A Student want to prepare 250mL of .10 M NaCl solution
... The Number of atoms in 9.0g of Aluminum is equal to the Number of atoms in A) 8.1g Mg B) 12.1g Mg The Long way: C) 9.0g Mg Calculate # of atoms in 9.0 g Al D) 18.0g Mg Then calculate mass of that # of Mg atoms Look for ratios: Al has a molar mass of 27 This is 1/3 of a mole Al Which one is 1/3 mole ...
... The Number of atoms in 9.0g of Aluminum is equal to the Number of atoms in A) 8.1g Mg B) 12.1g Mg The Long way: C) 9.0g Mg Calculate # of atoms in 9.0 g Al D) 18.0g Mg Then calculate mass of that # of Mg atoms Look for ratios: Al has a molar mass of 27 This is 1/3 of a mole Al Which one is 1/3 mole ...
CHEMISTRY FINAL EXAM REVIEW SHEET
... 11. How many electrons are in the valence shells of these atoms and ions? Cl ...
... 11. How many electrons are in the valence shells of these atoms and ions? Cl ...
What You Need to Know to Pass the Chemistry
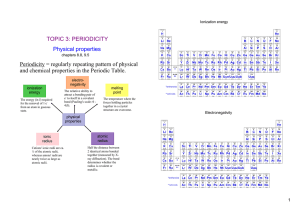
... 12. The succession of elements within a group demonstrates characteristic trends in properties. As you progress down a group: atomic radius increases. electronegativity decreases. first ionization energy decreases. metallic character increases. 13. The succession of elements within a period ...
... 12. The succession of elements within a group demonstrates characteristic trends in properties. As you progress down a group: atomic radius increases. electronegativity decreases. first ionization energy decreases. metallic character increases. 13. The succession of elements within a period ...
Exam 3 Review - Iowa State University
... a. What are the formal charges of Cl, N, and O in each structure? b. Are any of the oxygen atoms equivalent? c. What nitrogen- and oxygen-containing ion is isoelectronic with ClNO2? 22. Which contains a multiple bond in its Lewis structure? a. ICl b. SO2 c. Cl2 d. NH4+ e. CaI2 23. Find the enthalpy ...
... a. What are the formal charges of Cl, N, and O in each structure? b. Are any of the oxygen atoms equivalent? c. What nitrogen- and oxygen-containing ion is isoelectronic with ClNO2? 22. Which contains a multiple bond in its Lewis structure? a. ICl b. SO2 c. Cl2 d. NH4+ e. CaI2 23. Find the enthalpy ...
Class Notes
... What is an Element? Element: A substance that can not be changed into a simpler substance(s) under normal laboratory conditions A substance made up of only one type of atom is an elemental substance Material engineers are working on the process of creating man-made diamonds to be used as supe ...
... What is an Element? Element: A substance that can not be changed into a simpler substance(s) under normal laboratory conditions A substance made up of only one type of atom is an elemental substance Material engineers are working on the process of creating man-made diamonds to be used as supe ...
In 1869, Russia`s Dmitri Mendeleev and Germany`s Lothar Meyer
... are needed t o s ee thi s pi c ture. ...
... are needed t o s ee thi s pi c ture. ...
HOMEWORK 6-1 - losbanosusd.k12.ca.us
... 1. A polyatomic ion is a group of covalently bonded atoms. ____________ 2. NaCl is a molecular compound. _____________ 3. Lattice energy is released when the ions in a crystal lattice are separated from each other. ____________ 4. Ionic compounds are electrical conductors in their molten states. ___ ...
... 1. A polyatomic ion is a group of covalently bonded atoms. ____________ 2. NaCl is a molecular compound. _____________ 3. Lattice energy is released when the ions in a crystal lattice are separated from each other. ____________ 4. Ionic compounds are electrical conductors in their molten states. ___ ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... Solid lithium hydroxide is used in space vehicles to remove the carbon dioxide exhaled by astronauts. The lithium hydroxide reacts with gaseous carbon dioxide to form solid lithium carbonate and liquid water. How many grams of carbon dioxide can be absorbed by 1.00 g of lithium hydroxide? ...
... Solid lithium hydroxide is used in space vehicles to remove the carbon dioxide exhaled by astronauts. The lithium hydroxide reacts with gaseous carbon dioxide to form solid lithium carbonate and liquid water. How many grams of carbon dioxide can be absorbed by 1.00 g of lithium hydroxide? ...
Chemistry Midterm Review 2006
... 3. Differentiate characteristics of pure substances and mixtures. 4. Pure substances can be broken down into________________ and ___________________________. 5. Differentiate characteristics of an element and a compound. State examples. 6. Mixtures can be classified as _______________________ and __ ...
... 3. Differentiate characteristics of pure substances and mixtures. 4. Pure substances can be broken down into________________ and ___________________________. 5. Differentiate characteristics of an element and a compound. State examples. 6. Mixtures can be classified as _______________________ and __ ...
The mole
... the mass relationships of elements in compounds. 1. A mole (mol) is the amount of a substance that contains as many particles as there are atoms in exactly 12 g of carbon-12. The mole is the SI unit for amount of substance. The number of particles in a mole has been experimentally determined as 6.02 ...
