Microsoft Word
... Redox reactions are characterized by a transfer of electrons. An atom is oxidized (loses electrons) if its oxidation number increases (becomes more positive) in a chemical reaction; an atom is reduced (gains electrons) if its oxidation number decreases. ...
... Redox reactions are characterized by a transfer of electrons. An atom is oxidized (loses electrons) if its oxidation number increases (becomes more positive) in a chemical reaction; an atom is reduced (gains electrons) if its oxidation number decreases. ...
Chemical Reactions Chapter 11
... • Predicting the products of a chemical reaction involve 1st determining the type of reaction that is occurring. – Combination: starts with 2 elements – Decomposition: starts with 1 compound – Single Replacement: Starts with 1 element & 1 compound – Double Replacement: starts with 2 compounds – Neut ...
... • Predicting the products of a chemical reaction involve 1st determining the type of reaction that is occurring. – Combination: starts with 2 elements – Decomposition: starts with 1 compound – Single Replacement: Starts with 1 element & 1 compound – Double Replacement: starts with 2 compounds – Neut ...
Unit 3: Bonding and Nomenclature Content Outline: Calculating
... a. You have 8.5 moles of Fluorine (F) gas. How grams of Fluorine do you have? 8.5 moles x 19.00 grams = 160.0 grams or 1.6 x102 grams 1 mole V. Calculating Percent Composition from Molar Mass A. This calculation allows for us to find the percentage (out of 100%) of one element from the total molecul ...
... a. You have 8.5 moles of Fluorine (F) gas. How grams of Fluorine do you have? 8.5 moles x 19.00 grams = 160.0 grams or 1.6 x102 grams 1 mole V. Calculating Percent Composition from Molar Mass A. This calculation allows for us to find the percentage (out of 100%) of one element from the total molecul ...
Introductory Review
... For ionic compounds, e.g. sodium chloride, the formula shows the ratio of elements that form the compound. Solid sodium chloride consists of a collection of positively charged sodium ions and negatively charged chloride ions in a three-dimensional structure. You cannot say which sodium ion is assoc ...
... For ionic compounds, e.g. sodium chloride, the formula shows the ratio of elements that form the compound. Solid sodium chloride consists of a collection of positively charged sodium ions and negatively charged chloride ions in a three-dimensional structure. You cannot say which sodium ion is assoc ...
Redox
... This method is typically used for organic compounds, which contain many carbon, hydrogen, and oxygen atoms The advantage of the effective charge method is that you can determine which atom has been oxidized or reduced To determine effective charges, we will need to use some more advanced topics, suc ...
... This method is typically used for organic compounds, which contain many carbon, hydrogen, and oxygen atoms The advantage of the effective charge method is that you can determine which atom has been oxidized or reduced To determine effective charges, we will need to use some more advanced topics, suc ...
Nature of Molecules and Water
... – H2O, CO2, C6H12O2, NH3, O2, N2, H2… • Compounds are molecules containing more than one type of element – H2O, CO2, C6H12O2 ...
... – H2O, CO2, C6H12O2, NH3, O2, N2, H2… • Compounds are molecules containing more than one type of element – H2O, CO2, C6H12O2 ...
Types of Chemical Reactions
... general form of: A + BC → AC + B 5) Double Replacement: This is when the metals and nonmetals of two different molecules switch places, forming two entirely different compounds. These reactions are in the general form: ...
... general form of: A + BC → AC + B 5) Double Replacement: This is when the metals and nonmetals of two different molecules switch places, forming two entirely different compounds. These reactions are in the general form: ...
Midterm Review - Closter Public Schools
... liquids, they _____________________________. In gases they ________________________. Matter is said to be ______________ when it is has only one type of particle. Matter is said to be ______________when it has more than one type of particle. A ______________ is a pure substance that contains only a ...
... liquids, they _____________________________. In gases they ________________________. Matter is said to be ______________ when it is has only one type of particle. Matter is said to be ______________when it has more than one type of particle. A ______________ is a pure substance that contains only a ...
Ionic Bonding - Effingham County Schools
... Example: A sodium ion, Na+, has a charge of 1+. A chloride ion, Cl-, has a charge of 1-. There is an electrical force of attraction between oppositely charged ions. In sodium chloride, these ions combine in a one – to – one ratio so that each positive charge is balanced by a negative charge. attract ...
... Example: A sodium ion, Na+, has a charge of 1+. A chloride ion, Cl-, has a charge of 1-. There is an electrical force of attraction between oppositely charged ions. In sodium chloride, these ions combine in a one – to – one ratio so that each positive charge is balanced by a negative charge. attract ...
Chemistry - Beachwood City Schools
... 1. What is a chemical bond? Why do atoms form chemical bonds? How are covalent bonds and ionic bonds different? How are they the same? 2. How is the valence of an atom related to the number of bonds it usually forms? 3. What types of substances contain covalent bonds? 4. List the atoms in each of th ...
... 1. What is a chemical bond? Why do atoms form chemical bonds? How are covalent bonds and ionic bonds different? How are they the same? 2. How is the valence of an atom related to the number of bonds it usually forms? 3. What types of substances contain covalent bonds? 4. List the atoms in each of th ...
