CHAPTER 2 ATOMS, MOLECULES, AND IONS 1 CHAPTER TWO
... a. The smaller parts are electrons and the nucleus. The nucleus is broken down into protons and neutrons which can be broken down into quarks. For our purpose, electrons, neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isot ...
... a. The smaller parts are electrons and the nucleus. The nucleus is broken down into protons and neutrons which can be broken down into quarks. For our purpose, electrons, neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isot ...
Chapter 4 Notes - Atomic Theory
... Ca = calcium; N = nitrogen Drop the end of the anion and add –ide ...
... Ca = calcium; N = nitrogen Drop the end of the anion and add –ide ...
Chapter 12
... –Ex. C4H10 butane ; isobutane Alkyl Groups & IUPAC names Use the following rules to properly name hydrocarbon molecules: –1. Identify the longest, continuous chain of C atoms and name it. –2. # the C atoms in the chain from the end nearest an alkyl group. –3. # and name the attached alkyl group(s). ...
... –Ex. C4H10 butane ; isobutane Alkyl Groups & IUPAC names Use the following rules to properly name hydrocarbon molecules: –1. Identify the longest, continuous chain of C atoms and name it. –2. # the C atoms in the chain from the end nearest an alkyl group. –3. # and name the attached alkyl group(s). ...
Chemistry Exam Review
... 3. Particles are always in constant random motion. 4. The particles in a substance attract each other. 5. The particles of a substance move faster when ...
... 3. Particles are always in constant random motion. 4. The particles in a substance attract each other. 5. The particles of a substance move faster when ...
Balancing Chemical Equations
... compounds. Therefore you add another to the front of the compound. Since the compounds cannot be separated, you cannot just add a two in front of the oxygen atom only, it has to be in front of the whole compound. ...
... compounds. Therefore you add another to the front of the compound. Since the compounds cannot be separated, you cannot just add a two in front of the oxygen atom only, it has to be in front of the whole compound. ...
All chemical equations must be balanced, that is, they must have the
... There are times, however, that hydrocarbons do not combust completely; this is called incomplete combustion and the products are slightly different. You would always be told if the reaction was compete or incomplete, if nothing is said, assume it is complete combustion Incomplete combustion gives th ...
... There are times, however, that hydrocarbons do not combust completely; this is called incomplete combustion and the products are slightly different. You would always be told if the reaction was compete or incomplete, if nothing is said, assume it is complete combustion Incomplete combustion gives th ...
history of the atom ppt student copy
... 4. Atoms of different elements combined in whole-number ratios to form chemical compounds. 5. In chemical reactions, ____________________________________ ________________________________________________________ •Dalton’s theory helped explain the law of conservation of mass because it stated that at ...
... 4. Atoms of different elements combined in whole-number ratios to form chemical compounds. 5. In chemical reactions, ____________________________________ ________________________________________________________ •Dalton’s theory helped explain the law of conservation of mass because it stated that at ...
CHE 1401 - Fall 2013 - Chapter 7 Homework 7 (Chapter 7: Periodic
... 12) Alkali metals tend to be more reactive than alkaline earth metals because __________. A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more r ...
... 12) Alkali metals tend to be more reactive than alkaline earth metals because __________. A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more r ...
General Chemistry - Review for final exam: (Make sure you bring
... 34. What is the formula for the speed of light? 35. How is a photon created as an atom is heated? 36. According to Schrodinger, where are you most likely to find the electron in an s orbital? 37. Where are periods and groups on the periodic table? 38. Where are the following found on the periodic ta ...
... 34. What is the formula for the speed of light? 35. How is a photon created as an atom is heated? 36. According to Schrodinger, where are you most likely to find the electron in an s orbital? 37. Where are periods and groups on the periodic table? 38. Where are the following found on the periodic ta ...
Tutorial 1
... 1. Use the second member of each group from Group 1A to Group 7A to show that the number of valance electrons on an atom of the element is the same as its group number. 2. Use Lewis dot symbol to show the formation of aluminum oxide (Al2O3) 3. Explain what an ionic bond is? And name five metals and ...
... 1. Use the second member of each group from Group 1A to Group 7A to show that the number of valance electrons on an atom of the element is the same as its group number. 2. Use Lewis dot symbol to show the formation of aluminum oxide (Al2O3) 3. Explain what an ionic bond is? And name five metals and ...
Chapter 4: Chemical Reactions Elements can be characterized as
... Oxidation numbers (Table 4-10) are used to track electron transfer in oxidation-reduction (redox) reactions. Oxidation numbers are more important in binary ionic compounds than molecular compounds. Some abbreviated rules that apply are below (full list on page 138): 1. Oxidation number of free eleme ...
