File
... Coefficients Interpretation of above example: 2 atoms of solid iron (metal) react with 3 molecules of chlorine gas to produce 2 formula units of solid iron (III) chloride. Coefficients can also be interpreted in a more useful way: MOLES! This is just as if we multiplied the whole equation by 6.02 x ...
... Coefficients Interpretation of above example: 2 atoms of solid iron (metal) react with 3 molecules of chlorine gas to produce 2 formula units of solid iron (III) chloride. Coefficients can also be interpreted in a more useful way: MOLES! This is just as if we multiplied the whole equation by 6.02 x ...
Investigating Chemistry - Chemistry at Winthrop University
... periodic table combine, we use a different system for naming these covalent compounds. • This results in discrete molecules with directional bonds. For example, H2O. • It can also result in an infinite network of covalently bonded atoms as in diamond, C, or sand, SiO2, which is not a discrete molecu ...
... periodic table combine, we use a different system for naming these covalent compounds. • This results in discrete molecules with directional bonds. For example, H2O. • It can also result in an infinite network of covalently bonded atoms as in diamond, C, or sand, SiO2, which is not a discrete molecu ...
periodic table - Mesa Community College
... to the second task: assembling these symbols and names and learning how to write formulas and name compounds. Compounds are defined as pure substances that can be broken down to simpler pure substances by a chemical reaction. The smallest particle of an element that can take part in a chemical react ...
... to the second task: assembling these symbols and names and learning how to write formulas and name compounds. Compounds are defined as pure substances that can be broken down to simpler pure substances by a chemical reaction. The smallest particle of an element that can take part in a chemical react ...
TEST REVIEW S Valence Electrons TEST REVIEW SHEET 2017
... NOTE: If an element has <4 valence electrons it will give them away during an ionic bond and become a positive ion. If >4, it will take them and become a negative ion For the most part…. metals will give away their valence electrons and nonmetals will take enough valence electrons to fill their oute ...
... NOTE: If an element has <4 valence electrons it will give them away during an ionic bond and become a positive ion. If >4, it will take them and become a negative ion For the most part…. metals will give away their valence electrons and nonmetals will take enough valence electrons to fill their oute ...
key - Greenslime.info
... Define “atomic mass” as used and listed on the periodic table. What does it tell about an element? The average number of protons plus neutrons in the nucleus of an atom. For any one element, the number of protons does not change, but the number of neutrons can change with different isotopes. ...
... Define “atomic mass” as used and listed on the periodic table. What does it tell about an element? The average number of protons plus neutrons in the nucleus of an atom. For any one element, the number of protons does not change, but the number of neutrons can change with different isotopes. ...
Chapters 1-4 Numbers and Measurements in Chemistry Units SI
... • Ions are formed when the number of protons and electrons in an atom are not equal. – Ions with more protons than electrons are called cations. • net positive charge ...
... • Ions are formed when the number of protons and electrons in an atom are not equal. – Ions with more protons than electrons are called cations. • net positive charge ...
Chapter 2
... The salt calcium fluoride is an ionic substance with formula CaF2, meaning that, while the atoms of Ca and F are in a 1-to-2 ratio, it does not exist as molecules of CaF2, but as a crystal lattice containing 1 Ca for every 2 F atoms. ...
... The salt calcium fluoride is an ionic substance with formula CaF2, meaning that, while the atoms of Ca and F are in a 1-to-2 ratio, it does not exist as molecules of CaF2, but as a crystal lattice containing 1 Ca for every 2 F atoms. ...
Solute
... Can be elements or compounds Element – substances made up of only one kind of atom Every element has a unique atomic number Atomic number – number of protons in the nucleus ...
... Can be elements or compounds Element – substances made up of only one kind of atom Every element has a unique atomic number Atomic number – number of protons in the nucleus ...
g - Porterville College Home
... c. Prefix exception: Mono is never used on the first word regardless if there is one atom of that element. (e.g. carbon monoxide not monocarbon monoxide) d. Prefix vowel omitted if similar vowel sounds run together by addition of the prefix. (e.g. carbon monoxide NOT carbon monooxide) ...
... c. Prefix exception: Mono is never used on the first word regardless if there is one atom of that element. (e.g. carbon monoxide not monocarbon monoxide) d. Prefix vowel omitted if similar vowel sounds run together by addition of the prefix. (e.g. carbon monoxide NOT carbon monooxide) ...
13.2 Chemical Formulas
... oxidation numbers are the key. An element’s oxidation number is the number of electrons it will gain or lose in a chemical reaction. We can use the periodic table to find the oxidation number for an element. When we add up the oxidation numbers of the elements in an ionic compound, the sum must be z ...
... oxidation numbers are the key. An element’s oxidation number is the number of electrons it will gain or lose in a chemical reaction. We can use the periodic table to find the oxidation number for an element. When we add up the oxidation numbers of the elements in an ionic compound, the sum must be z ...
Chemistry Test Study Guide
... classify as either metal, nonmetal, or metalloid: Ca, Cl, I, Ir, Si, and Ti. ...
