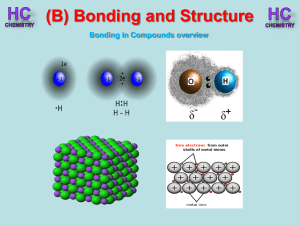
Lesson 1 - Bonding in compounds overview
... Silicon, like carbon, can form giant covalent networks. Silicon carbide exist in a similar structure to diamond. Tetrahedral shape ...
... Silicon, like carbon, can form giant covalent networks. Silicon carbide exist in a similar structure to diamond. Tetrahedral shape ...
Chapter 3 - pennsaukenchemistry2
... element present, if necessary. 2. Calculate the number of _________ of each element. ...
... element present, if necessary. 2. Calculate the number of _________ of each element. ...
Objective 3 Stations Student Sheet
... 1. How is the periodic table organized? 2. What family of elements has valence electrons at two energy levels? 3. What are the elements called that are between metals and nonmetals? 4. Which family of nonmetals has seven valence electrons? 5. What are some properties of noble gases? 6. What is anoth ...
... 1. How is the periodic table organized? 2. What family of elements has valence electrons at two energy levels? 3. What are the elements called that are between metals and nonmetals? 4. Which family of nonmetals has seven valence electrons? 5. What are some properties of noble gases? 6. What is anoth ...
Chemistry
... The atomic mass listed on the periodic table is an average mass for all the different isotopes that exist, taking into account the percent and mass of each different isotope. C4.10e C4.10c Calculate the average atomic mass of an element given the percent abundance and mass of the individual isotopes ...
... The atomic mass listed on the periodic table is an average mass for all the different isotopes that exist, taking into account the percent and mass of each different isotope. C4.10e C4.10c Calculate the average atomic mass of an element given the percent abundance and mass of the individual isotopes ...
Page 1 of 4 FOSS California Mixtures and Solutions
... Bends: A condition that causes pain in deep-sea divers’ arms and legs after returning to the surface. Caisson: A large box with no bottom. These boxes were used to provide environments for workers under water. Carbohydrate: A group of carbon-based nutrients, such as sugars and starches. Carbon-14 da ...
... Bends: A condition that causes pain in deep-sea divers’ arms and legs after returning to the surface. Caisson: A large box with no bottom. These boxes were used to provide environments for workers under water. Carbohydrate: A group of carbon-based nutrients, such as sugars and starches. Carbon-14 da ...
matter
... • A substance made of two or more elements • When they combine, they form compounds with properties different from the elements. • Example: C12H22011=table sugar It has 12 atoms of carbon, 22 atoms of hydrogen, and 11 atoms of oxygen Carbon is black but sugar is not Oxygen is a gas but sugar is not ...
... • A substance made of two or more elements • When they combine, they form compounds with properties different from the elements. • Example: C12H22011=table sugar It has 12 atoms of carbon, 22 atoms of hydrogen, and 11 atoms of oxygen Carbon is black but sugar is not Oxygen is a gas but sugar is not ...
Power point types of chemical rxn
... 1. Sulfuric acid is used to neutralize calcium hydroxide: • H2SO4 + Ca(OH) 2 CaSO4 + 2H2O 2. Phosphoric acid helps to neutralize the compounds that cause rust, such as iron(II) hydroxide. • H3PO4 + 3Fe(OH)2 Fe3(PO4)2 + 6H2O NEUTRALIZATION: http://www.youtube.com/watch?v=_P5hGzA6Vb0 See page 263 ...
... 1. Sulfuric acid is used to neutralize calcium hydroxide: • H2SO4 + Ca(OH) 2 CaSO4 + 2H2O 2. Phosphoric acid helps to neutralize the compounds that cause rust, such as iron(II) hydroxide. • H3PO4 + 3Fe(OH)2 Fe3(PO4)2 + 6H2O NEUTRALIZATION: http://www.youtube.com/watch?v=_P5hGzA6Vb0 See page 263 ...
math_in_chemistry
... 3) You find 7.36 g of a compound has decomposed to give 6.93 g of oxygen. The rest is hydrogen. If the molecular mass is 34.0 g/mol, what is the molecular formula? ...
... 3) You find 7.36 g of a compound has decomposed to give 6.93 g of oxygen. The rest is hydrogen. If the molecular mass is 34.0 g/mol, what is the molecular formula? ...
Semester 2 Review WS
... d. What is represented in letter C? _________________________________________________ e. How many total ATOMS are there in letter D? _____________________________________ f. How many total MOLECULES are there in letter D? ________________________________ 2. Distinguish between the structure of molec ...
... d. What is represented in letter C? _________________________________________________ e. How many total ATOMS are there in letter D? _____________________________________ f. How many total MOLECULES are there in letter D? ________________________________ 2. Distinguish between the structure of molec ...
File
... 2.7 Physical properties and Intermolecular Interactions • Alkanes are insoluble in water. That is because water molecules are polar, whereas alkanes are nopolar. (all C-C and C-H bonds are nearly purely covalent.) • Alkanes have lower boiling points for a given molecular weight than most other orga ...
... 2.7 Physical properties and Intermolecular Interactions • Alkanes are insoluble in water. That is because water molecules are polar, whereas alkanes are nopolar. (all C-C and C-H bonds are nearly purely covalent.) • Alkanes have lower boiling points for a given molecular weight than most other orga ...
biology biology - Napa Valley College
... Runoff from agricultural land may have high levels of some elements such as selenium. Plants can take up selenium and convert it to gas, removing it from the water. We can use these plants to remove selenium = ...
... Runoff from agricultural land may have high levels of some elements such as selenium. Plants can take up selenium and convert it to gas, removing it from the water. We can use these plants to remove selenium = ...
