Atoms and Materials for Engineering
... connected to at least one other atom. The connections are made by atomic bonds. There are three important kinds of primary atomic bonds: 1) ionic 2) covalent 3) metallic. To really understand each kind, you would need to read many pages of explanation. So let us just try for some simple descriptions ...
... connected to at least one other atom. The connections are made by atomic bonds. There are three important kinds of primary atomic bonds: 1) ionic 2) covalent 3) metallic. To really understand each kind, you would need to read many pages of explanation. So let us just try for some simple descriptions ...
CHEMISTry is life - World of Teaching
... Precipitation reactions: When an insoluble solid called a precipitate forms when reactants are formed together. For example, when Carbon Dioxide is mixed with Calcium Hydroxide (limewater), the precipitate Calcium carbonate is formed. Ca(OH)2+CO2→H2O+Ca C03 Calcium Carbonate is found in chalk! ...
... Precipitation reactions: When an insoluble solid called a precipitate forms when reactants are formed together. For example, when Carbon Dioxide is mixed with Calcium Hydroxide (limewater), the precipitate Calcium carbonate is formed. Ca(OH)2+CO2→H2O+Ca C03 Calcium Carbonate is found in chalk! ...
X CHEMISTRY-1-2006 TIME-1½ MARKS 80 SECTION
... is soluble in water. The solution is tested with lit mns. Write only the word which will correctly complete each of the following sentences: a. If ‘x’ is a metal, then the litmust will turn _________. b. 2f ‘x’ is a non-mental, then the litmus will turn ________. c. If ‘x’ is a reactive metal, then ...
... is soluble in water. The solution is tested with lit mns. Write only the word which will correctly complete each of the following sentences: a. If ‘x’ is a metal, then the litmust will turn _________. b. 2f ‘x’ is a non-mental, then the litmus will turn ________. c. If ‘x’ is a reactive metal, then ...
CHEMISTRY Periodic Table of the Elements
... 1. Determine which element is the cation. The cations are usually found toward the left of the periodic table and are written first in the name and formula of a molecule. 2. Determine which element is the anion. The anions are non-metals (or hydrogen), located on the right side of the periodic table ...
... 1. Determine which element is the cation. The cations are usually found toward the left of the periodic table and are written first in the name and formula of a molecule. 2. Determine which element is the anion. The anions are non-metals (or hydrogen), located on the right side of the periodic table ...
1 - Montville.net
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
Balancing Chemical Equations Guided Inquiry (CC)
... A subscript is a small number that tells you how many atoms are in a compound. For example, in CaCl2 the two is the subscript and it tells us that there are two chloride ions bonded to one calcium. A coefficient tells us how many atoms or compounds there are, but in a different way. For example in t ...
... A subscript is a small number that tells you how many atoms are in a compound. For example, in CaCl2 the two is the subscript and it tells us that there are two chloride ions bonded to one calcium. A coefficient tells us how many atoms or compounds there are, but in a different way. For example in t ...
ionic bond. - cloudfront.net
... of two or more elements; at least one of these elements is a metal. • Alloys are important because the properties of an alloy are often superior to those of its component elements. ...
... of two or more elements; at least one of these elements is a metal. • Alloys are important because the properties of an alloy are often superior to those of its component elements. ...
F324 summary - Macmillan Academy
... • Condensation polymers have chemical groups that are vulnerable to chemical attack from either acids or alkalis – polyesters (ester group) and polyamides (amide group). This process is known as hydrolysis and results in the breakdown of the polymer. • Disposing of polymers is an environmental probl ...
... • Condensation polymers have chemical groups that are vulnerable to chemical attack from either acids or alkalis – polyesters (ester group) and polyamides (amide group). This process is known as hydrolysis and results in the breakdown of the polymer. • Disposing of polymers is an environmental probl ...
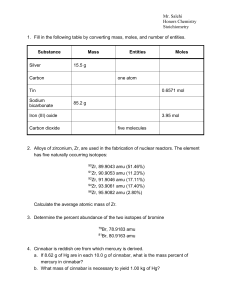
Stoichiometry - Cloudfront.net
... only carbon, hydrogen, and oxygen, determine the empirical formula of the unknown compound. 7. A 105.5 mg sample of a white substance is suspected to be cocaine, C 17H21NO4. The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white soli ...
... only carbon, hydrogen, and oxygen, determine the empirical formula of the unknown compound. 7. A 105.5 mg sample of a white substance is suspected to be cocaine, C 17H21NO4. The substance formed 279.3 mg of CO2 and 66.46 mg H2O on combustion. The compound contains 4.680% N by mass. Is the white soli ...