... classify as either metal, nonmetal, or metalloid: Ca, Cl, I, Ir, Si, and Ti. ...
9. Balancing Equations
... How many sodiums on the left? 2; how many on the right/ 1; put a coefficient 2 in front of the one on the right. How many oxygens on the left/ on the right/, etc. Na2O + BaCl2 - 2NaCl + BaO ...
... How many sodiums on the left? 2; how many on the right/ 1; put a coefficient 2 in front of the one on the right. How many oxygens on the left/ on the right/, etc. Na2O + BaCl2 - 2NaCl + BaO ...
Slide 1 - Herricks
... 1. With the energy term within the equation 2. With the energy term outside the equation ...
... 1. With the energy term within the equation 2. With the energy term outside the equation ...
Chapter 2 power point
... 2) Oxoacid names are similar to those of the oxoanions, except for two suffix changes: -ate in the anion becomes –ic in the acid -ite in the anion becomes –ous in the acid The oxoanion prefixes hypo- and per- are retained. Thus, BrO4– is perbromate, and HBrO4 is perbromic acid; IO2– is iodite, and H ...
... 2) Oxoacid names are similar to those of the oxoanions, except for two suffix changes: -ate in the anion becomes –ic in the acid -ite in the anion becomes –ous in the acid The oxoanion prefixes hypo- and per- are retained. Thus, BrO4– is perbromate, and HBrO4 is perbromic acid; IO2– is iodite, and H ...
4.1 & 4.2 LDP and R.A.M
... -Now, Dalton used hydrogen first because it was the lightest element and gave it a mass of 1. -he compared all the other element to this value For ex. : when Dalton looked at water, he saw thta 1 g of hydrogen combined with 8 g oxygen -so he gave oxygen a mass of 8 -this was a mistake since 2 atoms ...
... -Now, Dalton used hydrogen first because it was the lightest element and gave it a mass of 1. -he compared all the other element to this value For ex. : when Dalton looked at water, he saw thta 1 g of hydrogen combined with 8 g oxygen -so he gave oxygen a mass of 8 -this was a mistake since 2 atoms ...
SLE133 – “Chemistry in Our World” Summary Notes Week 1
... 1. Frying an egg (bonds break and new bonds form, cannot change the fried egg into the original state) 2. Burning a candle (the wax burns into CO2, H2O, and some other compounds) More examples in ‘exam hints’ section. ...
... 1. Frying an egg (bonds break and new bonds form, cannot change the fried egg into the original state) 2. Burning a candle (the wax burns into CO2, H2O, and some other compounds) More examples in ‘exam hints’ section. ...
New substances are formed by chemical reactions. When elements
... by covalent bonds. These bonds form when atoms share pairs of electrons. Chemical formulas The chemical formula of a compound shows how many of each type of atom join together to make the units which make up the compound. For example, in iron sulfide every iron atom is joined to one sulfur atom, so ...
... by covalent bonds. These bonds form when atoms share pairs of electrons. Chemical formulas The chemical formula of a compound shows how many of each type of atom join together to make the units which make up the compound. For example, in iron sulfide every iron atom is joined to one sulfur atom, so ...
CHM121 Exam I Review
... 5. What is the weight of a 165 pound person in grams? (1 lb = 0.4536 kg) 6. A procedure calls for 6.5 g thionyl chloride (SOCl2). SOCl2 is a liquid so you want to measure it using a syringe. Its density is 1.631, according to an Aldrich catalog. How many mL are ...
... 5. What is the weight of a 165 pound person in grams? (1 lb = 0.4536 kg) 6. A procedure calls for 6.5 g thionyl chloride (SOCl2). SOCl2 is a liquid so you want to measure it using a syringe. Its density is 1.631, according to an Aldrich catalog. How many mL are ...
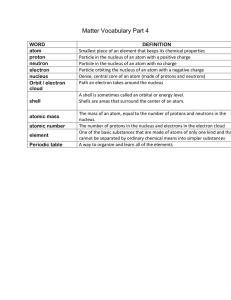
Matter Vocab Part 4
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
... Particle in the nucleus of an atom with a positive charge Particle in the nucleus of an atom with no charge Particle orbiting the nucleus of an atom with a negative charge Dense, central core of an atom (made of protons and neutrons) Path an electron takes around the nucleus A shell is sometimes cal ...
Review for Exam 1
... Determine how many of each ion type is needed for an overall charge of zero. When the cation and anion have different charges, use the ion charges to determine the number of ions of each needed. ...
... Determine how many of each ion type is needed for an overall charge of zero. When the cation and anion have different charges, use the ion charges to determine the number of ions of each needed. ...
1 - M*W
... a) Are the last column (to the right) on the periodic table b) Do not react with other elements c) Are the first column (on the left) on the periodic table d) B & C 34) Halogens, like fluorine, are very reactive because a) They want to gain an electron to complete their outer energy level b) They wa ...
... a) Are the last column (to the right) on the periodic table b) Do not react with other elements c) Are the first column (on the left) on the periodic table d) B & C 34) Halogens, like fluorine, are very reactive because a) They want to gain an electron to complete their outer energy level b) They wa ...