2202 Chapter 1 - Eric G. Lambert School
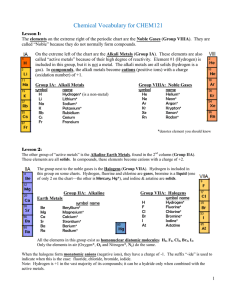
... - composed of a cation and an anion (usually metal and nonmetal) eg. NaCl Mg3N2 ...
... - composed of a cation and an anion (usually metal and nonmetal) eg. NaCl Mg3N2 ...
chapter2 2012 (no naming)
... Greeks: Empedocles and Democritus • Suggested the concept of atoms but were not taken seriously or credited with an atomic theory ...
... Greeks: Empedocles and Democritus • Suggested the concept of atoms but were not taken seriously or credited with an atomic theory ...
Matter and Atoms
... States of Matter – 3.3 Objectives •Describe the states of matter on Earth • Explain the reasons that matter exists in these ...
... States of Matter – 3.3 Objectives •Describe the states of matter on Earth • Explain the reasons that matter exists in these ...
File
... _____ 6. If two or more compounds are composed of the same two elements, then the ratio of the masses of the second element that is combined with a certain mass of the first element is always a ratio of small whole numbers. This statement is called the law of a. definite proportions. b. conservation ...
... _____ 6. If two or more compounds are composed of the same two elements, then the ratio of the masses of the second element that is combined with a certain mass of the first element is always a ratio of small whole numbers. This statement is called the law of a. definite proportions. b. conservation ...
Organic compounds are covalent compounds composed of carbon
... SPI 0807.9.4 – Differentiate between a mixture and a compound ...
... SPI 0807.9.4 – Differentiate between a mixture and a compound ...
The formula and name denote elements and relative composition in
... Empirical Formulas: chemical formula that indicates the relative proportions of the elements in a molecule rather than the actual number of atoms of the elements. It is a ratio. Ex: Determine the empirical formula for a compound containing 75% C and 25% H. 1. Assume 100g (make it easy for yourself) ...
... Empirical Formulas: chemical formula that indicates the relative proportions of the elements in a molecule rather than the actual number of atoms of the elements. It is a ratio. Ex: Determine the empirical formula for a compound containing 75% C and 25% H. 1. Assume 100g (make it easy for yourself) ...
Atoms, Molecules and Ions
... and closest to the bottom of a group on periodic table is placed first in formula − If more than one compound can be formed from the same elements, use prefixes to indicate number of each kind of atom − Last element name ends in -ide ...
... and closest to the bottom of a group on periodic table is placed first in formula − If more than one compound can be formed from the same elements, use prefixes to indicate number of each kind of atom − Last element name ends in -ide ...
Packet
... A. Iron (III) sulfate __________________________ B. Iron (II) sulfate __________________________ C. Oxygen gas _______________________ D. Nitric Acid ______________________________ ...
... A. Iron (III) sulfate __________________________ B. Iron (II) sulfate __________________________ C. Oxygen gas _______________________ D. Nitric Acid ______________________________ ...
PowerPoint Template
... matter usually occurs as mixtures, such as air, seawater, soil, and organisms A heterogeneous mixture has one or more visible boundaries between the components Example: rocks A homogeneous mixture has no visible boundaries because the components are mixed as individual atoms, ions, and ...
... matter usually occurs as mixtures, such as air, seawater, soil, and organisms A heterogeneous mixture has one or more visible boundaries between the components Example: rocks A homogeneous mixture has no visible boundaries because the components are mixed as individual atoms, ions, and ...
Atoms, Molecules and Ions
... must have two positive charges. Copper (II) nitrate. (b)The cation is K+ and the anion is PO4-3 (phosphate). Because potassium only forms one type of ion (K+), there is no need to use potassium (I) in the name. The compound is potassium phosphate. (c) The cation is NH4+1 (ammonium ion) and the anion ...
... must have two positive charges. Copper (II) nitrate. (b)The cation is K+ and the anion is PO4-3 (phosphate). Because potassium only forms one type of ion (K+), there is no need to use potassium (I) in the name. The compound is potassium phosphate. (c) The cation is NH4+1 (ammonium ion) and the anion ...
AtomMoleculeNaming_G1
... Metal Cations: Type II Type II: Metal ions that are other than Type I Common Examples: Fe2+/3+, Cu+/2+, Cr3+/6+, Mn2+/4+, Mn2+/4+, Pb2+/4+, Sn2+/4+, etc ) – have more than one possible charge – determine charge by charge on anion ...
... Metal Cations: Type II Type II: Metal ions that are other than Type I Common Examples: Fe2+/3+, Cu+/2+, Cr3+/6+, Mn2+/4+, Mn2+/4+, Pb2+/4+, Sn2+/4+, etc ) – have more than one possible charge – determine charge by charge on anion ...
Chemical Reactions
... • Synthesis – 2 substances (reactants) combine to form a new substance (product). – Substances are either atoms (elements) or compounds in this case. A + ...