... the mass relationships of elements in compounds. 1. A mole (mol) is the amount of a substance that contains as many particles as there are atoms in exactly 12 g of carbon-12. The mole is the SI unit for amount of substance. The number of particles in a mole has been experimentally determined as 6.02 ...
Percentage Composition
... determination. – Converting a massed sample of one element to a compound (to find the mass of the second that combined with the first). • Ex. 2Mg + O2 2 MgO ...
... determination. – Converting a massed sample of one element to a compound (to find the mass of the second that combined with the first). • Ex. 2Mg + O2 2 MgO ...
Itty-Bitty Atoms
... rows and columns. All of the elements in the ---,--vertical columns (up and H down) are alike in some ways. ...
... rows and columns. All of the elements in the ---,--vertical columns (up and H down) are alike in some ways. ...
Print this article - Bangladesh Journals Online
... hexane/CH2Cl2 (2:3, v/v) gave [Mn2(µ-η2-C7H5N2S)2(CO)6] (3) as reddish crystals (58 mg, 29%) from CH2Cl2 and hexane mixture at -200C. IR(νCO,CH2Cl2): 2036 vs, 2017 vs , 1922 br cm-1; 1H NMR (CD2 Cl2): δ 7.52 (d, H, J = 8.0 Hz), 7.25 (d, H, J=8.0 Hz), 6.9 (dd, H, J = 8.0 Hz), 6.50 (dd, H, J = 8.0 Hz ...
... hexane/CH2Cl2 (2:3, v/v) gave [Mn2(µ-η2-C7H5N2S)2(CO)6] (3) as reddish crystals (58 mg, 29%) from CH2Cl2 and hexane mixture at -200C. IR(νCO,CH2Cl2): 2036 vs, 2017 vs , 1922 br cm-1; 1H NMR (CD2 Cl2): δ 7.52 (d, H, J = 8.0 Hz), 7.25 (d, H, J=8.0 Hz), 6.9 (dd, H, J = 8.0 Hz), 6.50 (dd, H, J = 8.0 Hz ...
Calculating the Empirical Formula from Mass % Elemental
... BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
... BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
Review Station Ideas
... 3 Stoichiometry Station 10 – HYDRATES A student is assigned the task of determining the number of moles of water in one mole of MgCl2 · n H2O. The student collects the data shown in the following table. Mass of empty container Initial mass of sample and container Mass of sample and container after ...
... 3 Stoichiometry Station 10 – HYDRATES A student is assigned the task of determining the number of moles of water in one mole of MgCl2 · n H2O. The student collects the data shown in the following table. Mass of empty container Initial mass of sample and container Mass of sample and container after ...
c2 atomic structure f pmh
... A lithium atom can lose one electron to form a lithium ion which can be written (2)+ A fluorine atom can gain one electron to form a fluoride ion. Choose from the list the correct way to write the fluoride ion. ...
... A lithium atom can lose one electron to form a lithium ion which can be written (2)+ A fluorine atom can gain one electron to form a fluoride ion. Choose from the list the correct way to write the fluoride ion. ...
Chapter 2_Application Problems
... have one oxygen atom and two hydrogen atoms – correct; according to Dalton, atoms combine together in compounds in small whole-number ratios, so that you could describe a compound by describing the number of atoms of each element in a molecule. He used this idea to explain why compounds obey the Law ...
... have one oxygen atom and two hydrogen atoms – correct; according to Dalton, atoms combine together in compounds in small whole-number ratios, so that you could describe a compound by describing the number of atoms of each element in a molecule. He used this idea to explain why compounds obey the Law ...
Honors Midterm Review – 2015-16
... a) What is the greatest mass of ammonia, NH3 (g), that will be formed when 12.80 grams of H2(g) reacts with 55.25 grams of N2(g)? (Hint: this is a limiting reactant problem) ...
... a) What is the greatest mass of ammonia, NH3 (g), that will be formed when 12.80 grams of H2(g) reacts with 55.25 grams of N2(g)? (Hint: this is a limiting reactant problem) ...
Trends in the periodic table - Brigham Young University
... to remove one electron from an atom • what do you think will happen with the ionization energy, based on what you have learned about atomic radius? ...
... to remove one electron from an atom • what do you think will happen with the ionization energy, based on what you have learned about atomic radius? ...
File
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
Chapter 2
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
chemistry-2nd-edition-julia-burdge-solution
... numerically equal to the charge on the cation. The correct formula is Mg3(PO4)2. Note that for its subscript to be changed, a polyatomic ion must be enclosed in parentheses. Calcium is an alkaline earth metal. It only forms a 2 cation. The polyatomic ion hydrogen phosphate, HPO 24 , has a 2 charg ...
... numerically equal to the charge on the cation. The correct formula is Mg3(PO4)2. Note that for its subscript to be changed, a polyatomic ion must be enclosed in parentheses. Calcium is an alkaline earth metal. It only forms a 2 cation. The polyatomic ion hydrogen phosphate, HPO 24 , has a 2 charg ...