2.5 THE NAMES AND FORMULAS OF COMPOUNDS
... compounds are weaker in comparison — adding a relatively small amount of heat will cause a solid molecular compound to change state from a solid to a liquid, and then to a gas. (b) Ionic compounds (many of which dissolve readily in water) form solutions that conduct electricity. Because the ionic bo ...
... compounds are weaker in comparison — adding a relatively small amount of heat will cause a solid molecular compound to change state from a solid to a liquid, and then to a gas. (b) Ionic compounds (many of which dissolve readily in water) form solutions that conduct electricity. Because the ionic bo ...
Day 72 TYPES OF CHEMICAL REACTIONS
... 1. Sulfuric acid is used to neutralize calcium hydroxide: • H2SO4 + Ca(OH) 2 CaSO4 + 2H2O 2. Phosphoric acid helps to neutralize the compounds that cause rust, such as iron(II) hydroxide. • H3PO4 + 3Fe(OH)2 Fe3(PO4)2 + 6H2O NEUTRALIZATION: http://www.youtube.com/watch?v=_P5hGzA6Vb0 See page 263 ...
... 1. Sulfuric acid is used to neutralize calcium hydroxide: • H2SO4 + Ca(OH) 2 CaSO4 + 2H2O 2. Phosphoric acid helps to neutralize the compounds that cause rust, such as iron(II) hydroxide. • H3PO4 + 3Fe(OH)2 Fe3(PO4)2 + 6H2O NEUTRALIZATION: http://www.youtube.com/watch?v=_P5hGzA6Vb0 See page 263 ...
Chapter 2 PowerPoint
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
chapt02_lecture from text
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
... • Gain or loss of electrons forms ions – Na atom loses an electron to become Na+ – Cl atom gains an electron to become Cl– – Opposite charges attract so that Na+ and Cl– remain associated as an ionic compound ...
Chemistry Note PowerPoint
... • An atom’s valance electrons are those that have the highest energy levels and are held most loosely. • The number of valance electrons determine many properties of that element, including the ways in which the atom combines with other atoms ...
... • An atom’s valance electrons are those that have the highest energy levels and are held most loosely. • The number of valance electrons determine many properties of that element, including the ways in which the atom combines with other atoms ...
Chemistry
... 95. A 78.0-g sample of an unknown compound contains 12.4 g of hydrogen. What is the percent by mass of hydrogen in the compound? ...
... 95. A 78.0-g sample of an unknown compound contains 12.4 g of hydrogen. What is the percent by mass of hydrogen in the compound? ...
1.Using the table above, decide if the element mercury (Hg) should
... electron deficiencies? One sentence for each is enough. You can draw structures but you don't need to. Aluminum chloride, Al2Cl6 This forms an intermolecular Lewis acid-base dimer where one Cl atom on each AlCl3 donates a pair of electrons to the neighboring Al atom. Boron trifluoride, BF3 This form ...
... electron deficiencies? One sentence for each is enough. You can draw structures but you don't need to. Aluminum chloride, Al2Cl6 This forms an intermolecular Lewis acid-base dimer where one Cl atom on each AlCl3 donates a pair of electrons to the neighboring Al atom. Boron trifluoride, BF3 This form ...
SUMMER WORK AP Chemistry
... nuclear charge, Z. (c) Use the relationship you derive in part (b) to predict the ground-state energy of the C5+ ion. 18. Write the Lewis symbol for atoms of each of the following elements: (a) Al, (b) Br, (c) Ar, (d) Sr. 19. Which of the following bonds are polar: (a) B – F, (b) Cl – Cl, (c) Se – O ...
... nuclear charge, Z. (c) Use the relationship you derive in part (b) to predict the ground-state energy of the C5+ ion. 18. Write the Lewis symbol for atoms of each of the following elements: (a) Al, (b) Br, (c) Ar, (d) Sr. 19. Which of the following bonds are polar: (a) B – F, (b) Cl – Cl, (c) Se – O ...
The Law of Definite Proportions
... The ratio of O to H in water is always 16:2 or 8:1. Example (2): KCl always contains one atom of K for every one atom of Cl In KCl, potassium and chlorine always have a ratio of “39.09 to 35.45” or “1.1 to 1” by mass. ...
... The ratio of O to H in water is always 16:2 or 8:1. Example (2): KCl always contains one atom of K for every one atom of Cl In KCl, potassium and chlorine always have a ratio of “39.09 to 35.45” or “1.1 to 1” by mass. ...
Molar Mass and Formulas
... Determine the molecular formula of benzopyrene if it has a molar mass of 252 g and an empirical formula of C5H3 • What is the empirical formula: C5H3 • Determine the molar mass of C5H3 ...
... Determine the molecular formula of benzopyrene if it has a molar mass of 252 g and an empirical formula of C5H3 • What is the empirical formula: C5H3 • Determine the molar mass of C5H3 ...
Final Exam Study Guide Page 1 Quiz
... b. 887.5 moles c. 1.5 x 1025 moles d. none of the above 2. How many grams are in 6.2 moles of NH4? a. .34 g b. 111.8 g c. 6.2 g d. 11.6 g 3. One mole of CaCO3 is equal to how many molecules of CaCO3? a. 765 molecules b. 249 molecules c. 7.6 x 1024 molecules d. 6.02 x 1023 molecules 4. How many grams ...