The Basics - I`m a faculty member, and I need web space. What
... • Name to Formula– Final compound must be neutral based on subscripts and charges • Magnesium Fluoride Mg2+ + F- MgF2 • Ammonium Sulfide NH4+ + S2- (NH4)2S • Tin(II) Carbonate Sn2+ + CO32- SnCO3 • Iron(III) Oxide Fe3+ + O2- Fe2O3 • Iron(II) Oxide Fe2+ + O2- FeO ...
... • Name to Formula– Final compound must be neutral based on subscripts and charges • Magnesium Fluoride Mg2+ + F- MgF2 • Ammonium Sulfide NH4+ + S2- (NH4)2S • Tin(II) Carbonate Sn2+ + CO32- SnCO3 • Iron(III) Oxide Fe3+ + O2- Fe2O3 • Iron(II) Oxide Fe2+ + O2- FeO ...
Unit 1: Building Blocks Homework
... Chlorine has a greater attraction than hydrogen for the bonded electrons in a hydrogen chloride molecule. What term is used to describe this type of covalent bond? ...
... Chlorine has a greater attraction than hydrogen for the bonded electrons in a hydrogen chloride molecule. What term is used to describe this type of covalent bond? ...
Chemistry IGCSE Revision PDF File
... If a metal is more reactive than hydrogen its ions stay in solution and hydrogen bubbles off ...
... If a metal is more reactive than hydrogen its ions stay in solution and hydrogen bubbles off ...
NOTES Atomic Structure Number Mass.docx
... all metals are malleable, shiny, conduct electricity, and are a similar color. Even where the colors were different, they could not be measured. So they tried organizing the elements based on something they could measure – atomic mass. Atomic mass is the relative average mass of an atom of the eleme ...
... all metals are malleable, shiny, conduct electricity, and are a similar color. Even where the colors were different, they could not be measured. So they tried organizing the elements based on something they could measure – atomic mass. Atomic mass is the relative average mass of an atom of the eleme ...
1 Mole
... reaction so SO4 can be treated like one atom: Mg(s) + CuSO4(aq) MgSO4(aq) + Cu(s) e.x. carbonate breaks apart so atoms must be balanced individually: CaCO3(aq) + HCl(aq) CaCl2(aq) + H2O(l) + CO2(g) ...
... reaction so SO4 can be treated like one atom: Mg(s) + CuSO4(aq) MgSO4(aq) + Cu(s) e.x. carbonate breaks apart so atoms must be balanced individually: CaCO3(aq) + HCl(aq) CaCl2(aq) + H2O(l) + CO2(g) ...
Chemical Bonding I
... summing the valence electrons of each atom. (Be sure to take ions into account!) 3) Distribute the electrons among the atoms giving octets to all atoms other than H (duet for it). 4) If any a ...
... summing the valence electrons of each atom. (Be sure to take ions into account!) 3) Distribute the electrons among the atoms giving octets to all atoms other than H (duet for it). 4) If any a ...
Sample Paper - Army Public School Jammu Cantt
... (a) the stability of Fe3+ in acid solution as compared to that of Cr3+ and Mn3+ (b) the ease with which iron can be oxidised as compared to the similar process for either Cr or Mn metals What can be inferred from the magnetic moment of the complex K4[Mn(CN)6] Magnetic moment: 2.2 BM? Describe the ty ...
... (a) the stability of Fe3+ in acid solution as compared to that of Cr3+ and Mn3+ (b) the ease with which iron can be oxidised as compared to the similar process for either Cr or Mn metals What can be inferred from the magnetic moment of the complex K4[Mn(CN)6] Magnetic moment: 2.2 BM? Describe the ty ...
Physical and Chemical Properties
... • The boiling point of an element or compound means the temperature at which the liquid form of an element or compound is at equilibrium with the gaseous form. • the boiling point of water is 100 degrees Celsius. ...
... • The boiling point of an element or compound means the temperature at which the liquid form of an element or compound is at equilibrium with the gaseous form. • the boiling point of water is 100 degrees Celsius. ...
Chemistry - Solutions
... chemical reaction – Chemical reaction: Breaking and forming of chemical bonds ...
... chemical reaction – Chemical reaction: Breaking and forming of chemical bonds ...
August 2010 Regents Exam part 1
... 5 An atom of which element has the largest atomic radius? Look on Table S (1) Fe (126) (2) Mg (160) (3) Si (132) (4) Zn (138) 6 Which element requires the least amount of energy to remove the most loosely held electron from a gaseous atom in the ground state? (lowest 1st Ionization energy) (1) bromi ...
... 5 An atom of which element has the largest atomic radius? Look on Table S (1) Fe (126) (2) Mg (160) (3) Si (132) (4) Zn (138) 6 Which element requires the least amount of energy to remove the most loosely held electron from a gaseous atom in the ground state? (lowest 1st Ionization energy) (1) bromi ...