2.1 Atoms and Bonds
... ◦ The properties of a compound are different than the properties of the elements in the compound ...
... ◦ The properties of a compound are different than the properties of the elements in the compound ...
Year 10 Chemistry Exam June 2011 Multiple Choice Section A
... A different atoms of the same element with a different number of protons B different atoms of the same element with a different number of electrons C different atoms of the same element with different mass numbers. D different atoms of the same element with different atomic numbers. 7 The maximum nu ...
... A different atoms of the same element with a different number of protons B different atoms of the same element with a different number of electrons C different atoms of the same element with different mass numbers. D different atoms of the same element with different atomic numbers. 7 The maximum nu ...
Fall Exam 1
... 13. This summer, a new mode of public transportation called the Hyperloop was proposed. The inventor claims the Hyperloop could reach average speeds of 598 miles per hour. Assuming that you could travel at this speed on the Hyperloop, how long would it take to go from Lexington to Chicago, IL (358.8 ...
... 13. This summer, a new mode of public transportation called the Hyperloop was proposed. The inventor claims the Hyperloop could reach average speeds of 598 miles per hour. Assuming that you could travel at this speed on the Hyperloop, how long would it take to go from Lexington to Chicago, IL (358.8 ...
Ch. 2 Chemistry
... • Are those in the outermost, or valence shell • Determines the chemical behavior of an atom The valence number of an atom is the number of unpaired electrons in its valence shell This valence number determines the number of bonds the atom can form. ...
... • Are those in the outermost, or valence shell • Determines the chemical behavior of an atom The valence number of an atom is the number of unpaired electrons in its valence shell This valence number determines the number of bonds the atom can form. ...
Topic 2 Microscopic World I
... 77 a) Use an electron diagram to show the electron transfer when each of the following pairs of elements combine, showing electrons in the outermost shells only. (3 marks) i) Calcium and chlorine ...
... 77 a) Use an electron diagram to show the electron transfer when each of the following pairs of elements combine, showing electrons in the outermost shells only. (3 marks) i) Calcium and chlorine ...
Chemistry Notes
... More Terms (You know you love ‘em.) WEAK ACID: An acid that only partially ionizes in an aqueous solution. That means not every molecule breaks apart. They usually have a pH close to 7 (3-6). WEAK BASE: A base that only partially ionizes in an aqueous solution. That means not every molecule breaks a ...
... More Terms (You know you love ‘em.) WEAK ACID: An acid that only partially ionizes in an aqueous solution. That means not every molecule breaks apart. They usually have a pH close to 7 (3-6). WEAK BASE: A base that only partially ionizes in an aqueous solution. That means not every molecule breaks a ...
Part of a Molecular Compound
... • On many of them you can round to the nearest gram, except for elements that atomic masses end in 0.5 • Use as many significant figures needed to maintain the number of significant figures given in the problem. ...
... • On many of them you can round to the nearest gram, except for elements that atomic masses end in 0.5 • Use as many significant figures needed to maintain the number of significant figures given in the problem. ...
9.1-10.5 Organic Chemistry
... branch of the same type, a locating number is given to each branch and a prefix indicating the number of that type of branch is attached to the name. This numbering prefix does not affect the alphabetical order of the branches Draw the structural formula for 3,4-dimethylhexane ...
... branch of the same type, a locating number is given to each branch and a prefix indicating the number of that type of branch is attached to the name. This numbering prefix does not affect the alphabetical order of the branches Draw the structural formula for 3,4-dimethylhexane ...
Pretest 4.3 2008
... How much power is lost if a high tension wire uses 50 000 V to transmit 100 000 W of power? (R for the high tension wire = 1000 Ω) ...
... How much power is lost if a high tension wire uses 50 000 V to transmit 100 000 W of power? (R for the high tension wire = 1000 Ω) ...
Remember Question words
... Law of Conservation of Mass = no detectable gain or loss in mass occurs in chemical reactions. However, the state of a substance may change in a chemical reaction. For example, substances in a chemical reaction can change from solid states to gaseous states but the total mass will not change. Or mor ...
... Law of Conservation of Mass = no detectable gain or loss in mass occurs in chemical reactions. However, the state of a substance may change in a chemical reaction. For example, substances in a chemical reaction can change from solid states to gaseous states but the total mass will not change. Or mor ...
Chapter 3 Reading Questions
... d. the number of electrons in a molecule 16. Which of the following statements concerning empirical formulas is TRUE? a. Empirical formulas tell the relative number of each type of atom b. Empirical formulas tells nothing about molecular structure c. Different compounds can have the same empirical f ...
... d. the number of electrons in a molecule 16. Which of the following statements concerning empirical formulas is TRUE? a. Empirical formulas tell the relative number of each type of atom b. Empirical formulas tells nothing about molecular structure c. Different compounds can have the same empirical f ...
Honors Chemistry Semester 1 Exam Review
... 2. For the following ionic names, write the missing chemical formula or chemical name. a. nickel (II) oxide _____________ b. calcium carbonate _____________ c. potassium nitrate ...
... 2. For the following ionic names, write the missing chemical formula or chemical name. a. nickel (II) oxide _____________ b. calcium carbonate _____________ c. potassium nitrate ...
File
... 2. The first element in the formula is named first, using the full element name. 3. The second element is named using the suffix ‘-ide.’ 4. Prefixes are used to denote the numbers of atoms present. See table below for prefixes. 5. The prefix ‘mono-’ is only used to name the second element. example: ...
... 2. The first element in the formula is named first, using the full element name. 3. The second element is named using the suffix ‘-ide.’ 4. Prefixes are used to denote the numbers of atoms present. See table below for prefixes. 5. The prefix ‘mono-’ is only used to name the second element. example: ...