... Oxidation numbers (Table 4-10) are used to track electron transfer in oxidation-reduction (redox) reactions. Oxidation numbers are more important in binary ionic compounds than molecular compounds. Some abbreviated rules that apply are below (full list on page 138): 1. Oxidation number of free eleme ...
Family
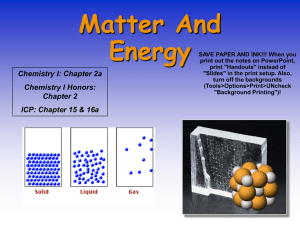
... In the solid phase, atoms cannot move past one another. Even in this state, atoms still have energy and move. Atoms in the solid phase vibrate. - As the temperature of a solid increases, the amount of vibration increases. The solid will keep its shape until the amount of energy added is greater than ...
... In the solid phase, atoms cannot move past one another. Even in this state, atoms still have energy and move. Atoms in the solid phase vibrate. - As the temperature of a solid increases, the amount of vibration increases. The solid will keep its shape until the amount of energy added is greater than ...
Chapter 1: Matter, Measurement and Problem Solving
... Free to move Compressible: forcing gas into a small container No fixed volume: volume of the container No fixed shape: take the shape of container Examples: Helium and Nitrogen ...
... Free to move Compressible: forcing gas into a small container No fixed volume: volume of the container No fixed shape: take the shape of container Examples: Helium and Nitrogen ...
Chapter 2 - Chemistry
... Postulates of Dalton s Atomic Theory 1.) All matter is composed of indivisible atoms. An atom is an extremely small particle of matter that retains its identity during chemical reactions. 2.) An element is a type of matter composed of only one kind of atom, each atom of a given kind having the same ...
... Postulates of Dalton s Atomic Theory 1.) All matter is composed of indivisible atoms. An atom is an extremely small particle of matter that retains its identity during chemical reactions. 2.) An element is a type of matter composed of only one kind of atom, each atom of a given kind having the same ...
Equation Intro Worksheet 1213
... c) potassium iodide + lead (II) nitrate → lead (II) iodide + potassium nitrate ...
... c) potassium iodide + lead (II) nitrate → lead (II) iodide + potassium nitrate ...
Chemical Equation
... • Usually the group number indicates the oxidation number of the elements in that group. • The cation simply has the same name as the element. • Transition Metals have multiple oxidation numbers. ...
... • Usually the group number indicates the oxidation number of the elements in that group. • The cation simply has the same name as the element. • Transition Metals have multiple oxidation numbers. ...
Chemistry PowerPoint
... What does the law of conservation of matter state? a. The total mass of the reactants is greater than the total mass of the products b. The total mass of the reactants is less than the total mass of the products c. The total mass of the reactants equals the total mass of the products d. Mass can be ...
... What does the law of conservation of matter state? a. The total mass of the reactants is greater than the total mass of the products b. The total mass of the reactants is less than the total mass of the products c. The total mass of the reactants equals the total mass of the products d. Mass can be ...
Mid-Term OR Study Guide
... polar bonds in all formulas, show where shared electrons come from with different symbols (x’s, open and solid dots, stars, different color dots, etc.), and put loops around shared electron pairs. (A) Is the bond type between a phosphorus atom and a fluorine atom ionic, polar covalent, or nonpolar c ...
... polar bonds in all formulas, show where shared electrons come from with different symbols (x’s, open and solid dots, stars, different color dots, etc.), and put loops around shared electron pairs. (A) Is the bond type between a phosphorus atom and a fluorine atom ionic, polar covalent, or nonpolar c ...
Chapter 3
... -3 Important Prop of Solutions 1.part not large enough to be seen 2.part. are evenly spread out(all parts of sol are ident) 3.solution doesn’t settle out of time ...
... -3 Important Prop of Solutions 1.part not large enough to be seen 2.part. are evenly spread out(all parts of sol are ident) 3.solution doesn’t settle out of time ...
CHEMISTRY
... ATOMS will switch partners when provoked. This is what chemical reactions are all about. Don’t mess with an atom’s nucleus. ...
... ATOMS will switch partners when provoked. This is what chemical reactions are all about. Don’t mess with an atom’s nucleus. ...
The Periodic Table - Harlan Independent Schools
... Transition elements have properties similar to one another and to other metals, but their properties do not fit in with those of any other family. Many transition metals combine chemically with oxygen to form compounds called oxides. ...
... Transition elements have properties similar to one another and to other metals, but their properties do not fit in with those of any other family. Many transition metals combine chemically with oxygen to form compounds called oxides. ...