HIGHEST TO LOWEST) OH hydroxyl NH2 amino F, Cl, Br, I fluoro, chloro, bromo, iodo CH2CH2CH3 propyl CH2CH3 ethyl CH3 methyl ...
Topic 1 - Coral Gables Senior High
... You will notice that often the letter or letters used are derived from the English name of the element, but in some cases they derive from other languages. For example, Hg for mercury comes from Latin, whereas W for tungsten has its origin in European dialects. Happily, these symbols are all accepte ...
... You will notice that often the letter or letters used are derived from the English name of the element, but in some cases they derive from other languages. For example, Hg for mercury comes from Latin, whereas W for tungsten has its origin in European dialects. Happily, these symbols are all accepte ...
GCE Getting Started - Edexcel
... Know the relative mass and relative charge of protons, neutrons and electrons. Know what is meant by the terms ‘atomic (proton) number’ and ‘mass number’. Be able to determine the number of each type of subatomic particle in an atom, molecule or ion from the atomic (proton) number and mass number. U ...
... Know the relative mass and relative charge of protons, neutrons and electrons. Know what is meant by the terms ‘atomic (proton) number’ and ‘mass number’. Be able to determine the number of each type of subatomic particle in an atom, molecule or ion from the atomic (proton) number and mass number. U ...
Topic 4 - Lloyd Crosby
... (c) Known coordination numbers range from two to eight. (2) Rule of thumb The coordination number will be double the charge on the metal ion. ...
... (c) Known coordination numbers range from two to eight. (2) Rule of thumb The coordination number will be double the charge on the metal ion. ...
1ST CHAPTER Long-questions-basic-concept
... their natural abundance. Natural abundances of atoms are given as atomic percentages. The mass contributed by each isotope is equal to fractional abundance multiplied by the isotopic mass. The average or fractional atomic mass for the element is obtained by taking the sum of the masses contributed b ...
... their natural abundance. Natural abundances of atoms are given as atomic percentages. The mass contributed by each isotope is equal to fractional abundance multiplied by the isotopic mass. The average or fractional atomic mass for the element is obtained by taking the sum of the masses contributed b ...
3: Haloalkanes, Alcohols, Ethers, and Amines
... With the exception of the protonated amines and alcohols just mentioned, all of the organic molecules that we have considered have no ionic charge so they are electrically neutral. A molecule is electrically neutral because the total number of its electrons (-1 charge) is equal to the number of prot ...
... With the exception of the protonated amines and alcohols just mentioned, all of the organic molecules that we have considered have no ionic charge so they are electrically neutral. A molecule is electrically neutral because the total number of its electrons (-1 charge) is equal to the number of prot ...
Chapter 4 Chemical Quantities and Aqueous Reactions
... • Predicting whether a compound will dissolve in water is not easy • The best way to do it is to do some experiments to test whether a compound will dissolve in water, then develop some rules based on those experimental results we call this method the empirical method Tro, Chemistry: A Molecular Ap ...
... • Predicting whether a compound will dissolve in water is not easy • The best way to do it is to do some experiments to test whether a compound will dissolve in water, then develop some rules based on those experimental results we call this method the empirical method Tro, Chemistry: A Molecular Ap ...
Chemical Equations and Reactions
... mass of products for any given chemical reaction. Chemical reactions are described by chemical equations. A chemical equation represents, with symbols and formulas, the identities and relative amounts of the reactants and products in a chemical reaction. For example, the following chemical equation ...
... mass of products for any given chemical reaction. Chemical reactions are described by chemical equations. A chemical equation represents, with symbols and formulas, the identities and relative amounts of the reactants and products in a chemical reaction. For example, the following chemical equation ...
380 KB / 39 pages
... (b) When a bottle of milk left too long in the refrigerator turns sour, chemical reactions have occurred. New compounds (some of which taste and/or smell bad) have been formed, so souring of milk is a chemical reaction. (c) When equal volumes of solutions of blue food coloring and yellow food colori ...
... (b) When a bottle of milk left too long in the refrigerator turns sour, chemical reactions have occurred. New compounds (some of which taste and/or smell bad) have been formed, so souring of milk is a chemical reaction. (c) When equal volumes of solutions of blue food coloring and yellow food colori ...
Title
... • Topics in the first semester. • They cover the main parts of general and organic chemistry • Form: short revision of a chapter of general chemistry + practicing chemical calculations • revision of the essentials of organic chemistry ...
... • Topics in the first semester. • They cover the main parts of general and organic chemistry • Form: short revision of a chapter of general chemistry + practicing chemical calculations • revision of the essentials of organic chemistry ...
Unit5C - OCCC.edu
... Rules for Oxidation Numbers • Oxidation numbers are always reported for individual atoms or ions not groups of atoms or ions!!!!!!!!!!! • For an atom in its elemental form, the oxidation number is always zero. – H2: oxidation # = 0 for each H atom – Cu: oxidation number = 0 – Cl2: oxidation # = 0 f ...
... Rules for Oxidation Numbers • Oxidation numbers are always reported for individual atoms or ions not groups of atoms or ions!!!!!!!!!!! • For an atom in its elemental form, the oxidation number is always zero. – H2: oxidation # = 0 for each H atom – Cu: oxidation number = 0 – Cl2: oxidation # = 0 f ...