... • Synthesis – 2 substances (reactants) combine to form a new substance (product). – Substances are either atoms (elements) or compounds in this case. A + ...
nature of Matter
... Nitrogen-14 (7 protons & 7 neutrons) An isotope is when atoms of the same element have a different number of NEUTRONS. Isotopes are identified by their mass number. Since they still have the same number of electrons, all isotopes of an element have the same chemical properties. A compound is a sub ...
... Nitrogen-14 (7 protons & 7 neutrons) An isotope is when atoms of the same element have a different number of NEUTRONS. Isotopes are identified by their mass number. Since they still have the same number of electrons, all isotopes of an element have the same chemical properties. A compound is a sub ...
The Periodic table and subatomic particles
... 3. Go through your grade 9 and 10 notes (should you still have them). 4. Topics to be covered include: Periodic table and its organization, subatomic particles, Bohr-Rutherford diagrams, ions, counting atoms, chemical reactions, names an formulas, balancing chemical equations, law of conservation of ...
... 3. Go through your grade 9 and 10 notes (should you still have them). 4. Topics to be covered include: Periodic table and its organization, subatomic particles, Bohr-Rutherford diagrams, ions, counting atoms, chemical reactions, names an formulas, balancing chemical equations, law of conservation of ...
JJ Thompson Webquest
... theory of chemistry was the Law of Multiple Proportions. He found that when carbon combined with oxygen to form a gas, there were two possible outcomes, depending on the conditions - and in one outcome each gram of carbon combined with precisely twice as much oxygen as in the other. He correctly int ...
... theory of chemistry was the Law of Multiple Proportions. He found that when carbon combined with oxygen to form a gas, there were two possible outcomes, depending on the conditions - and in one outcome each gram of carbon combined with precisely twice as much oxygen as in the other. He correctly int ...
Atomic Structure (history of atom)
... ATOMS of any one ELEMENT are different from those of any other element Atoms of different elements can physically mix together or chemically combine to form ...
... ATOMS of any one ELEMENT are different from those of any other element Atoms of different elements can physically mix together or chemically combine to form ...
CHEM 1305 - HCC Learning Web
... B) Ternary ionic C) Binary molecular D) Binary acid -------12. What is the term for a single atom bearing a positive or a negative charge as the result of gaining or losing valence electrons? A) Anion B) Cation C) Monoatomic ion D) Polyatomic ion -------13. The compound Na2SO4 is classified as which ...
... B) Ternary ionic C) Binary molecular D) Binary acid -------12. What is the term for a single atom bearing a positive or a negative charge as the result of gaining or losing valence electrons? A) Anion B) Cation C) Monoatomic ion D) Polyatomic ion -------13. The compound Na2SO4 is classified as which ...
Terminology 1
... Matter that has a definite or constant composition and distinct properties Matter that has a particular set of characteristics differs from the characteristics of another kind of matter ...
... Matter that has a definite or constant composition and distinct properties Matter that has a particular set of characteristics differs from the characteristics of another kind of matter ...
chapter 6 sec 2 resonance structure
... H is 2.1 and O is 3.5. 3.5 – 2.1 = 1.4 so the bond between H and O is a polar covalent bond. By definition a neutral group of atoms held together by covalent bonds is a molecule. So, the H2O particle is a molecule H2O is a molecule which makes H2O a molecular compound and a molecular formula. But H2 ...
... H is 2.1 and O is 3.5. 3.5 – 2.1 = 1.4 so the bond between H and O is a polar covalent bond. By definition a neutral group of atoms held together by covalent bonds is a molecule. So, the H2O particle is a molecule H2O is a molecule which makes H2O a molecular compound and a molecular formula. But H2 ...
Dalton Model Reading
... constituent elements, then the masses of the constituents will always have the same proportions, regardless of the quantity or source of the original substance. John Dalton studied and expanded upon this previous work and developed the law of multiple proportions: if two elements can together form m ...
... constituent elements, then the masses of the constituents will always have the same proportions, regardless of the quantity or source of the original substance. John Dalton studied and expanded upon this previous work and developed the law of multiple proportions: if two elements can together form m ...
Unit 1 - Measurement Atomic Theory
... (f) Formula units are structure where cation charges = anion charges (i) Anions and Cation take on noble gas configuration ...
... (f) Formula units are structure where cation charges = anion charges (i) Anions and Cation take on noble gas configuration ...
Chapters 6, 8
... When non-metals react among themselves, the resulting compound is a molecular compound. Each reacting atom delivers its valence electrons to a pool of electrons, and the resulting compound is made by sharing these electrons among all atoms in the compound. Each shared electron pair produces a covale ...
... When non-metals react among themselves, the resulting compound is a molecular compound. Each reacting atom delivers its valence electrons to a pool of electrons, and the resulting compound is made by sharing these electrons among all atoms in the compound. Each shared electron pair produces a covale ...