Matter
... • Solute – substance that is dissolved. A solute is soluble, or able to dissolve. • A substance that is insoluble is unable to dissolve, forms a mixture that is not homogeneous, and therefore NOT a solution. ...
... • Solute – substance that is dissolved. A solute is soluble, or able to dissolve. • A substance that is insoluble is unable to dissolve, forms a mixture that is not homogeneous, and therefore NOT a solution. ...
Biochemistry Introduction day 1
... Isotopes: Atoms of an element that have the same number of protons but a different number of neutrons. Ex: Oxygen usually has 8 neutrons but 9 and 10 neutrons can be found in some oxygen atoms. Some isotopes are unstable in the nucleus which makes it more likely to decay and release energy. This i ...
... Isotopes: Atoms of an element that have the same number of protons but a different number of neutrons. Ex: Oxygen usually has 8 neutrons but 9 and 10 neutrons can be found in some oxygen atoms. Some isotopes are unstable in the nucleus which makes it more likely to decay and release energy. This i ...
Study Island Copyright © 2012 Study Island
... A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are different than the properties of its elements. D. The compound shares ...
... A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are different than the properties of its elements. D. The compound shares ...
Chapter One Outline
... Chemical reactions are usually accompanied by transfers of energy Substance and Mixtures A heterogeneous mixture is one in which properties in one region are different from properties in another region A homogeneous mixture, or a solution, is completely uniform and consists of two or more substances ...
... Chemical reactions are usually accompanied by transfers of energy Substance and Mixtures A heterogeneous mixture is one in which properties in one region are different from properties in another region A homogeneous mixture, or a solution, is completely uniform and consists of two or more substances ...
24 Sept 08 - Seattle Central College
... • Name the following compound: MnO2 manganese(IV) oxide ...
... • Name the following compound: MnO2 manganese(IV) oxide ...
MatterPP4
... elements that are chemically combined. Most compounds have totally different properties from the elements of which they are composed. Chemical bonds are the forces that hold the elements together in a compound creating a state of stability. ...
... elements that are chemically combined. Most compounds have totally different properties from the elements of which they are composed. Chemical bonds are the forces that hold the elements together in a compound creating a state of stability. ...
AP Chemistry
... CH3Cl = 1 carbon, 3 hydrogen, 1 chlorine Some elements actually exist in nature as ...
... CH3Cl = 1 carbon, 3 hydrogen, 1 chlorine Some elements actually exist in nature as ...
Matter Classification
... • The purest substance of matter • There is only one type of atom for every element. In other words, all carbon atoms are identical to other carbon atoms; all aluminum atoms are identical to all aluminum atoms. ...
... • The purest substance of matter • There is only one type of atom for every element. In other words, all carbon atoms are identical to other carbon atoms; all aluminum atoms are identical to all aluminum atoms. ...
Unit 3 Test - hrsbstaff.ednet.ns.ca
... ___ Combustibility is the ability of a substance to react with acids ___ Sugar disappearing in water is an example of a solution ___ Raisins in Raisin Bran are an example of a solution ___ Lighting a test tube of acetylene gas is an example of a reaction with acid ___ Lighting a test tube of acetyle ...
... ___ Combustibility is the ability of a substance to react with acids ___ Sugar disappearing in water is an example of a solution ___ Raisins in Raisin Bran are an example of a solution ___ Lighting a test tube of acetylene gas is an example of a reaction with acid ___ Lighting a test tube of acetyle ...
Chapter 2 - Speedway High School
... • A cation is a positively charged ion • An ionic bond is an attraction between an anion and a cation ...
... • A cation is a positively charged ion • An ionic bond is an attraction between an anion and a cation ...
Topic 3&4 Atoms and the per.table
... Units 3 and 4 Revision. Q8. Identify the element (a) with an atomic number of 12. (b) with an electron arrangement of 2,5 (c) which has 9 electrons in its atoms. ...
... Units 3 and 4 Revision. Q8. Identify the element (a) with an atomic number of 12. (b) with an electron arrangement of 2,5 (c) which has 9 electrons in its atoms. ...
Document
... Inorganic Nomenclature • Write the name of the cation. • If the anion is an monoatomic ion, change its ending to -ide; if the anion is a polyatomic ion … do you want to memorize 400+ of these? • If the cation can have more than one possible charge, write the charge as a Roman numeral in parentheses ...