... b. 887.5 moles c. 1.5 x 1025 moles d. none of the above 2. How many grams are in 6.2 moles of NH4? a. .34 g b. 111.8 g c. 6.2 g d. 11.6 g 3. One mole of CaCO3 is equal to how many molecules of CaCO3? a. 765 molecules b. 249 molecules c. 7.6 x 1024 molecules d. 6.02 x 1023 molecules 4. How many grams ...
SEPARATION OF MATTER - Los Angeles City College
... symbolized as H+ or p+. • Neutrons: neutral particles with the same mass as the ...
... symbolized as H+ or p+. • Neutrons: neutral particles with the same mass as the ...
Analysis of a Matter
... symbolized as H+ or p+. • Neutrons: neutral particles with the same mass as the ...
... symbolized as H+ or p+. • Neutrons: neutral particles with the same mass as the ...
Ch6-Energy in Chemical Reactions-Chemical Reactions
... Chemists measure chemical in grams as the amount in the reaction. Therefore, we need a conversion factor to convert grams to atoms or molecules. Mole is the connection or the conversion factor between atoms and grams. Mole is just a large number 6.022 x 1023 for counting atoms like dozen -12 for co ...
... Chemists measure chemical in grams as the amount in the reaction. Therefore, we need a conversion factor to convert grams to atoms or molecules. Mole is the connection or the conversion factor between atoms and grams. Mole is just a large number 6.022 x 1023 for counting atoms like dozen -12 for co ...
document
... You need to keep track of how you are doing in the class and take action if you fall behind or have trouble with the material. A. Fellow students - meet others in the class. Even though you and the other student may be perplexed about a subject, you will find that talking together in the language of ...
... You need to keep track of how you are doing in the class and take action if you fall behind or have trouble with the material. A. Fellow students - meet others in the class. Even though you and the other student may be perplexed about a subject, you will find that talking together in the language of ...
chemistry_chapter_3
... If the numbers are both whole numbers, these will be the subscripts of the elements in the formula If the whole numbers are identical, substitute the number 1 Example: C2H2 and C8H8 have an empirical formula of CH If either or both numbers are not whole numbers, numbers in the ratio must be mu ...
... If the numbers are both whole numbers, these will be the subscripts of the elements in the formula If the whole numbers are identical, substitute the number 1 Example: C2H2 and C8H8 have an empirical formula of CH If either or both numbers are not whole numbers, numbers in the ratio must be mu ...
02_Lecture_Presentation
... • Atoms of the various elements differ in number of subatomic particles • An element’s atomic number is the number of protons in its nucleus • An element’s mass number is the sum of protons plus neutrons in the nucleus • Atomic mass, the atom’s total mass, can be approximated by the mass number ...
... • Atoms of the various elements differ in number of subatomic particles • An element’s atomic number is the number of protons in its nucleus • An element’s mass number is the sum of protons plus neutrons in the nucleus • Atomic mass, the atom’s total mass, can be approximated by the mass number ...
Chapter3 Solutions
... a number, derived from a formula that includes factors such as electron affinity and ionization potential, that indicates an element’s ability to attract electrons within a chemical bond. 2. (a) Zn−O, ∆EN = EN O − EN Zn = 3.44 − 1.65 = 1.79 (b) Mg−I, ∆EN = EN I − EN Mg = 2.66 − 1.31 = 1.35 (c) Co−Cl ...
... a number, derived from a formula that includes factors such as electron affinity and ionization potential, that indicates an element’s ability to attract electrons within a chemical bond. 2. (a) Zn−O, ∆EN = EN O − EN Zn = 3.44 − 1.65 = 1.79 (b) Mg−I, ∆EN = EN I − EN Mg = 2.66 − 1.31 = 1.35 (c) Co−Cl ...
Predicting Products online assistance #3
... 3. single replacement - an element replaces another in a compound. 4. double replacement - the elements in two compounds switch partners to form two new compounds. Writing Balanced Equations A chemical reaction is the process by which one or more substances are changed into one or more different sub ...
... 3. single replacement - an element replaces another in a compound. 4. double replacement - the elements in two compounds switch partners to form two new compounds. Writing Balanced Equations A chemical reaction is the process by which one or more substances are changed into one or more different sub ...
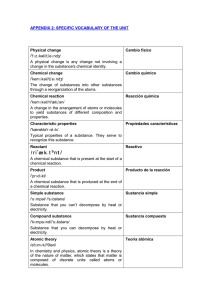
specific vocabulary of the unit
... The order of an element in Mendeleyev's table of the elements; equal to the number of protons in the nucleus or electrons in the neutral state of an atom of an element. Mass number Número másico /mæs//'nʌmbər / The sum of the number of neutrons and protons in an atomic nucleus. Atomic mass ...
... The order of an element in Mendeleyev's table of the elements; equal to the number of protons in the nucleus or electrons in the neutral state of an atom of an element. Mass number Número másico /mæs//'nʌmbər / The sum of the number of neutrons and protons in an atomic nucleus. Atomic mass ...
CHM100PracticeExam2
... A) the amount of a substance that contains as many elementary entities as there are atoms in exactly 12g of the N-14 isotope. B) a mass exactly equal to one-twelfth the mass of one C-12 atom. C) a mass of 1mole of units of a substance. D) the amount of a substance that contains as many elementary en ...
... A) the amount of a substance that contains as many elementary entities as there are atoms in exactly 12g of the N-14 isotope. B) a mass exactly equal to one-twelfth the mass of one C-12 atom. C) a mass of 1mole of units of a substance. D) the amount of a substance that contains as many elementary en ...