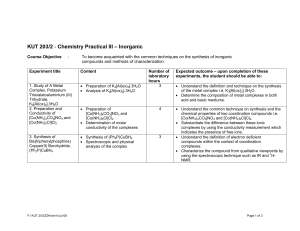
KUT 203/2 - Chemistry Practical III (Inorganic Chemistry)
... • Understand the correlation between the colors of transition metal or metal ion with its oxidation state i.e. VO2+ (yellow), VO2+ (blue) etc. • Determine the composition of a metal complex of which the metal exists in various oxidation states by using the titration technique. • Synthesize several c ...
... • Understand the correlation between the colors of transition metal or metal ion with its oxidation state i.e. VO2+ (yellow), VO2+ (blue) etc. • Determine the composition of a metal complex of which the metal exists in various oxidation states by using the titration technique. • Synthesize several c ...
Calculations with Chemical Formulas and Equations
... • So, the formula weight of calcium chloride, CaCl2, would be Ca: 1(40.1 amu) + Cl: 2(35.5 amu) 111.1 amu ...
... • So, the formula weight of calcium chloride, CaCl2, would be Ca: 1(40.1 amu) + Cl: 2(35.5 amu) 111.1 amu ...
2015 AP Chemistry Summer Assignment
... hydrogen, and 14.14% oxygen. Calculate the empirical formula for nylon-6. 36. Maleic acid is a compound composed of 41.39% C, 3.47% H, and the rest oxygen. If 0.129 mole of maleic acid has a mass of 15.0 g, what are the empirical and molecular formulas? 37. Many homes in rural America are heated by ...
... hydrogen, and 14.14% oxygen. Calculate the empirical formula for nylon-6. 36. Maleic acid is a compound composed of 41.39% C, 3.47% H, and the rest oxygen. If 0.129 mole of maleic acid has a mass of 15.0 g, what are the empirical and molecular formulas? 37. Many homes in rural America are heated by ...
I. Why Atoms Combine - Manchester High School
... • If charges cancel, just write the symbols. • If not, crisscross the charges to find subscripts. Use parentheses when more than one ...
... • If charges cancel, just write the symbols. • If not, crisscross the charges to find subscripts. Use parentheses when more than one ...
word-doc Practice for the final exam!
... 6. What is the volume of a 12.2 g piece of metal with a density of 9.43 g/cm3? a. 12.2 cm3 b. 1.29 cm3 c. 0.773 cm3 d. 115 cm3 e. none of these 7. "Absolute zero" refers to _______. a. 0 Kelvin b. 0o Fahrenheit c. 0o Celsius d. oC + 9/5(oF - 32) e. 273.15 oC 8. 1.035 x 10-4L = _____ mL a. 1.035 x 1 ...
... 6. What is the volume of a 12.2 g piece of metal with a density of 9.43 g/cm3? a. 12.2 cm3 b. 1.29 cm3 c. 0.773 cm3 d. 115 cm3 e. none of these 7. "Absolute zero" refers to _______. a. 0 Kelvin b. 0o Fahrenheit c. 0o Celsius d. oC + 9/5(oF - 32) e. 273.15 oC 8. 1.035 x 10-4L = _____ mL a. 1.035 x 1 ...
Unit B: Matter and Chemical Change
... Read and interpret chemical formulas for compounds of two elements, and give the IUPAC (international Union of Pure and Applied Chemistry) name and common name of these compounds (e.g., give, verbally and in writing, the name for NaCl(s) (sodium chloride), CO2(g) (carbon dioxide), MgO(s) (magnesium ...
... Read and interpret chemical formulas for compounds of two elements, and give the IUPAC (international Union of Pure and Applied Chemistry) name and common name of these compounds (e.g., give, verbally and in writing, the name for NaCl(s) (sodium chloride), CO2(g) (carbon dioxide), MgO(s) (magnesium ...
F. The Quantum Atom Theory - River Dell Regional School District
... 1. Develops Law of Definite Composition- all samples of a specific substance contain the same mass ratio of the same elements a. ex: all samples of CO2 contains 27.3% carbon and 72.7% oxygen b. therefore ‘elements’ are combining in a whole number ratio – WHY???? ...
... 1. Develops Law of Definite Composition- all samples of a specific substance contain the same mass ratio of the same elements a. ex: all samples of CO2 contains 27.3% carbon and 72.7% oxygen b. therefore ‘elements’ are combining in a whole number ratio – WHY???? ...
chemistry form iii - Covington Latin School
... Prerequisites: Algebra I and Geometry Course Description: Students enrolled in this course will receive an introduction to the following topics: measurements in chemistry; elements and compounds; matter and energy; periodic table and chemical nomenclature; chemical reactions; quantities in chemistry ...
... Prerequisites: Algebra I and Geometry Course Description: Students enrolled in this course will receive an introduction to the following topics: measurements in chemistry; elements and compounds; matter and energy; periodic table and chemical nomenclature; chemical reactions; quantities in chemistry ...
Document
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
Bacteria and Virus Research Jigsaw
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
Covalent Bonding - Effingham County Schools
... •These representations are all Lewis Structures, formulas in which atomic symbols represent nuclei and inner-shell electrons, dot-pairs or dashes between two atomic symbols represent electron pairs in covalent bonds, and dots adjacent to only one atomic symbol represent unshared electrons. Unshared ...
... •These representations are all Lewis Structures, formulas in which atomic symbols represent nuclei and inner-shell electrons, dot-pairs or dashes between two atomic symbols represent electron pairs in covalent bonds, and dots adjacent to only one atomic symbol represent unshared electrons. Unshared ...