... Inorganic Nomenclature • Write the name of the cation. • If the anion is an monoatomic ion, change its ending to -ide; if the anion is a polyatomic ion … do you want to memorize 400+ of these? • If the cation can have more than one possible charge, write the charge as a Roman numeral in parentheses ...
4. bonding - New Hartford Central Schools
... Look up the name of the polyatomic ion on chart table E When polyatomic ion is second 1. write the name of the first element 2. write the name of the polyatomic ion Na2SO4 Sodium sulfate If polyatomic ion first 1. look up ion name 2. follow ending for binary compounds ...
... Look up the name of the polyatomic ion on chart table E When polyatomic ion is second 1. write the name of the first element 2. write the name of the polyatomic ion Na2SO4 Sodium sulfate If polyatomic ion first 1. look up ion name 2. follow ending for binary compounds ...
S1-2-02: What is the basic subatomic structure of an atom?
... S1-0-04: What symbols and systems help people use chemicals safely at home, work and for the environment? 4. What are some similarities and differences between WHMIS and HHPS? ...
... S1-0-04: What symbols and systems help people use chemicals safely at home, work and for the environment? 4. What are some similarities and differences between WHMIS and HHPS? ...
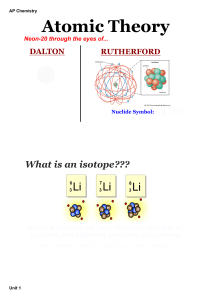
The Atomic Theory of Matter
... • The rest of the subatomic particles were found when scientists made theories on where the electrons were in an atom. In 1910, a scientist named Rutherford examined the effects of passing alpha rays through a gold foil a few thousand atoms thick. He found that most passed right through the gold foi ...
... • The rest of the subatomic particles were found when scientists made theories on where the electrons were in an atom. In 1910, a scientist named Rutherford examined the effects of passing alpha rays through a gold foil a few thousand atoms thick. He found that most passed right through the gold foi ...
Chapter 4.1 and 4.2 - science-b
... In a chemical reaction, one substance changes to another by reorganizing the way the atoms are attached to each other ...
... In a chemical reaction, one substance changes to another by reorganizing the way the atoms are attached to each other ...
Chapter One
... Negative ions - anions one or more electrons more than neutral 23 F , O , N 23- - polyatomic anions SO4 , PO4 Cations and anions can combine to form electroneutral ionic compounds ...
... Negative ions - anions one or more electrons more than neutral 23 F , O , N 23- - polyatomic anions SO4 , PO4 Cations and anions can combine to form electroneutral ionic compounds ...
ChLM Final Review Name: Period: Base Knowledge 1. Classify the
... 22. Draw arrows showing where periods and groups are on the periodic table. ...
... 22. Draw arrows showing where periods and groups are on the periodic table. ...
Review Sheet: Unit 6 Name__________________ CHEMISTRY: A
... compounds are composed of a metal and a nonmetal while ____________ compounds are formed between nonmetals. In formulas for binary ionic compounds, the ____________ ion (or ____________) is always written first and named first. The ____________ ion (or ...
... compounds are composed of a metal and a nonmetal while ____________ compounds are formed between nonmetals. In formulas for binary ionic compounds, the ____________ ion (or ____________) is always written first and named first. The ____________ ion (or ...
ChemFinalgeocities
... a. AlBr3 c. Al3Br9 b. Al2Br6 d. Al4Br12 Based on its position in the periodic table, the most likely charge of an iodide ion is _____. a. 1+ c. 2+ b. 1d. 7Which of the following formulas is incorrect? a. Al2(SO4)3 c. Ca(OH)2 b. AlOH3 d. (NH4)2S The correct name for Fe2S3 is _____. a. iron(III) sulfi ...
... a. AlBr3 c. Al3Br9 b. Al2Br6 d. Al4Br12 Based on its position in the periodic table, the most likely charge of an iodide ion is _____. a. 1+ c. 2+ b. 1d. 7Which of the following formulas is incorrect? a. Al2(SO4)3 c. Ca(OH)2 b. AlOH3 d. (NH4)2S The correct name for Fe2S3 is _____. a. iron(III) sulfi ...